Research projects in synthetic organic and analytical chemistry dr. khan (2006 2015)
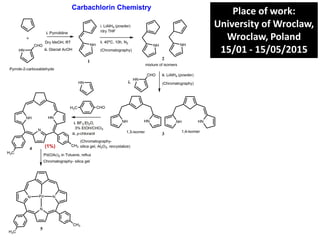
- 1. Place of work: University of Wroclaw, Wroclaw, Poland 15/01 - 15/05/2015 (1%)
- 2. 21 5, 20 o- tol p-Me 7, 8 17, 18 12, 13 2, 3 NH m- tol Figure. 1H NMR spectrum of 10,15-Ditolyl-21-carbachlorin 4 in CDCl3. HR-MS(ESI+): m/z calcd for C35H29N3+ H+: 492.2440; found 492.2460
- 3. 3 Modified cucurbit[n]urils: Synthesis and supramolecular interactions Ph.D. Defense Presentation Date: 1st October 2012 Place of work: Department of Chemistry, Masaryk University Brno, Czech Republic 09/2006 - 09/2012
- 4. 4 Chemical structure of cucurbit[n]uril and the reactive sites of it’s modification? Sites for functionalization CB[n]: Cucurbit[n]uril (n = 5-8, 10) Glycoluril (Building block of CB[n]) Cucurbit[6]uril Peripheral (equatorial) position Methylene bridge (axial) position , 100°C, 24 h 1. Kim, J.; et. al. J. Am. Chem. Soc. 2000, 122, 540-541. 2
- 5. 5 1. Extremely low solubility in common organic/aqueous solvents (except for aqueous solution of strong acids). 2. Problem of accessing CB[n] derivatives/analogues by tailor-made synthetic procedures What are the limitations in the CB[n] chemistry ? 1. To prepare functionalized CB[n] compounds. 2. To improve their solubility in both aqueous and organic solvents. 3. To access their wide range of applications. Why is modification of CB[n] compounds necessary ?
- 6. 6 Methods of preparing functionalized cucurbit[n]uril: Three possible routes 2. Kim, K.; et. al. Chem. Soc. Rev., 2007, 36, 267-279. Route 1 Route 2 Direct method of functionalization Indirect method of functionalization Route 3 Indirect method of functionalization Substitution at the peripheral position Substitution at the CH2 bridge position (so far found to be unsuccessful) Substitution at the peripheral position
- 7. 7 Research aims of this dissertation work Two major projects Project 1 Synthesis and separation of new monosubstituted CB[6] compounds modified at the CH2 bridge (axial) position. Project 2 Synthesis, purification and supramolecular interactions of a water soluble CB[6] derivative, namely, hexamethylcucurbit[6]uril (MeCB[6]). Here, different pyridinium guests (G1, G3, G4) were used to study their supramolecular complexations with the MeCB[6] macrocycle both in the solution and solid state.
- 8. Scheme. Reaction Scheme for Synthesis of Monosubstituted CB[6] Figure. X-ray crystal structure of Monosubstituted CB[6] 3. L. Gilberg, M. Shamsul Azim Khan, M. Enderesova, V.Sindelar. Organic Letters, 16 (9), 2446-2449, 2014. Project 1: Synthesis of monosubstituted CB[6] (0.2%)
- 9. N N NN O O N N NN O O C H C H2 5 1 I-01 3. L. Gilberg, M. Shamsul Azim Khan, M. Enderesova, V.Sindelar. Organic Letters, 16 (9), 2446-2449, 2014.
- 10. 10 Project 2: Synthesis of hexamethylcucurbit[6]uril (MeCB[6]) and its supramolecular interactions with different pyridinium guests 4. Khan, M.S.A.; Heger, D., Necas, M.; Sindelar, V. J. Phys. Chem B. 2009, 113, 11054-11057. X-ray crystal structure of MeCB[6]•acetone•19H2O complex The “acetone molecule” was not possible to remove completely from MeCB[6] cavity even after drying at 100 °C for 24 h.
- 11. 11 Figure. 1H NMR spectrum (500 MHz, D2O) of pure hexamethylcucurbit[6]uril, MeCB[6]. 4. Khan, M.S.A.; Heger, D., Necas, M.; Sindelar, V. J. Phys. Chem B. 2009, 113, 11054-11057.
- 12. 12 Supramolecular interactions of MeCB[6] with pyridinium guests (G1, G3, G4) Two different modes of interaction between MeCB[6] and the pyridinium guests were studied 1. Binding mode in the solid state (using X-ray crystallography) 2. Binding mode in the solution using a) 1H NMR titration method b) UV-vis spectrophotometric titration method
- 13. 13 Supramolecular interactions of MeCB[6] with methylviologen (MeV2+ = G1) guest 4. Khan, M.S.A.; Heger, D., Necas, M.; Sindelar, V. J. Phys. Chem B. 2009, 113, 11054-11057. Binding mode between MeCB[6] and MeV2+in the pure D2O solution: 1H NMR fast exchange kinetics NN CH3 CH3 + + Crystal preparation of MeCB[6]•MeV2+•8H2O complex Slow evaporation of aqueous solution of the host and guest in 1:1 ratio resulted X-ray quality single crystals. Binding mode between MeCB[6] and MeV2+in solid state: X-ray crystallography - wireframe model Methyl protons of MeV2+ (red ●), MeCB[6] (green ▲), and acetone (blue ♦). α β β α β α α β (A) (B) (C) Free MeV2+ 0.5 : 1 1 : 1 MeCB[6]: MeV2+
- 14. 14 Determination of binding constants (K / M-1) for the complexation between MeCB[6] and methylviologen (MeV2+) guest : UV-vis spectrophotometric titration method 4. Khan, M.S.A.; Heger, D., Necas, M.; Sindelar, V. J. Phys. Chem B. 2009, 113, 11054-11057. Figure. Electronic absorption spectrum of the complexation between MeV2+ and MeCB[6] in pure water. P S I - P l o t W o r k i n g D e m w w w . p o l y s o f t w a r e . c o m P S I - P l o t W o r w w w . p o l y s o f P S I - P l o t W o r k i n g D e m w w w . p o l y s o f t w a r e . c o m P S I - P l o t W o r w w w . p o l y s o f P S I - P l o t W o r k i n g w w w . p o l y s o f t w a r e P S I - P l o t w w w . p o l P S I - P l o t W o r k i n g w w w . p o l y s o f t w a r e P S I - P l o t w w w . p o l Legend 0.1mM NaCl fit1 0.5mM fit2 1mM fit3 3mM fit4 5mM fit5 10mM fit6 20mM fit7 P S I - P l o t W o r k i n g D e m o w w w . p o l y s o f t w a r e . c o m P S I - P l o t W o r k i n g D e m o w w w . p o l y s o f t w a r e . c o m P S I - P l o t W o r k i n g D e m o w w w . p o l y s o f t w a r e . c o m P S I - P l o t W o r k i n g D e m o w w w . p o l y s o f t w a r e . c o m P S I - P l o t W o r k i n g D e m o w w w . p o l y s o f t w a r e . c o m P S I - P l o t W o r k i n g D e m o w w w . p o l y s o f t w a r e . c o m P S I - P l o t W o r k i n g D e m o w w w . p o l y s o f t w a r e . c o m P S I - P l o t W o r k i n g D e m o w w w . p o l y s o f t w a r e . c o m c(MeCB[6]) in mol / L 0.0000 0.0001 0.0002 0.0003 0.0004 Absorbanceat257nm(a.u.) 0.40 0.45 0.50 0.55 0.60 0.65 0.70 0.75 0.80 Effect of NaCl on the binding constant K Figure. Fitting of the absorbance data for the 1:1 complex formation between MeV2+ and MeCB[6]. 257 nm K = (2.05 ± 0.21) × 105 M-1 MeCB[6] concentration increases
- 15. Table: Values of Association Constants of the MeCB[6]-MeV2+ Complex (KMeCB[6]) and the CB[7]-MeV2+ Complex (KCB[7]): UV-vis Spectrophotometric and 1H-NMR Titration methods 4. Khan, M.S.A.; Heger, D., Necas, M.; Sindelar, V. J. Phys. Chem B. 2009, 113, 11054-11057. NaCl/mM KMeCB[6] / M-1 KCB[7] / M-1 0 (1.23 ± 0.15) × 105c Ndd 0 (2.05 ± 0.21) × 105 (1.68 ± 0.30) × 106 0.1 (1.97 ± 0.13) × 105 ndd 0.2 (1.03 ± 0.06) × 105 ndd 0.4 (6.85 ± 0.43) × 104 ndd 0.5 (7.17 ± 0.45) × 104 ndd 0.8 (3.15 ± 0.28) × 104 ndd 1 (2.84 ± 0.31) × 104 ndd 2 (2.47 ± 0.21) × 104 ndd 3 (1.17 ± 0.12) × 104 ndd 5 (3.76 ± 0.35) × 103 ndd 10 (2.15 ± 0.20) × 103 (8.3 ± 1.8) × 105 20 (7.6 ± 1.4) × 102 ndd 30 (3.02 ± 0.55) × 102 ndd 40 (1.25 ± 0.63) × 102 ndd 50 (1.00 ± 0.67) × 102 (3.45 ± 0.52) × 105 100 nbb (1.49 ± 0.16) × 105 200 (6.0 ± 2.5) × 101c (9.4 ± 1.0) × 104 a The standard deviation of the fit is given for each measurement. b K not obtained by UV-vis spectroscopy. c Determined by 1H NMR titration. d Not measured. CB[7]•MeV2+ complex NN CH3 CH3 + + MeCB[6]•MeV2+ complex > 2000-fold decrease only 5-fold decrease
- 16. 16 Table: Values of Association Constants (K/M-1): UV-vis Spectrophotometry Unpublished results: KMeCB[6]●G3 and KMeCB[6]●G4 5. Kolman, V.; Khan, M.S. A.; Babinský, M.; Marek, R.; Sindelar, V. Org. Lett. 2011, 13, 6148-6151. K / M-1 NaCl / M MeCB[6]∙G1 MeCB[6]∙G3 MeCB[6]∙G4 0 (2.05 ± 0.21) × 105 (3.47 ± 1.95) × 107 (9.79 ± 3.63) × 107 0.005 (3.76 ± 0.35) × 103 (7.41 ± 1.78 ) × 106 - 0.01 (2.15 ± 0.2) × 103 (2.04 ± 1.32) × 106 (1.65 ± 1.51) × 107 0.02 (7.60 ± 1.40) × 102 (1.05 ± 1.30) × 106 (2.28 ± 1.05) × 106 0.03 (3.02 ± 0.55) × 102 (4.79 ± 1.23) × 105 (1.15 ± 1.12) × 106 0.04 (1.25 ± 0.63) × 102 (2.75 ± 1.17) × 105 (1.08 ± 1.2) × 106 0.05 (1.0 ± 0.67) × 102 (1.95 ± 1.12) × 105 (4.19 ± 1.23) × 105 (2.10 ± 0.20) × 106 * 0.1 n.d (6.46 ± 1.40) × 104 (3.09 ± 1.35) × 105 0.2 (6.0 ± 2.5) × 101 ** (4.20 ± 2.0) × 104 (7.88 ± 1.26) × 104 0.3 - (3.31 ± 1.82) × 104 (8.8 ± 1.12) × 103 * value for the complex with CB[6], ** 1H NMR titration method Figure. Structures of host (MeCB[6]) and guests (G1, G3, G4) M-1 : 2.1 × 105 < 3.5 × 107 < 9.8 × 107 Extra stabilized: one hydrogen bond (+N-H···O), ion-dipole interactions Extra stabilized due to hydrophobicity of ethylene moiety Different binding mode of pyridinium guests inside MeCB[6] cavity
- 17. Collaborative project: Deprotection of propylene bridged glycoluril dimer 6. Stancl, M.; Khan, M.S.A.; Sindelar, V. Tetrahedron, 2011, 67, 8937-8941. Figure. Chemical structures of mono- (A), di- (B), tri- (C) and tetra- (D) hydroxy- bis(propylene)glycoluril dimers. N N NN O O N N NN O O NH N NNH O O N NH NHN O O K2 S2 O8 / H2 O (Glycoluril dimer, Yield: 37.4 %)Bis(propylene)glycoluril dimer N2 atm / 78°C, 6h Characterization: 1H, 13C NMR (d6-DMSO) spectroscopy and HRMS (ESI+)
- 18. 0,1 0,15 0,2 0,25 0,3 0,35 0 0,00001 0,00002 0,00003 0,00004 0,00005 Conc. of CB6 / M Absorbanceat317nm CB6·2 complex (1:1) at 0.05 M NaCl K = (2.1 ± 0.2) × 106 M-1 UV-vis titration method Figure. 1H NMR spectra (300 MHz, 0.05 M NaCl-D2O) 5. Kolman, V.; Khan, M.S. A.; Babinský, M.; Marek, R.; Sindelar, V. Org. Lett. 2011, 13, 6148-6151. Guest G4 0.5 : 1 1 : 1 CB6●2 Slow exchange process in the 1H NMR time scale Collaborative project: Supramolecular shuttle based on CB6 and guest 2 (BPE) Figure. Structures of synthesized host (CB6) and guest 2 (BPE).
- 19. Calix[n]phyrin chemistry Place of work: (current position- Postdoctoral fellow) University of Chemical Technology Prague, Czech Republic 05/2013 - 06/2015
- 20. 1. Synthesis of calix[n]phyrin conjugates with Tröger's bases (TB). 2. sensing of cations /anions of the synthesized compounds. Research goals Calix[4]phyrin-TB conjugate V shape
- 22. Tb Tc Td Te Cb Cc Cd Ce Ta Ca Tb Cb Tc Cc Cd Ce Td Te Tf Cf Figure 2: 1H NMR spectrum of Bis(nitro)calix[4]phyrin, 2a (cis-isomer, green) and 2b (trans-isomer, red) in CDCl3.
- 23. Synthesis of TB-calix[4]phyrin and TB-calix[6]phyrin
- 24. 2 x NH 18 x CH: TB 8 x CH: pyrrole 6 x CH2: CB 4 x CH3 3 x NH 27 x CH: TB 12 x CH: pyrrole 9 x CH2: TB 6 x CH3 A) TB-calix[4]phyrin, 8a B) TB-calix[6]phyrin, 8b Compound 8a: HRMS-ESI+: for [M+H]+ (C62H53N8) calcd: 909.44; found 909.44 and for [M+2H]2+ (C62H54N8) calcd: 455.22; found 455.22 Compound 8b: HRMS-ESI+: for [M+H]+ (C93H79N12) calcd: 1364.66; found 1364.66 and for [M+2H]2+ (C93H80N12) calcd: 682.83; found 682.83
- 25. Tris-mannose methylamine Supramolecular object Construction of supramolecular host from TB
- 26. Synthesis of tris-mannose cluster from D(+)-mannose
- 28. Cyclodextrin chemistry - dimerization of cyclodextrins Place of work: (Position- Research assistant) Institute of Organic Chemistry and Biochemistry, Prague, Academy of Sciences of the Czech Republic 08/2011 - 12/2012
- 29. Concept: Duplex Cyclodextrins Connected with Reversible Linkages Guest Duplex CD Cleavage by external stimuli Guest release
- 30. 2 R1–SH H2OO2 pH ≥ 7 R2–S R1–S–S–R1 + R1–S + R1–S–S–R2 Concept: Duplex Cyclodextrins Connected with Disulfide Linkages
- 31. • Synthesis of cyclodextrin homoduplexes and heteroduplexes linked by disulfide bonds Cyclodextrin Duplexes Linked by Disulfide Bonds - Outline • Dynamic exchange of building blocks in the presence of a guest (template effect) • Thermodynamic stabilities of inclusion complexes with organic molecules • Cleavage of disulfide bonds by reducing thiols • Applications in supramolecular / material chemistry
- 32. 5 steps, 69 % 5 steps, 65 % J. Org. Chem. 2009, 74, 1082. α-CD O OH OHO O OH OH AcS O O OH OH OH O O O OH OH O OH OH SAcO OOH OH OH O OH OH β-CD O OH OH O O OH OHAcS O O OH OH OH O O OH OH OOH OH SAc O O OH OH OH O OH OH O OHOH OH O O Starting Cyclodextrin Disulfanyl Building Blocks Figure. Gradient parameters for the elution of the mixture reaction of β-cyclodextrin heteroduplex formation (volume of each collected fraction was 39 ml). = =
- 35. Synthesis of 3-hydroxyadamantane-1-acetamidohexanol in dry DMF Figure. Chromatograms of βCD- heteroduplex: A) in equimolar mixture with the template (guest), and for the synthesis of βCD- heteroduplex B) without template, and C) with template. The solvent system in HPLC analysis was 22-60% MeOH-water (30 min). A B C Host : template; 1:1 Host: no template Host: with template
- 37. Dimerization of Bifunctional Cyclodextrins by Oxidation of Sulfanyl Groups [O]
- 38. Duplexes with Cleavable Bonds for Drug Delivery
- 39. Monolayers on Glass Surfaces Using Cyclodextrin Hetroduplexes as Divalent Connectors
- 40. 5.7 Å 7.8 Å ~ 14 Å Monolayers on Glass Surfaces Using Cyclodextrin Hetroduplexes as Divalent Connectors
- 41. Si / glass H2SO4 H2O2 activation α β (15 min, drops) SAM: preparation μCP (5 min, 3x5 µm, lines) Rh-Ad3
- 42. lissamin-Ad3 (Rh-Ad3) excitace: 510-550 nm emise: 590 nm (červená) 3.5 μM, voda/EtOH (1:1) Fluorescent Trivalent Ligand
- 44. Duplexes for Selective Sensors
