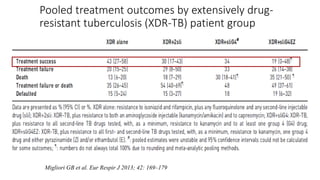
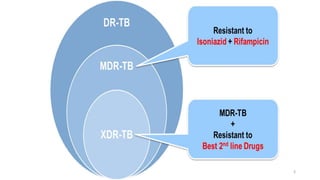
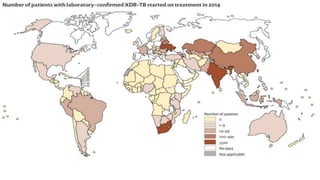
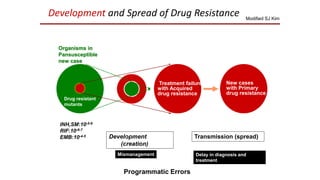
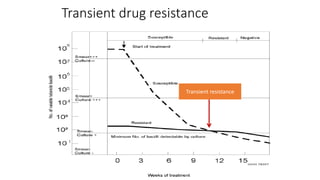
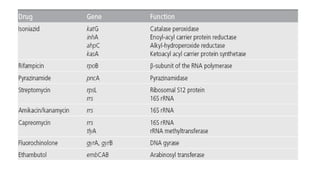
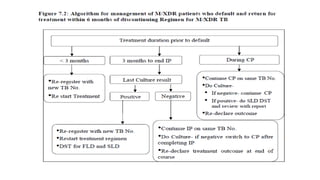
This document discusses extensively drug resistant tuberculosis (XDR TB). It defines XDR TB as resistance to at least rifampin and isoniazid among first-line drugs, plus resistance to any fluoroquinolone and at least one second-line injectable drug. Globally, an estimated 9.7% of MDR TB cases have XDR TB. XDR TB has been reported in 105 countries as of 2014, with the highest rates in several Eastern European and Southern African countries. Treatment of XDR TB requires utilizing the few drugs the infection remains susceptible to in a lengthy 24-30 month regimen.


















































![J Antimicrob Chemother. 2014 Jun 30. pii: dku235. [Epub ahead of print]
Clofazimine in the treatment of extensively drug-resistant tuberculosis with
HIV coinfection in South Africa: a retrospective cohort study.
Padayatchi N, Gopal M, Naidoo R, Werner L, Naidoo K, Master I, O'Donnell MR.
• Between August 2009 and July 2011, eligible XDR-TB patients (n = 85) were initiated on treatment
for XDR-TB.
• Most patients (86%) were HIV-infected and receiving antiretroviral therapy (90%).
• Patients receiving a clofazimine-containing regimen (n = 50) had a higher percentage of
culture conversion (40%) compared with patients (n = 35) receiving a non-clofazimine
regimen (28.6%).
• On multivariate analysis, there was a 2-fold increase in TB culture conversion at 6 months
(hazard rate ratio 2.54, 95% CI 0.99-6.52, P = 0.05) in the group receiving a clofazimine-
containing regimen.
• Adverse effects due to clofazimine were minor and rarely life-threatening.](https://image.slidesharecdn.com/prapulxdrtb-170426171732/85/XDR-TB-59-320.jpg)