Presentation1.pptx
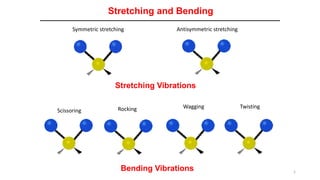
- 1. 1 Stretching and Bending Symmetric stretching Antisymmetric stretching Scissoring Rocking Wagging Twisting Bending Vibrations Stretching Vibrations
- 2. 2 Q1) What is the correct order of stretching frequency of C=O bond for the following compounds? Ans. (i) > (iii) > (ii) In (ii) lone pair of electrons from oxygen as well as alkene double bond electrons can be delocalized to carbonyl double bond and will decrease its bond order significantly. In (iii), oxygen only oxygen lone pairs can be delocalized to carbonyl double bond. So in this case C=O bond order will be higher than (ii) case. Finally, in (i), oxygen lone pairs can be delocalized into alkene double bond as well as carbonyl double bond, therefore, C=O in (i) will have highest bond order and thus stretching frequency.
- 3. 3 Q2) What is the correct order of stretching frequency of C=O bond for the following compounds? Ans. (i) > (iii) > (ii) Explanation similar to question no. 1
- 4. 4 Q3) What is the correct order of stretching frequency of C=O bond for the following compounds? Ans. (iii) > (i) > (ii) Because of the resonance, C=O bond will get partial double bond character in all the three cases with a positive charge on carbon at para position. OMe (electron donating) will stabilize the positive charge, whereas, nitro group will destabilize the positive charge. Therefore, bond strength of C=O will be highest in (iii) and least in case of (ii).
- 5. 5 Q4) What is the correct order of stretching frequency of C=O bond for the following compounds? Ans. (iv) > (iii) > (ii) > (i) External C=O double bond give an increase in absorption frequency with decreasing ring size. Smaller rings require the use of more p character to make the ring C-C bonds form the requisite small angles (recall the trend: sp = 180˚, sp2 = 120˚, sp3 = 109˚, sp>3 = < 109˚). This removes the p character from the sigma bond of the external double bond (C=O) but gives it more s character, thus strengthening and stiffening the double bond. The force constant K is then increased, and the absorption frequency increases.
- 6. 6 C5H10O
- 7. 7 C6H12O
- 8. 8
- 9. 9
- 10. 10 For CnHn+r For CnHn+r o For CnHn+r Nx or CnHn+r NxO Hydrogen deficiency: [n-(r-x)]+2 2 For CnHn+r Xx or CnHn+r XxO (X: F, Cl, Br, I) Hydrogen deficiency: [n-(r+x)]+2 2 Hydrogen deficiency Index