UK Facilities
•Download as PPT, PDF•
0 likes•353 views
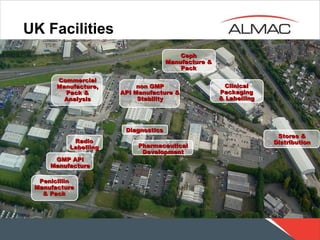
This document outlines various pharmaceutical manufacturing and production activities including non-GMP API manufacture and stability testing in UK facilities, GMP API manufacture, pharmaceutical development, clinical packaging and labeling, radio labeling, diagnostics, stores and distribution, ceph manufacture and packaging, and penicillin manufacture and packaging.
Report
Share
Report
Share
Recommended
JND Packaging 6-8

JND Packaging is a leading innovator of hygienic plastic packaging solutions in China, focusing on the food, dairy, pharmaceutical, and healthcare industries. It has GMP and ISO certifications and provides design, mold development, molding, and surface decoration services. JND has state-of-the-art curve surface offset printing technology and pioneered high barrier multilayer packaging. It is committed to supplying cutting-edge packaging solutions to help brands adapt to changing markets.
Jnd in marketing consumer behavior 

The Just Noticeable Difference (JND) refers to the smallest detectable difference between two stimuli that a person can perceive. In marketing, it is important to determine the JND for products so that any negative changes are not noticeable to customers, while improvements are apparent. The concept of the JND can also be applied to pricing, promotion, packaging, and product modifications - any changes should be below the JND threshold so customers accept them gradually without discomfort to the perceived change.
20 Interesting Things: QR Codes

This is the fifth deck in our summer series 20 Interesting Things. It takes a quick look at what is happening in the Crowdsourcing space from both a marketing and general use perspective. Look for our other decks on Foursquare, Augmented Reality, Crowdsourcing and Goodness.
Colour Perception and Consumer Behaviour

The document discusses how colour influences consumer behaviour and marketing. It provides examples of how different colours like red, yellow, blue, green, orange, pink, purple, black are perceived and can stimulate certain emotions. It also gives examples of how companies have successfully used colour in their branding and product packaging to influence sales and target certain demographics like Coca Cola switching to black packaging for Coke Zero to target males. Culture can influence colour meanings so marketers must be aware of this. Colour appropriateness is also important for products.
Perception- Interpretation Gestalt & Semiotics 

Gestalt theory states that the brain perceives whole forms or patterns rather than individual parts. There are several Gestalt laws including figure-ground, closure, similarity, and proximity. Semiotics is the study of signs and symbols and how they influence interpretation. Signs have a signifier, signified, and together they form a sign. There are three types of signs: iconic, indexical, and symbolic. Advertisements often use metonymic codes by creating associations between products and concepts. Analogical codes create mental comparisons, while displaced codes transfer meaning between symbols.
Almac Protein Labelling Technology

This document discusses site-specific protein modification using Almac's proprietary protein ligation technology. The technology enables proteins to be modified at their C-terminus in a highly selective and high-yielding manner, resulting in homogeneous conjugated protein products with retained biological activity. This overcomes limitations of existing nonspecific protein modification methods. The technology has been demonstrated on various therapeutic proteins and can attach a wide range of labels and molecules like PEG for half-life extension.
14C Labelled Peptide API\'s by Sean Kitson

This document discusses carbon-14 labeling of peptides for use in ADME studies. It provides an overview of carbon-14, its production and starting materials. Synthetic strategies for incorporating carbon-14 into peptides are described, including direct labeling of amino acids or terminal residues. Case studies demonstrate labeling strategies for two peptides, one involving a biotinylation reaction. The document concludes that carbon-14 labeling is well-suited for assessing a drug's ADME profile and that limitations in specific activity can be overcome through accelerated mass spectrometry.
SelectAZyme

This document summarizes the key biocatalysis services offered by Almac, including enzyme screening and process optimization, chiral building block synthesis, metabolite synthesis, and dedicated support for integrating biocatalytic technology. Almac has capabilities across enzyme selection, scale-up from grams to tons, and case studies in areas like using carbonyl reductase to synthesize chiral alcohols with high enantiomeric excess, resolving difficult alcohols with hydrolase, and producing metabolites at multi-kilogram scale for further studies. Contact information is provided for inquiries regarding Almac's biocatalysis services.
Recommended
JND Packaging 6-8

JND Packaging is a leading innovator of hygienic plastic packaging solutions in China, focusing on the food, dairy, pharmaceutical, and healthcare industries. It has GMP and ISO certifications and provides design, mold development, molding, and surface decoration services. JND has state-of-the-art curve surface offset printing technology and pioneered high barrier multilayer packaging. It is committed to supplying cutting-edge packaging solutions to help brands adapt to changing markets.
Jnd in marketing consumer behavior 

The Just Noticeable Difference (JND) refers to the smallest detectable difference between two stimuli that a person can perceive. In marketing, it is important to determine the JND for products so that any negative changes are not noticeable to customers, while improvements are apparent. The concept of the JND can also be applied to pricing, promotion, packaging, and product modifications - any changes should be below the JND threshold so customers accept them gradually without discomfort to the perceived change.
20 Interesting Things: QR Codes

This is the fifth deck in our summer series 20 Interesting Things. It takes a quick look at what is happening in the Crowdsourcing space from both a marketing and general use perspective. Look for our other decks on Foursquare, Augmented Reality, Crowdsourcing and Goodness.
Colour Perception and Consumer Behaviour

The document discusses how colour influences consumer behaviour and marketing. It provides examples of how different colours like red, yellow, blue, green, orange, pink, purple, black are perceived and can stimulate certain emotions. It also gives examples of how companies have successfully used colour in their branding and product packaging to influence sales and target certain demographics like Coca Cola switching to black packaging for Coke Zero to target males. Culture can influence colour meanings so marketers must be aware of this. Colour appropriateness is also important for products.
Perception- Interpretation Gestalt & Semiotics 

Gestalt theory states that the brain perceives whole forms or patterns rather than individual parts. There are several Gestalt laws including figure-ground, closure, similarity, and proximity. Semiotics is the study of signs and symbols and how they influence interpretation. Signs have a signifier, signified, and together they form a sign. There are three types of signs: iconic, indexical, and symbolic. Advertisements often use metonymic codes by creating associations between products and concepts. Analogical codes create mental comparisons, while displaced codes transfer meaning between symbols.
Almac Protein Labelling Technology

This document discusses site-specific protein modification using Almac's proprietary protein ligation technology. The technology enables proteins to be modified at their C-terminus in a highly selective and high-yielding manner, resulting in homogeneous conjugated protein products with retained biological activity. This overcomes limitations of existing nonspecific protein modification methods. The technology has been demonstrated on various therapeutic proteins and can attach a wide range of labels and molecules like PEG for half-life extension.
14C Labelled Peptide API\'s by Sean Kitson

This document discusses carbon-14 labeling of peptides for use in ADME studies. It provides an overview of carbon-14, its production and starting materials. Synthetic strategies for incorporating carbon-14 into peptides are described, including direct labeling of amino acids or terminal residues. Case studies demonstrate labeling strategies for two peptides, one involving a biotinylation reaction. The document concludes that carbon-14 labeling is well-suited for assessing a drug's ADME profile and that limitations in specific activity can be overcome through accelerated mass spectrometry.
SelectAZyme

This document summarizes the key biocatalysis services offered by Almac, including enzyme screening and process optimization, chiral building block synthesis, metabolite synthesis, and dedicated support for integrating biocatalytic technology. Almac has capabilities across enzyme selection, scale-up from grams to tons, and case studies in areas like using carbonyl reductase to synthesize chiral alcohols with high enantiomeric excess, resolving difficult alcohols with hydrolase, and producing metabolites at multi-kilogram scale for further studies. Contact information is provided for inquiries regarding Almac's biocatalysis services.
Analytical Capabilities

Analytical Services provides analytical testing and support for drug development from early stage research through commercialization. This includes API and drug product testing, method development and validation, stability testing, physical and chemical characterization, and analytical support for clinical trials. Analytical Services has over 150 analysts and state-of-the-art laboratories and equipment to meet all analytical needs throughout drug development.
Solid State Capabilities

Almac provides solid state services to support drug development from invention to launch. Their services include polymorph screening and selection, crystallization development, physical form characterization, analytical method development and validation, and troubleshooting. They have highly trained scientists with the ability to solve any solid state problems. Their goal is to find and produce the optimal physical form of drugs to ensure development success.
API Supply

Almac provides chemistry and manufacturing solutions for APIs from discovery through commercialization. They offer multi-purpose GMP manufacturing facilities, process R&D, preclinical through commercial API supply, analytical development and validation, potent API production, biotransformation, micronization, and process validation using quality by design approaches. Almac has expertise in chemical development, biocatalysis, physical sciences, formulation development, and analytical services to support clients' API needs.
Rapidd™

Almac offers an accelerated drug development process that can take a lead candidate from selection to Phase I regulatory submission in just 12 months. Their integrated single-site capabilities include preclinical safety assessment studies, API and drug product development, analytical support, regulatory support, and dedicated project management. Using a single contract and commercial contact, Almac aims to reduce the effort spent on auditing, coordinating, and managing multiple vendors during the early clinical development process.
Radiolabelling Capabilities

Almac provides radiolabeling services including the synthesis of 14C labeled APIs and drug products for human ADME studies. They have a 14C manufacturing license and cGMP compliance. Almac uses integrated teams of radiochemists and analytical experts along with state-of-the-art facilities to efficiently develop and manufacture radiolabeled compounds. Case studies demonstrate successful projects involving peptide radiolabeling and developing powder-filled capsules and sterile solutions for clinical studies.
Chemokines

This document provides information on human synthetic wild-type chemokines that are site-specifically labelled and fully functional. It lists 17 chemokines along with their systematic names, native chemokine names, receptors they bind to, and product codes for native, biotin-labelled, and AlexaFluor 647-labelled versions. Additional services offered by the company include custom human and non-human chemokines, 14C labelling, GMP peptide synthesis, analytical services, and peptide pre-formulation.
Peptide & Protein Capabilities

Almac provides services related to peptide synthesis and protein engineering including GMP manufacturing for clinical trials, a chemokine catalogue, fluorescence lifetime assays, and proprietary technologies for site-specific protein modification. They offer a complete package for first-in-man clinical trials including chemical and analytical development, material supply, oversight of formulation and clinical trials. Case studies demonstrate their work synthesizing modified chemokines and developing sterile drug products for clinical use.
More Related Content
More from ejmckee
Analytical Capabilities

Analytical Services provides analytical testing and support for drug development from early stage research through commercialization. This includes API and drug product testing, method development and validation, stability testing, physical and chemical characterization, and analytical support for clinical trials. Analytical Services has over 150 analysts and state-of-the-art laboratories and equipment to meet all analytical needs throughout drug development.
Solid State Capabilities

Almac provides solid state services to support drug development from invention to launch. Their services include polymorph screening and selection, crystallization development, physical form characterization, analytical method development and validation, and troubleshooting. They have highly trained scientists with the ability to solve any solid state problems. Their goal is to find and produce the optimal physical form of drugs to ensure development success.
API Supply

Almac provides chemistry and manufacturing solutions for APIs from discovery through commercialization. They offer multi-purpose GMP manufacturing facilities, process R&D, preclinical through commercial API supply, analytical development and validation, potent API production, biotransformation, micronization, and process validation using quality by design approaches. Almac has expertise in chemical development, biocatalysis, physical sciences, formulation development, and analytical services to support clients' API needs.
Rapidd™

Almac offers an accelerated drug development process that can take a lead candidate from selection to Phase I regulatory submission in just 12 months. Their integrated single-site capabilities include preclinical safety assessment studies, API and drug product development, analytical support, regulatory support, and dedicated project management. Using a single contract and commercial contact, Almac aims to reduce the effort spent on auditing, coordinating, and managing multiple vendors during the early clinical development process.
Radiolabelling Capabilities

Almac provides radiolabeling services including the synthesis of 14C labeled APIs and drug products for human ADME studies. They have a 14C manufacturing license and cGMP compliance. Almac uses integrated teams of radiochemists and analytical experts along with state-of-the-art facilities to efficiently develop and manufacture radiolabeled compounds. Case studies demonstrate successful projects involving peptide radiolabeling and developing powder-filled capsules and sterile solutions for clinical studies.
Chemokines

This document provides information on human synthetic wild-type chemokines that are site-specifically labelled and fully functional. It lists 17 chemokines along with their systematic names, native chemokine names, receptors they bind to, and product codes for native, biotin-labelled, and AlexaFluor 647-labelled versions. Additional services offered by the company include custom human and non-human chemokines, 14C labelling, GMP peptide synthesis, analytical services, and peptide pre-formulation.
Peptide & Protein Capabilities

Almac provides services related to peptide synthesis and protein engineering including GMP manufacturing for clinical trials, a chemokine catalogue, fluorescence lifetime assays, and proprietary technologies for site-specific protein modification. They offer a complete package for first-in-man clinical trials including chemical and analytical development, material supply, oversight of formulation and clinical trials. Case studies demonstrate their work synthesizing modified chemokines and developing sterile drug products for clinical use.
More from ejmckee (7)
UK Facilities
- 1. UK Facilities non GMP API Manufacture & Stability Commercial Manufacture, Pack & Analysis GMP API Manufacture Pharmaceutical Development Clinical Packaging & Labelling Radio Labelling Diagnostics Stores & Distribution Ceph Manufacture & Pack Penicillin Manufacture & Pack