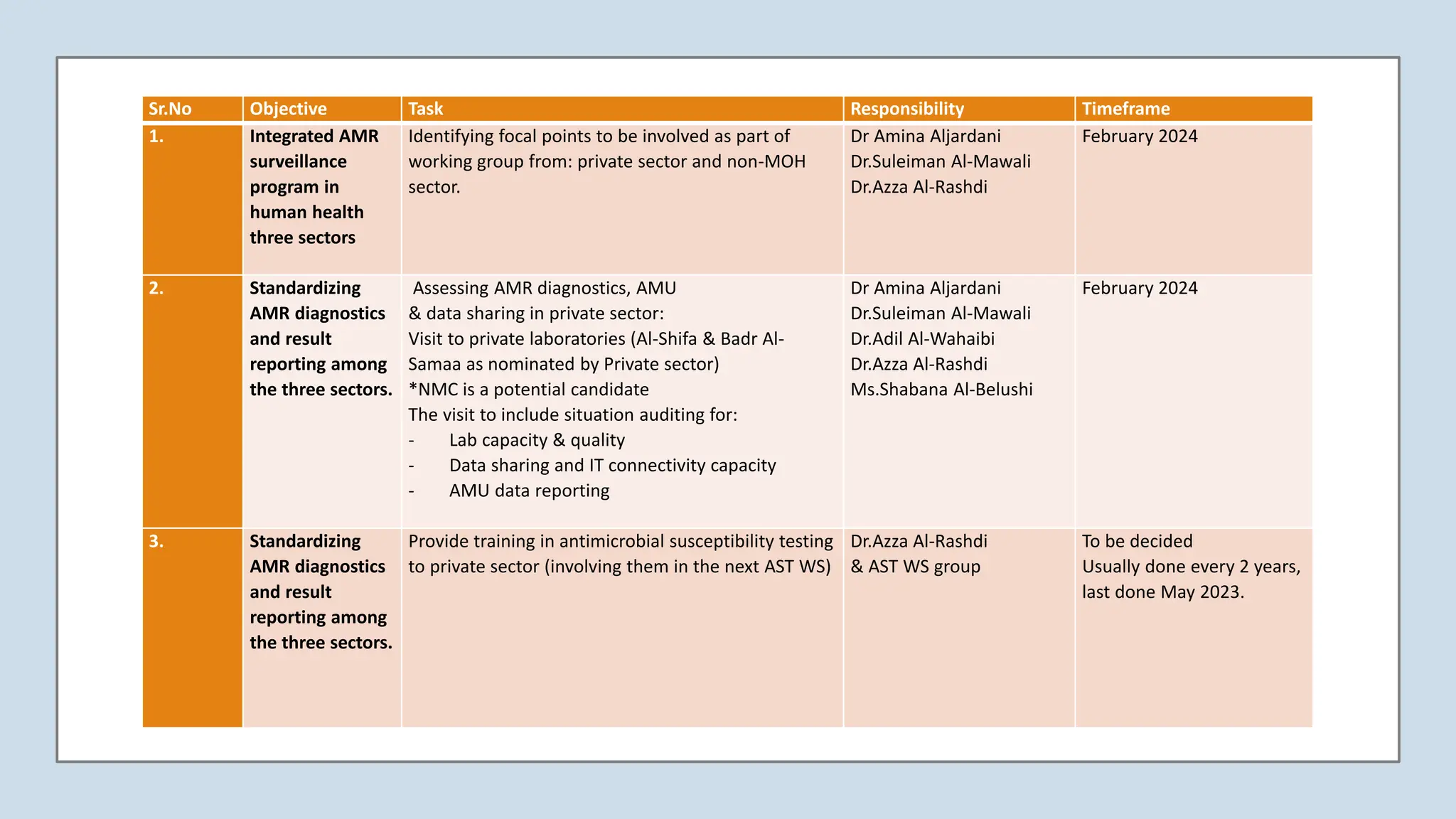
The Surveillance & Research Subcommittee aims to establish an integrated AMR surveillance program across human health sectors in Oman. Key goals include standardizing AMR diagnostics and reporting, expanding surveillance to non-MOH sectors and the environment, improving data sharing and connectivity, and establishing benchmarks. The subcommittee will work with private and non-MOH sectors, identify focal points, provide trainings, and assess capacities. It also seeks to create a research platform bringing together all relevant stakeholders to identify priorities, opportunities, and policies to assess the AMR burden and distribute research projects. Progress will be monitored through activity and outcome indicators established in the operational plan.