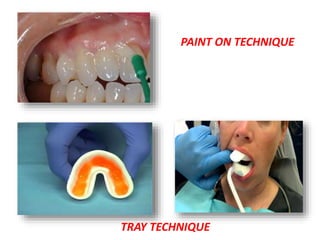
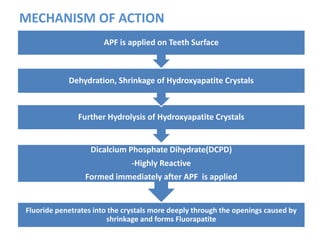
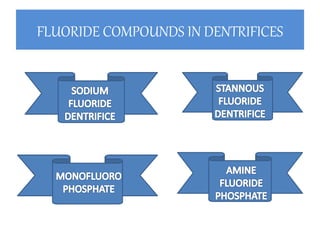
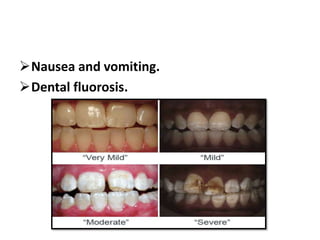
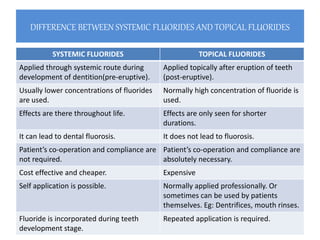
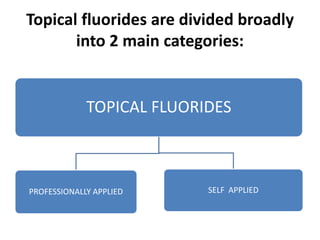
The document discusses the use of fluoride in dentistry, focusing on delivery methods, types of fluorides, and their applications in preventive dental care. It distinguishes between topical and systemic fluorides, and outlines the various types of topical fluorides, their indications, advantages, and techniques for application. The document emphasizes the importance of fluoride in preventing dental caries and provides a comprehensive overview of different fluoride agents used in clinical practice.



















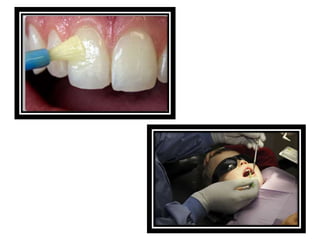
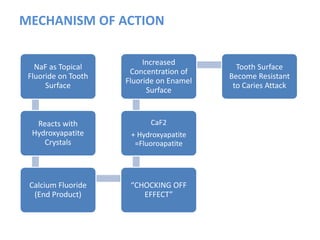




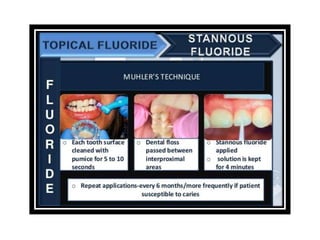
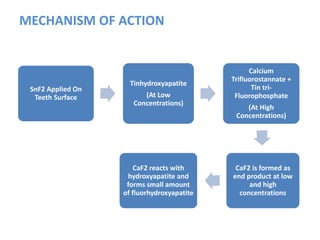


![ACIDULATED PHOSPHATE FLUORIDE (APF)
APF introduced in 1960’s by Brudevold and is his
co-workers at the Forsyth Dental Centre, Boston,
Massachusetts.
Fluoride concentration- 12300 ppm
PREPARATION:
An aq. solution of APF is prepared by dissolving
20 gms of sodium fluoride in 1 litre of 0.1 M
Phosphoric acid.
50% hydrofluoric acid is added to this to adjust
the pH at 3 and fluoride ion concentration at
1.23% [Brudevold’s Solution].](https://image.slidesharecdn.com/topicalfluorides2-190208052850/85/TOPICAL-FLUORIDES-33-320.jpg)