More Related Content
Similar to Hu Posterboard Presentation 081011
Similar to Hu Posterboard Presentation 081011 (20)
Hu Posterboard Presentation 081011
- 1. RESEARCH POSTER PRESENTATION DESIGN © 2011
www.PosterPresentations.com
• We study solution unimer plus
aggregates (P3HT, P3OT, and P3BT) that
are controlled by inducing in solutions
• Good Solvent solubilizes polythiophene
• Bad Solvent induces formation of
aggregates
• Control of aggregate formation by
volume ratio
• Characterize unimer and aggregates of
good/bad solvents by optical
spectroscopy of UV-Absorption spectrum
and photoluminescent spectrum
ABSTRACT
• Dissolve P3HT, P3OT, and P3BT in Chloroform (Good
Solvent)
• Add control amount of bad solvents (acetone,
acetonitrile, dimethylformamide, ethanol, hexane,
methanol, and tetrahydrofuran)
• Measure UV-Vis absorption spectrum and
photoluminescent spectrum
– Band Gap
– HOMO-LUMO (High Occupied Molecular Orbital
and Low Unoccupied Molecular Orbital)
– π – π stacking
– Energy Diagrams:
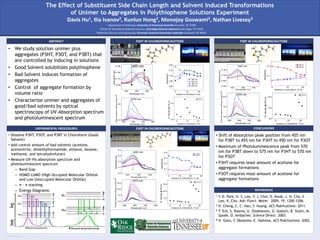
EXPERIMENTAL PROCEDURES P3OT IN CHLOROFORM/ACETONE
P3BT IN CHLOROFORM/ACETONE
CONCLUSIONS
• Shift of Absorption peak position from 455 nm
for P3BT to 455 nm for P3HT to 450 nm for P3OT
• Maximum of Photoluminescence peak from 570
nm for P3BT down to 575 nm for P3HT to 570 nm
for P3OT
• P3HT requires least amount of acetone for
aggregate formations
• P3OT requires most amount of acetone for
aggregate formations
REFERENCES
1 Y. D. Park, H. S. Lee, Y. J. Choi, D. Kwak, J. H. Cho, S.
Lee, K. Cho, Adv. Funct. Mater. 2009, 19, 1200-1206.
2 H. Cheng, C. C. Han, Y. Huang, ACS Publications. 2011.
3 T. Erb, S. Raleva, U. Zhokhavets, G. Gobsch, B. Stuhn, M.
Spode, O. Ambacher, Science Direct. 2003.
4 H. Goto, Y. Okamoto, E. Yashima, ACS Publications. 2002.
The Effect of Substituent Side Chain Length and Solvent Induced Transformations
of Unimer to Aggregates in Polythiophene Solutions Experiment
1 Department of Chemistry, University of Tennessee-Knoxville Knoxville, TN 37996
2Center for Nanophase Materials Sciences, Oak Ridge National Laboratory Oak Ridge, TN 37830
3Materials Sciences and Engineering, Tennessee Technical University-Cookeville Cookeville, TN 38505
Davis Hu1, Ilia Ivanov2, Kunlun Hong2, Monojoy Goswami2, Nathan Livesey3
400 500 600
0.076
0.114
0.152
0.190
Absorbance(au)
Wavelength (nm)
450 nm
0 10 20
0.00
0.05
0.10
0.15
0.20
455 nm
520 nm
560 nm
605 nm
Absorbance
Acetone in Chloroform, % wt.
500 600 700 800
0.0
5.0
PhotoluminescenceIntensity(counts)x10
6
Wavelength (nm)
575 nm
0 10 20
0.0
5.0
575 nm
PLIntensity@575nm
Acetone in Chloroform, % wt
400 500 600
0.0
0.1
0.2
0.3
0.4
Absorbance(au)
Wavelength (nm)
450 nm
500 600 700 800
0.0
5.0
10.0
15.0
PhotoluminescenceIntensity(counts)x106
Wavelength (nm)
573 nm
0 10 20 30
5.0
10.0
15.0
575 nm
PLIntensity@575nm
Acetone in Chloroform, % wt
0 10 20 30
0.1
0.2
0.3
0.4
448 nm
550 nm
Absorbance
Acetone in Chloroform, % wt.
400 500 600
0.000
0.063
0.126
0.189
Absorbance(au)
Wavelength (nm)
450 nm
500 600 700 800
0.0
5.0
10.0
PhotoluminescenceIntensity(counts)x106
Wavelength (nm)
570 nm
0 10 20
0.1
0.2
450 nm
565 nm
610 nm
Absorbance
Acetone in Chloroform, % wt.
0 10 20
0.0
5.0
10.0
570 nm
PLIntensity@570nm
Acetone in Chloroform, % wt
P3HT IN CHLOROFORM/ACETONE