Report
Share
Download to read offline
Recommended
Recommended
More than Just Lines on a Map: Best Practices for U.S Bike Routes
This session highlights best practices and lessons learned for U.S. Bike Route System designation, as well as how and why these routes should be integrated into bicycle planning at the local and regional level.
Presenters:
Presenter: Kevin Luecke Toole Design Group
Co-Presenter: Virginia Sullivan Adventure Cycling AssociationMore than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike RoutesProject for Public Spaces & National Center for Biking and Walking
More Related Content
Featured
More than Just Lines on a Map: Best Practices for U.S Bike Routes
This session highlights best practices and lessons learned for U.S. Bike Route System designation, as well as how and why these routes should be integrated into bicycle planning at the local and regional level.
Presenters:
Presenter: Kevin Luecke Toole Design Group
Co-Presenter: Virginia Sullivan Adventure Cycling AssociationMore than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike RoutesProject for Public Spaces & National Center for Biking and Walking
Featured (20)
Content Methodology: A Best Practices Report (Webinar)

Content Methodology: A Best Practices Report (Webinar)
How to Prepare For a Successful Job Search for 2024

How to Prepare For a Successful Job Search for 2024
Social Media Marketing Trends 2024 // The Global Indie Insights

Social Media Marketing Trends 2024 // The Global Indie Insights
Trends In Paid Search: Navigating The Digital Landscape In 2024

Trends In Paid Search: Navigating The Digital Landscape In 2024
5 Public speaking tips from TED - Visualized summary

5 Public speaking tips from TED - Visualized summary
Google's Just Not That Into You: Understanding Core Updates & Search Intent

Google's Just Not That Into You: Understanding Core Updates & Search Intent
The six step guide to practical project management

The six step guide to practical project management
Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...

Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...
Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...

Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...
More than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike Routes
Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...

Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...
Wilkins Poster-TSM-2015
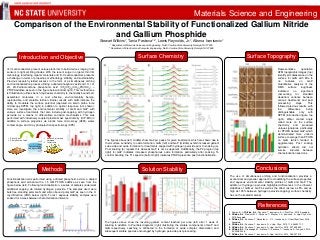
- 1. III-V semiconductors present unique platforms to build devices ranging from sensors to light emitting diodes. With the recent surge in organic thin film technology, interfacing organic materials and III-V semiconductors presents a challenge in terms of properties, methodology, stability, and sustainability. Previous research yielded success in the form of a simultaneous etching and functionalization process utilizing a diluted phosphoric acid and 1H, 1H, 2H, 2H-Perfluorooctane phosphonic acid (CF3(CF2)5(CH2)2PO(OH)2) – PFOPA additive as seen in the figure below (bottom right). This method (see left illustration) utilizes bench-top beaker chemistry to chemically bound self- assembled molecules in a cost effective, environmentally benign, reproducible, and scalable fashion. Earlier results with GaN indicate the ability to modulate the surface potential (captured via kelvin probe force microscopy-KPFM, top right) in addition to optical response (not shown). Here we investigate the environmental stability of GaN and GaP with various surface treatments that also include post-capping with hydrogen peroxide as a means to differentiate corrosion mechanisms. This was performed with inductively coupled plasma-mass spectrometry (ICP-MS) in addition to surface properties via atomic force microscopy (AFM), water contact angle, and X-ray photoelectron spectroscopy (XPS) Comparison of the Environmental Stability of Functionalized Gallium Nitride and Gallium Phosphide Stewart Wilkins1, Tania Paskova1,2, Lewis Reynolds, Jr.1, Albena Ivanisevic1 1 Department of Materials Science and Engineering, North Carolina State University, Raleigh, NC 27695 2 Department of Electrical and Computer Engineering, North Carolina State University, Raleigh, NC 27695 Materials Science and Engineering Representative qualitative AFM topographic images (top) identify pit coalescence on the surface for GaN with little to no features on GaP. Quantitative analysis of the RMS surface roughness indicated no significant (accounting for standard deviation) variance between various treatments and post processing steps. This followed previous results, with the differences being distinguishable only with KPFM (introduction figure, top right). Water contact angle relied more on the surface chemistry and produced unique signatures, particularly for PFOPA treated GaP, which demonstrated more uniform surface coverage than PFOPA on GaN (which tended to agglomerate). Post soaking hydration effects did not survive intervals between characterization sessions. Polar Bulk Nonpolar Bulk Etched Nonpolar on Al2O3 1. Pearce, B. L.; Wilkins, S. J.; Paskova, T.; Ivanisevic, A.; J. Mat. Res., 2015, Accepted. 2. Wilkins, S. J.; Slomski, M. J.; Paskova, T.; Weyher, J. L.; Ivanisevic, A.; App. Phys. Lttrs., 2015, Accepted. 3. Wilkins, S. J.; Paskova, T.; Reynolds, Jr., C. L.; Ivanisevic, A.; ChemPhysChem, 2015, Accepted. 4. Wilkins, S. J.; Paskova, T.; Ivanisevic, A.; J. App. Phys., 2013, 114, 064907-1-7. 5. Wilkins, S. J.; Paskova, T.; Ivanisevic, A.; App. Surf. Sci., 2015, 327, 498-503. 6. Wilkins, S. J.; Greenough, M.; Arellano, C.; Paskova, T.; Ivanisevic, A.; Langmuir, 2014, 30, 2038-2046. 7. Wilkins, S. J.; Paskova, T.; Ivanisevic, A.; App. Surf. Sci., 2014, 295, 207-213. Etched The figures above (left, middle) show Ga 2p3/2 peaks for given treatments, which are shown due to their surface sensitivity to oxide formation. GaN (left) and GaP (middle) exhibit increased gallium sub-oxide and oxide formation for treatments capped with hydrogen peroxide prior to soaking (a). Post soaking (b) repeats this whereas GaP is not as apparent. Additionally, the P2p region (top right) for GaP indicates increased phosphorous oxide formation, particularly after soaking. To confirm bonding, the F1s spectra (bottom right) indicates PFOPA presence post functionalization. Introduction and Objective Methods Surface Chemistry References The use of simultaneous etching and functionalization provides a economical and greener approach to modulating the surface properties and aqueous environmental stability profiles of GaN and GaP. The addition of hydrogen peroxide highlighted differences in the inherent stabilities of GaN vs. GaP as well as the effect (as low as 8% and as high as 145% between hydrogen peroxide capping) surface chemistry has on the treated material. Conclusions Functionalization was performed using a diluted phosphoric acid or a diluted phosphoric acid etchant with a 1.5 mM PFOPA additive as seen from the figure below (left). Following functionalization, a series of samples underwent additional capping via diluted hydrogen peroxide. The samples used were bulk-free standing polar GaN and silicon bound polar GaP as seen in the X- ray diffraction (XRD) below (right). To test aqueous stability, samples were soaked for a week between characterization intervals. Surface Topography Solution Stability The figures above show the resulting gallium content leached per area (left) after 1 week of soaking in addition to Pourbaix diagrams (right) illustrating the soluble components of GaP and GaN respectively. Leaching is attributed to the formation of oxide complex dissociation and subsequent soluble species encouraged by hydrogen peroxide post-processing. Functionalization and Etchant Bath 1. Solvent Clean 2. Etch to Remove Oxide 1. Remove Physisorbed Adsorbate 2. Characterize