33-09_ Infective Endocarditis.pdf
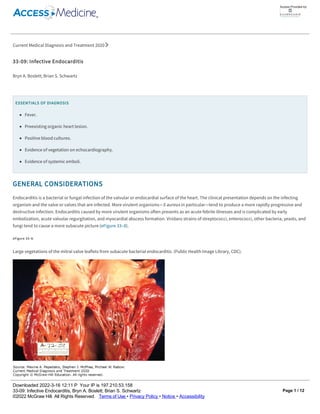
- 1. Current Medical Diagnosis and Treatment 2020 3309: Infective Endocarditis Bryn A. Boslett; Brian S. Schwartz ESSENTIALS OF DIAGNOSIS Fever. Preexisting organic heart lesion. Positive blood cultures. Evidence of vegetation on echocardiography. Evidence of systemic emboli. GENERAL CONSIDERATIONS Endocarditis is a bacterial or fungal infection of the valvular or endocardial surface of the heart. The clinical presentation depends on the infecting organism and the valve or valves that are infected. More virulent organisms—S aureus in particular—tend to produce a more rapidly progressive and destructive infection. Endocarditis caused by more virulent organisms often presents as an acute febrile illnesses and is complicated by early embolization, acute valvular regurgitation, and myocardial abscess formation. Viridans strains of streptococci, enterococci, other bacteria, yeasts, and fungi tend to cause a more subacute picture (eFigure 33–8). eFigure 33–8. Large vegetations of the mitral valve leaflets from subacute bacterial endocarditis. (Public Health Image Library, CDC). Underlying valvular disease, less common than in the past, is present in about 50% of cases. Valvular disease alters blood flow and produces jet effects that disrupt the endothelial surface, providing a nidus for attachment and infection of microorganisms that enter the bloodstream. Predisposing valvular abnormalities include rheumatic involvement of any valve, bicuspid aortic valves, calcific or sclerotic aortic valves, hypertrophic subaortic stenosis, mitral valve prolapse, and a variety of congenital disorders such as ventricular septal defect, tetralogy of Fallot, coarctation of the aorta, or patent ductus arteriosus. Rheumatic disease is no longer the major predisposing factor in developed countries. Regurgitation lesions are more Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 1 / 12
- 2. Underlying valvular disease, less common than in the past, is present in about 50% of cases. Valvular disease alters blood flow and produces jet effects that disrupt the endothelial surface, providing a nidus for attachment and infection of microorganisms that enter the bloodstream. Predisposing valvular abnormalities include rheumatic involvement of any valve, bicuspid aortic valves, calcific or sclerotic aortic valves, hypertrophic subaortic stenosis, mitral valve prolapse, and a variety of congenital disorders such as ventricular septal defect, tetralogy of Fallot, coarctation of the aorta, or patent ductus arteriosus. Rheumatic disease is no longer the major predisposing factor in developed countries. Regurgitation lesions are more susceptible than stenotic ones. The initiating event in native valve endocarditis is colonization of the valve by bacteria or yeast that gain access to the bloodstream. Transient bacteremia is common during dental, upper respiratory, urologic, and lower gastrointestinal diagnostic and surgical procedures. It is less common during upper gastrointestinal and gynecologic procedures. Intravascular devices are increasingly implicated as a portal of access of microorganisms into the bloodstream. A large proportion of cases of S aureus endocarditis are attributable to health care–associated bacteremia. Native valve endocarditis is usually caused by S aureus, viridans streptococci, enterococci, or HACEK organisms (an acronym for Haemophilus aphrophilus [now Aggregatibacter aphrophilus], Actinobacillus actinomycetemcomitans [now Aggregatibacter actinomycetemcomitans], Cardiobacterium hominis, Eikenella corrodens, and Kingella species). Streptococcal species formerly accounted for the majority of native valve endocarditis cases; S aureus is now the leading cause. Gramnegative organisms and fungi account for a small percentage. In injection drug users, S aureus accounts for over 60% of all endocarditis cases and for 80–90% of cases in which the tricuspid valve is infected. Enterococci and streptococci comprise the balance in about equal proportions. Gramnegative aerobic bacilli, fungi, and unusual organisms may cause endocarditis in injection drug users. The microbiology of prosthetic valve endocarditis also is distinctive. Early infections (ie, those occurring within 2 months after valve implantation) are commonly caused by staphylococci—both coagulasepositive and coagulasenegative—gramnegative organisms, and fungi. In late prosthetic valve endocarditis, streptococci are commonly identified, although coagulasenegative and coagulasepositive staphylococci still cause many cases. CLINICAL FINDINGS A. Symptoms and Signs Virtually all patients have fever at some point in the illness, although it may be very low grade (less than 38°C) in elderly individuals and in patients with heart failure or kidney failure. Rarely, there may be no fever at all. The duration of illness typically is a few days to a few weeks. Nonspecific symptoms are common. The initial symptoms and signs of endocarditis may be caused by direct arterial, valvular, or cardiac damage (eFigure 33–9). Although a changing regurgitant murmur is significant diagnostically, it is the exception rather than the rule. Symptoms also may occur as a result of embolization, metastatic infection or immunologically mediated phenomena. These include cough; dyspnea; arthralgias or arthritis; diarrhea; and abdominal, back, or flank pain. eFigure 33–9. Subacute bacterial endocarditis. Calcific embolus impacted in arteriole below the disk, producing a distal area of retinal infarction. (Reproduced, with permission, from Vaughan DG, Asbury T, RiordanEva P [editors]. General Ophthalmology, 15th ed. Originally published by Appleton & Lange. Copyright © 1999 by The McGrawHill Companies, Inc.) Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 2 / 12
- 3. eFigure 33–9. Subacute bacterial endocarditis. Calcific embolus impacted in arteriole below the disk, producing a distal area of retinal infarction. (Reproduced, with permission, from Vaughan DG, Asbury T, RiordanEva P [editors]. General Ophthalmology, 15th ed. Originally published by Appleton & Lange. Copyright © 1999 by The McGrawHill Companies, Inc.) The characteristic peripheral lesions—petechiae (on the palate or conjunctiva or beneath the fingernails), subungual (“splinter”) hemorrhages (Figure 33–4), Osler nodes (painful, violaceous raised lesions of the fingers, toes, or feet) (Figure 33–5), Janeway lesions (painless erythematous lesions of the palms or soles), and Roth spots (exudative lesions in the retina)—occur in about 25% of patients (eFigures 33–10 and 33–11). Strokes and major systemic embolic events are present in about 25% of patients and tend to occur before or within the first week of antimicrobial therapy. Hematuria and proteinuria may result from emboli or immunologically mediated glomerulonephritis, which can cause kidney dysfunction. Figure 33–4. Splinter hemorrhages appearing as red lineal streaks under the nail plate and within the nail bed, in endocarditis, psoriasis, and trauma. (Reproduced, with permission, from Richard P. Usatine, MD.) Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 3 / 12
- 4. Figure 33–4. Splinter hemorrhages appearing as red lineal streaks under the nail plate and within the nail bed, in endocarditis, psoriasis, and trauma. (Reproduced, with permission, from Richard P. Usatine, MD.) Figure 33–5. Osler node causing pain within the pulp of the big toe and multiple painless flat Janeway lesions over the sole of the foot. (Used, with permission, from David A. Kasper, DO, MBA, in Usatine RP, Smith MA, Mayeaux EJ Jr, Chumley H, Tysinger J. The Color Atlas of Family Medicine. McGrawHill, 2009.) Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 4 / 12
- 5. Figure 33–5. Osler node causing pain within the pulp of the big toe and multiple painless flat Janeway lesions over the sole of the foot. (Used, with permission, from David A. Kasper, DO, MBA, in Usatine RP, Smith MA, Mayeaux EJ Jr, Chumley H, Tysinger J. The Color Atlas of Family Medicine. McGrawHill, 2009.) eFigure 33–10. Roth spots are retinal hemorrhages with white centers found in infective endocarditis. (From Paul D. Comeau; Reproduced, with permission, from Usatine RP, Smith MA, Mayeaux EJ Jr, Chumley H, Tysinger J. The Color Atlas of Family Medicine. McGrawHill, 2009.) Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 5 / 12
- 6. eFigure 33–10. Roth spots are retinal hemorrhages with white centers found in infective endocarditis. (From Paul D. Comeau; Reproduced, with permission, from Usatine RP, Smith MA, Mayeaux EJ Jr, Chumley H, Tysinger J. The Color Atlas of Family Medicine. McGrawHill, 2009.) eFigure 33–11. Close up of a Roth spot, actually a cotton wool spot (from ischemic bursting of axons) surrounded by hemorrhage (from ischemic bursting of an arteriole). (From Paul D. Comeau; Reproduced, with permission, from Usatine RP, Smith MA, Mayeaux EJ Jr, Chumley H, Tysinger J. The Color Atlas of Family Medicine. McGrawHill, 2009.) B. Imaging Chest radiograph may show evidence for the underlying cardiac abnormality and, in rightsided endocarditis, pulmonary infiltrates. The electrocardiogram is nondiagnostic, but new conduction abnormalities suggest myocardial abscess formation. Echocardiography is useful in identifying vegetations and other characteristic features suspicious for endocarditis and may provide adjunctive information about the specific valve or valves that are infected. The sensitivity of transthoracic echocardiography is between 55% and 65%; it cannot reliably rule out endocarditis but may confirm a clinical suspicion. Transesophageal echocardiography is 90% sensitive in detecting vegetations and is particularly useful for identifying valve ring abscesses as well as prosthetic valve endocarditis. Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 6 / 12
- 7. B. Imaging Chest radiograph may show evidence for the underlying cardiac abnormality and, in rightsided endocarditis, pulmonary infiltrates. The electrocardiogram is nondiagnostic, but new conduction abnormalities suggest myocardial abscess formation. Echocardiography is useful in identifying vegetations and other characteristic features suspicious for endocarditis and may provide adjunctive information about the specific valve or valves that are infected. The sensitivity of transthoracic echocardiography is between 55% and 65%; it cannot reliably rule out endocarditis but may confirm a clinical suspicion. Transesophageal echocardiography is 90% sensitive in detecting vegetations and is particularly useful for identifying valve ring abscesses as well as prosthetic valve endocarditis. C. Diagnostic Studies 1. Blood cultures Three sets of blood cultures are recommended before starting antibiotics to maximize microbiologic diagnosis. To maximize the yield of blood cultures, adequate volume is important. Each culture bottle should be filled with 10 mL of blood since half of adults have less than 1 colony forming unit of bacteria per mL blood. The yield of bacteria may be up to 5% higher for every additional milliliter collected. Optimal yield is with two or three sets of cultures from different sites. There is no difference in yield if blood is collected simultaneously or several hours apart. Approximately 5% of cases will be culturenegative, usually attributable to administration of antimicrobials prior to cultures. If antimicrobial therapy has been administered prior to obtaining cultures and the patient is clinically stable, it is reasonable to withhold antimicrobial therapy for 2–3 days so that appropriate cultures can be obtained. Culturenegative endocarditis may also be due to organisms that require special media for growth (eg, Legionella, Bartonella, Abiotrophia species, formerly referred to as nutritionally deficient streptococci), organisms that do not grow on artificial media (Tropheryma whipplei, or pathogens of Q fever or psittacosis), or those that may require prolonged incubation (eg, Brucella, anaerobes, HACEK organisms). Bartonella quintana is an important cause of culturenegative endocarditis. 2. Modified Duke criteria The Modified Duke criteria are useful for the diagnosis of endocarditis. Major criteria include (1) two positive blood cultures for a microorganism that typically causes infective endocarditis or persistent bacteremia, or a single positive blood culture for Coxiella burnetii or anti–phase 1 IgG antibody titer greater than or equal to 1:800; and (2) evidence of endocardial involvement documented by echocardiography showing definite vegetation, myocardial abscess, new partial dehiscence of a prosthetic valve, or new valvular regurgitation (increase or change in murmur is not sufficient). Minor criteria include the presence of a predisposing condition; fever of 38°C or higher; vascular phenomena, such as cutaneous hemorrhages, aneurysm, systemic emboli, or pulmonary infarction; immunologic phenomena, such as glomerulonephritis, Osler nodes, Roth spots, or rheumatoid factor; and positive blood cultures not meeting the major criteria or serologic evidence of an active infection. A definite diagnosis can be made with 80% accuracy if two major criteria, one major criterion and three minor criteria, or five minor criteria are fulfilled. A possible diagnosis of endocarditis is made if one major and one minor criterion or three minor criteria are met. If fewer criteria are found, or a sound alternative explanation for illness is identified, or the patient’s febrile illness has resolved within 4 days, endocarditis is unlikely. COMPLICATIONS The course of infective endocarditis is determined by the degree of damage to the heart, by the site of infection (rightsided versus leftsided, aortic versus mitral valve), by the presence of metastatic foci of infection, by the occurrence of embolization, and by immunologically mediated processes. Destruction of infected heart valves is especially common and precipitous with S aureus, but can occur with any organism and can progress even after bacteriologic cure. The infection can also extend into the myocardium, resulting in abscesses leading to conduction disturbances, and involving the wall of the aorta, creating sinus of Valsalva aneurysms. Peripheral embolization to the brain and myocardium may result in infarctions. Embolization to the spleen and kidneys is also common. Peripheral emboli may initiate metastatic infections or may become established in vessel walls, leading to mycotic aneurysms. Rightsided endocarditis, which usually involves the tricuspid valve, causes septic pulmonary emboli, occasionally with infarction and lung abscesses. PREVENTION The American Heart Association recommends antibiotic prophylaxis for infective endocarditis in a relatively small group of patients with predisposing congenital or valvular anomalies (Table 33–3) undergoing select dental procedures, operations involving the respiratory tract, or operations of infected skin, skin structure, or musculoskeletal tissue (Table 33–4). Current antimicrobial recommendations are given in Table 33–5. Table 33–3. Cardiac conditions with high risk of adverse outcomes from endocarditis for which prophylaxis with dental procedures is recommended.1,2 Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 7 / 12
- 8. 1Reproduced, with permission, from Wilson W et al. Prevention of infective endocarditis. Circulation. 2007 Oct 9;116(15):1736–54. Copyright © 2007 American Heart Association, Inc. 2See Table 33–5 for prophylactic regimens. 3Except for the conditions listed above, antibiotic prophylaxis is no longer recommended for other forms of CHD. 4Prophylaxis is recommended because endothelialization of prosthetic material occurs within 6 months after procedure. 1Reproduced, with permission, from Wilson W et al. Prevention of infective endocarditis. Circulation. 2007 Oct 9;116(15):1736–54. Copyright © 2007 American Heart Association, Inc. PREVENTION The American Heart Association recommends antibiotic prophylaxis for infective endocarditis in a relatively small group of patients with predisposing congenital or valvular anomalies (Table 33–3) undergoing select dental procedures, operations involving the respiratory tract, or operations of infected skin, skin structure, or musculoskeletal tissue (Table 33–4). Current antimicrobial recommendations are given in Table 33–5. Table 33–3. Cardiac conditions with high risk of adverse outcomes from endocarditis for which prophylaxis with dental procedures is recommended.1,2 Prosthetic cardiac valve Previous infective endocarditis Congenital heart disease (CHD)3 Unrepaired cyanotic CHD, including palliative shunts and conduits Completely repaired congenital heart defect with prosthetic material or device, whether placed by surgery or by catheter intervention, during the first 6 months after the procedure4 Repaired CHD with residual defects at the site or adjacent to the site of a prosthetic patch or prosthetic device Cardiac transplantation recipients in whom cardiac valvulopathy develops Table 33–4. Recommendations for administration of bacterial endocarditis prophylaxis for patients according to type of procedure.1 Prophylaxis Recommended Prophylaxis Not Recommended Dental procedures All dental procedures that involve manipulation of gingival tissue or the periapical region of the teeth or perforation of the oral mucosa Respiratory tract procedures Only respiratory tract procedures that involve incision of the respiratory mucosa Procedures on infected skin, skin structure, or musculoskeletal tissue Dental procedures Routine anesthetic injections through noninfected tissue, taking dental radiographs, placement of removable prosthodontic or orthodontic appliances, adjustment of orthodontic appliances, placement of orthodontic brackets, shedding of deciduous teeth, and bleeding from trauma to the lips or oral mucosa Gastrointestinal tract procedures Genitourinary tract procedures Table 33–5. American Heart Association recommendations for endocarditis prophylaxis for dental procedures for patients with cardiac conditions.1–3 Oral Amoxicillin 2 g 1 hour before procedure Penicillin allergy Clindamycin 600 mg 1 hour before procedure or Cephalexin 2 g 1 hour before procedure (contraindicated if there is history of a betalactam immediate hypersensitivity reaction) Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 8 / 12
- 9. 1Reproduced, with permission, from Wilson W et al. Prevention of infective endocarditis. Circulation. 2007 Oct 9;116(15):1736–54. Copyright © 2007 American Heart Association, Inc. 1Data from the American Heart Association. Circulation. 2007 Oct 9;116(15):1736–54. 2For patients undergoing respiratory tract procedures involving incision of respiratory tract mucosa to treat an established infection or a procedure on infected skin, skin structure, or musculoskeletal tissue known or suspected to be caused by S aureus, the regimen should contain an antistaphylococcal penicillin or cephalosporin. Vancomycin can be used to treat patients unable to tolerate a betalactam or if the infection is known or suspected to be caused by a methicillin resistant strain of S aureus. 3See Table 33–3 for list of cardiac conditions. structure, or musculoskeletal tissue Table 33–5. American Heart Association recommendations for endocarditis prophylaxis for dental procedures for patients with cardiac conditions.1–3 Oral Amoxicillin 2 g 1 hour before procedure Penicillin allergy Clindamycin 600 mg 1 hour before procedure or Cephalexin 2 g 1 hour before procedure (contraindicated if there is history of a betalactam immediate hypersensitivity reaction) or Azithromycin or clarithromycin 500 mg 1 hour before procedure Parenteral Ampicillin 2 g intramuscularly or intravenously 30 minutes before procedure Penicillin allergy Clindamycin 600 mg intravenously 1 hour before procedure or Cefazolin 1 g intramuscularly or intravenously 30 minutes before procedure (contraindicated if there is history of a beta lactam immediate hypersensitivity reaction) TREATMENT Empiric regimens for endocarditis while culture results are pending should include agents active against staphylococci, streptococci, and enterococci. Vancomycin 1 g every 12 hours intravenously plus ceftriaxone 2 g every 24 hours provides appropriate coverage pending definitive diagnosis; consultation with an infectious disease expert is strongly recommended when initiating treatment. A. Viridans Streptococci For penicillinsusceptible viridans streptococcal endocarditis (ie, MIC 0.1 mcg/mL or less), penicillin G, 18 million units intravenously either continuously or in four to six equally divided doses, or ceftriaxone, 2 g intravenously once daily for 4 weeks, is recommended. The duration of therapy can be shortened to 2 weeks if gentamicin, 3 mg/kg intravenously every 24 hours, is used with penicillin or ceftriaxone. The 2week regimen is reasonable and can be considered in patients with uncomplicated endocarditis, rapid response to therapy, and no underlying kidney disease. For the patient unable to tolerate penicillin or ceftriaxone, vancomycin, 15 mg/kg intravenously every 12 hours for 4 weeks, is given with a desired trough level of 10–15 mcg/mL. Prosthetic valve endocarditis is treated with a 6week course of penicillin or ceftriaxone and the clinician can consider adding 2 weeks of gentamicin at the start of therapy. Viridans streptococci relatively resistant to penicillin (ie, MIC greater than 0.12 mcg/mL but less than or equal to 0.5 mcg/mL) should be treated for 4 weeks. Penicillin G, 24 million units intravenously either continuously or in four to six equally divided doses, is combined with gentamicin, 3 mg/kg intravenously every 24 hours for the first 2 weeks. Ceftriaxone may be a reasonable alternative treatment option for isolates that are susceptible to Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 9 / 12
- 10. continuously or in four to six equally divided doses, or ceftriaxone, 2 g intravenously once daily for 4 weeks, is recommended. The duration of therapy can be shortened to 2 weeks if gentamicin, 3 mg/kg intravenously every 24 hours, is used with penicillin or ceftriaxone. The 2week regimen is reasonable and can be considered in patients with uncomplicated endocarditis, rapid response to therapy, and no underlying kidney disease. For the patient unable to tolerate penicillin or ceftriaxone, vancomycin, 15 mg/kg intravenously every 12 hours for 4 weeks, is given with a desired trough level of 10–15 mcg/mL. Prosthetic valve endocarditis is treated with a 6week course of penicillin or ceftriaxone and the clinician can consider adding 2 weeks of gentamicin at the start of therapy. Viridans streptococci relatively resistant to penicillin (ie, MIC greater than 0.12 mcg/mL but less than or equal to 0.5 mcg/mL) should be treated for 4 weeks. Penicillin G, 24 million units intravenously either continuously or in four to six equally divided doses, is combined with gentamicin, 3 mg/kg intravenously every 24 hours for the first 2 weeks. Ceftriaxone may be a reasonable alternative treatment option for isolates that are susceptible to ceftriaxone. In the patient with IgEmediated allergy to penicillin, vancomycin alone, 15 mg/kg intravenously every 12 hours for 4 weeks, should be administered. Prosthetic valve endocarditis is treated with a 6week course of penicillin or ceftriaxone plus gentamicin as above. Endocarditis caused by viridans streptococci with an MIC greater than 0.5 mcg/mL or by nutritionally deficient streptococci should be treated the same as enterococcal endocarditis. B. Other Streptococci Endocarditis caused by S pneumoniae, S pyogenes (group A streptococcus), or groups B, C, and G streptococci is unusual. S pneumoniae sensitive to penicillin (MIC less than 0.1 mcg/mL) can be treated with penicillin 18 million units intravenously either continuously or in four to six equally divided doses or cefazolin 6 g intravenously either continuously or in three equally divided doses, or ceftriaxone 2 g daily intravenously for 4 weeks. Highdose penicillin (24 million units) or a thirdgeneration cephalosporin may be required for the treatment of endocarditis (without meningitis) caused by strains resistant to penicillin (MIC greater than 0.1 mcg/mL). The addition of vancomycin and rifampin to ceftriaxone may be considered in patients with S pneumoniae strains with cefotaxime MIC greater than 2 mcg/mL. Group A streptococcal infection can be treated with penicillin or ceftriaxone for 4–6 weeks. Groups B, C, and G streptococci tend to be more resistant to penicillin than group A streptococci, and some experts have recommended adding gentamicin, 3 mg/kg intravenously every 24 hours, to penicillin for the first 2 weeks of a 4 to 6week course. Endocarditis caused by S gallolyticus (bovis) is associated with liver disease, especially cirrhosis, and gastrointestinal abnormalities, especially colon cancer. Colonoscopy should be performed to exclude the latter. C. Enterococci For enterococcal endocarditis, penicillin or ampicillin alone is inadequate. The combination of penicillin or ampicillin with gentamicin had been the treatment of choice but several clinical studies of the combination of ampicillin plus ceftriaxone have demonstrated clinical outcomes equivalent to the combination of ampicillin plus gentamicin. One recommended regimen is ampicillin, 2 g intravenously every 4 hours, or penicillin G, 18–30 million units intravenously continuously or in six equally divided doses plus gentamicin, 1 mg/kg intravenously every 8 hours. The second recommended regimen is ampicillin (2 g intravenously every 4 hours) plus ceftriaxone 2 g intravenously every 12 hours. The recommended duration of therapy is 4–6 weeks (the longer duration for patients with symptoms for more than 3 months, relapse, or prosthetic valve endocarditis). The combination of ampicillin plus ceftriaxone is recommended for patients with creatinine clearance less than 50 mL/min or whose enterococcal isolates are resistant to gentamicin. In patients intolerant of penicillin and ampicillin or who have enterococcal isolate resistant to these agents, vancomycin plus gentamicin can be used. Endocarditis caused by strains resistant to penicillin and vancomycin are difficult to treat and should always be managed in consultation with an infectious diseases specialist. D. Staphylococci For methicillinsusceptible S aureus isolates, nafcillin or oxacillin, 12 g intravenously daily given continuously or in four to six divided doses or cefazolin, 6 g intravenously daily given continuously or in three divided doses for 6 weeks, is the preferred therapy. In cases of brain abscess resulting from methicillinsusceptible S aureus endocarditis, nafcillin should be used instead of cefazolin. For patients with history of immediate type hypersensitivity to betalactams, a desensitization protocol should be undertaken. For patients with a history of nonanaphylactoid reactions to penicillins, cefazolin should be used. Patients who are infected with methicillinresistant S aureus or who are unable to tolerate betalactam therapy should receive vancomycin, 30 mg/kg/day intravenously divided in two or three doses, to achieve a goal trough level of 15–20 mcg/kg, or daptomycin intravenously at greater than or equal to 8 mg/kg/day. Aminoglycoside combination regimens are not recommended. The effect of rifampin with antistaphylococcal drugs is variable, and its routine use is not recommended. Because coagulasenegative staphylococci—a common cause of prosthetic valve endocarditis—are routinely resistant to methicillin, betalactam antibiotics should not be used for this infection unless the isolate is demonstrated to be susceptible. A combination of vancomycin, 30 mg/kg/day intravenously divided in two or three doses for 6 weeks; rifampin, 300 mg every 8 hours for 6 weeks; and gentamicin, 3 mg/kg intravenously every 8 hours for the first 2 weeks, is recommended for prosthetic valve infection. If the organism is sensitive to methicillin, either nafcillin or oxacillin or cefazolin can be used in combination with rifampin and gentamicin. Combination therapy with nafcillin or oxacillin (vancomycin or daptomycin for methicillinresistant strains), rifampin, and gentamicin is also recommended for treatment of S aureus prosthetic valve infection. Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 10 / 12
- 11. intravenously at greater than or equal to 8 mg/kg/day. Aminoglycoside combination regimens are not recommended. The effect of rifampin with antistaphylococcal drugs is variable, and its routine use is not recommended. Because coagulasenegative staphylococci—a common cause of prosthetic valve endocarditis—are routinely resistant to methicillin, betalactam antibiotics should not be used for this infection unless the isolate is demonstrated to be susceptible. A combination of vancomycin, 30 mg/kg/day intravenously divided in two or three doses for 6 weeks; rifampin, 300 mg every 8 hours for 6 weeks; and gentamicin, 3 mg/kg intravenously every 8 hours for the first 2 weeks, is recommended for prosthetic valve infection. If the organism is sensitive to methicillin, either nafcillin or oxacillin or cefazolin can be used in combination with rifampin and gentamicin. Combination therapy with nafcillin or oxacillin (vancomycin or daptomycin for methicillinresistant strains), rifampin, and gentamicin is also recommended for treatment of S aureus prosthetic valve infection. E. HACEK Organisms HACEK organisms are slowgrowing, fastidious gramnegative coccobacilli or bacilli (H aphrophilus [now A aphrophilus], A actinomycetemcomitans, C hominis, E corrodens, and Kingella species) that are normal oral flora and cause less than 5% of all cases of endocarditis. They may produce beta lactamase, and thus the treatment of choice is ceftriaxone (or another thirdgeneration cephalosporin), 2 g intravenously once daily for 4 weeks. Prosthetic valve endocarditis should be treated for 6 weeks. In the penicillinallergic patient, experience is limited, but fluoroquinolones have in vitro activity and should be considered. F. CultureNegative Endocarditis Failure to culture microorganisms from patients with suspected infective endocarditis may be due to infection from organisms not recovered in routine microbiology testing or previous administration of antimicrobial agents before blood cultures were obtained. These cases must be managed with the assistance of an infectious disease specialist. Pathogens that are not able to be cultured by commonly used techniques include Bartonella species, Chlamydia species, Brucella species, and Tropheryma whipplei. Serologic testing should be performed in patients who have epidemiologic risk factors for these infections. Treatment should be directed at likely pathogens while awaiting serologic results; treatment of patients given prior antimicrobials before cultures were obtained must also consider likely pathogens. G. Role of Surgery While many cases can be successfully treated medically, operative management is frequently required. Acute heart failure unresponsive to medical therapy is an indication for valve replacement even if active infection is present. Infections unresponsive to appropriate antimicrobial therapy after 7– 10 days (ie, persistent fevers, positive blood cultures despite therapy) are more likely to be eradicated if the valve is replaced. Surgery is nearly always required for cure of fungal endocarditis and is more often necessary with highly resistant bacteria. It is also indicated when the infection involves the sinus of Valsalva or produces septal abscesses. Recurrent infection with the same organism prompts an operative approach, especially with infected prosthetic valves. Continuing embolization presents a difficult problem when the infection is otherwise responding; surgery may be the proper approach. Particularly challenging is a large and fragile vegetation demonstrated by echocardiography in the absence of embolization. Most clinicians favor an operative approach with vegetectomy and valve repair if the patient is a good candidate. Operation without delay may be considered in patients with endocarditis and an ischemic stroke who have an indication for surgery. If not urgent or if intracranial hemorrhage is present, a delay of at least 4 weeks should be considered. Embolization after bacteriologic cure does not necessarily imply recurrence of endocarditis. H. Role of Anticoagulation Anticoagulation is contraindicated in native valve endocarditis because of an increased risk of intracerebral hemorrhage from mycotic aneurysms or embolic phenomena. The role of anticoagulant therapy during prosthetic valve endocarditis is more controversial. Reversal of anticoagulation may result in thrombosis of the mechanical prosthesis, particularly in the mitral position. Conversely, anticoagulation during active prosthetic valve endocarditis caused by S aureus has been associated with fatal intracerebral hemorrhage. One approach is to discontinue anticoagulation during the septic phase of S aureus prosthetic valve endocarditis. In patients with S aureus prosthetic valve endocarditis complicated by a CNS embolic event, anticoagulation should be discontinued for the first 2 weeks of therapy. Indications for anticoagulation following prosthetic valve implantation for endocarditis are the same as for patients with prosthetic valves without endocarditis (eg, nonporcine mechanical valves and valves in the mitral position). RESPONSE TO THERAPY If infection is caused by viridans streptococci, enterococci, or coagulasenegative staphylococci, defervescence occurs in 3–4 days on average; with S aureus or Pseudomonas aeruginosa, fever may persist for longer. Blood cultures should be obtained every 1–2 days to document sterilization. Other causes of persistent fever are myocardial or metastatic abscess, sterile embolization, superimposed hospitalacquired infection, and drug reaction. Most relapses occur within 1–2 months after completion of therapy. Obtaining one or two blood cultures during this period is prudent. WHEN TO REFER Consider consulting an infectious diseases specialist in all cases of suspected infective endocarditis. Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 11 / 12
- 12. RESPONSE TO THERAPY If infection is caused by viridans streptococci, enterococci, or coagulasenegative staphylococci, defervescence occurs in 3–4 days on average; with S aureus or Pseudomonas aeruginosa, fever may persist for longer. Blood cultures should be obtained every 1–2 days to document sterilization. Other causes of persistent fever are myocardial or metastatic abscess, sterile embolization, superimposed hospitalacquired infection, and drug reaction. Most relapses occur within 1–2 months after completion of therapy. Obtaining one or two blood cultures during this period is prudent. WHEN TO REFER Consider consulting an infectious diseases specialist in all cases of suspected infective endocarditis. Consult a cardiac surgeon in the situations mentioned in the Role of Surgery section above to prevent further embolic disease, heart failure, and other complications including death. WHEN TO ADMIT Patients with infective endocarditis should be hospitalized for expedited evaluation and treatment. Baddour LM et al. Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications: a scientific statement for healthcare professionals from the American Heart Association. Circulation. 2015 Oct 13; 132(15):1435–86. Erratum in: Circulation. 2015 Oct 27; 132(17):e215. [PubMed: 26373316] Vincent LL et al. Infective endocarditis: update on epidemiology, outcomes and management. Curr Cardiol Rep. 2018 Aug 16;20(10):86. [PubMed: 30117004] Wilson W et al. Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group. Circulation. 2007 Oct 9; 116(15):1736–54. [PubMed: 17446442] Access Provided by: Downloaded 2022316 12:11 P Your IP is 197.210.53.158 3309: Infective Endocarditis, Bryn A. Boslett; Brian S. Schwartz ©2022 McGraw Hill. All Rights Reserved. Terms of Use • Privacy Policy • Notice • Accessibility Page 12 / 12