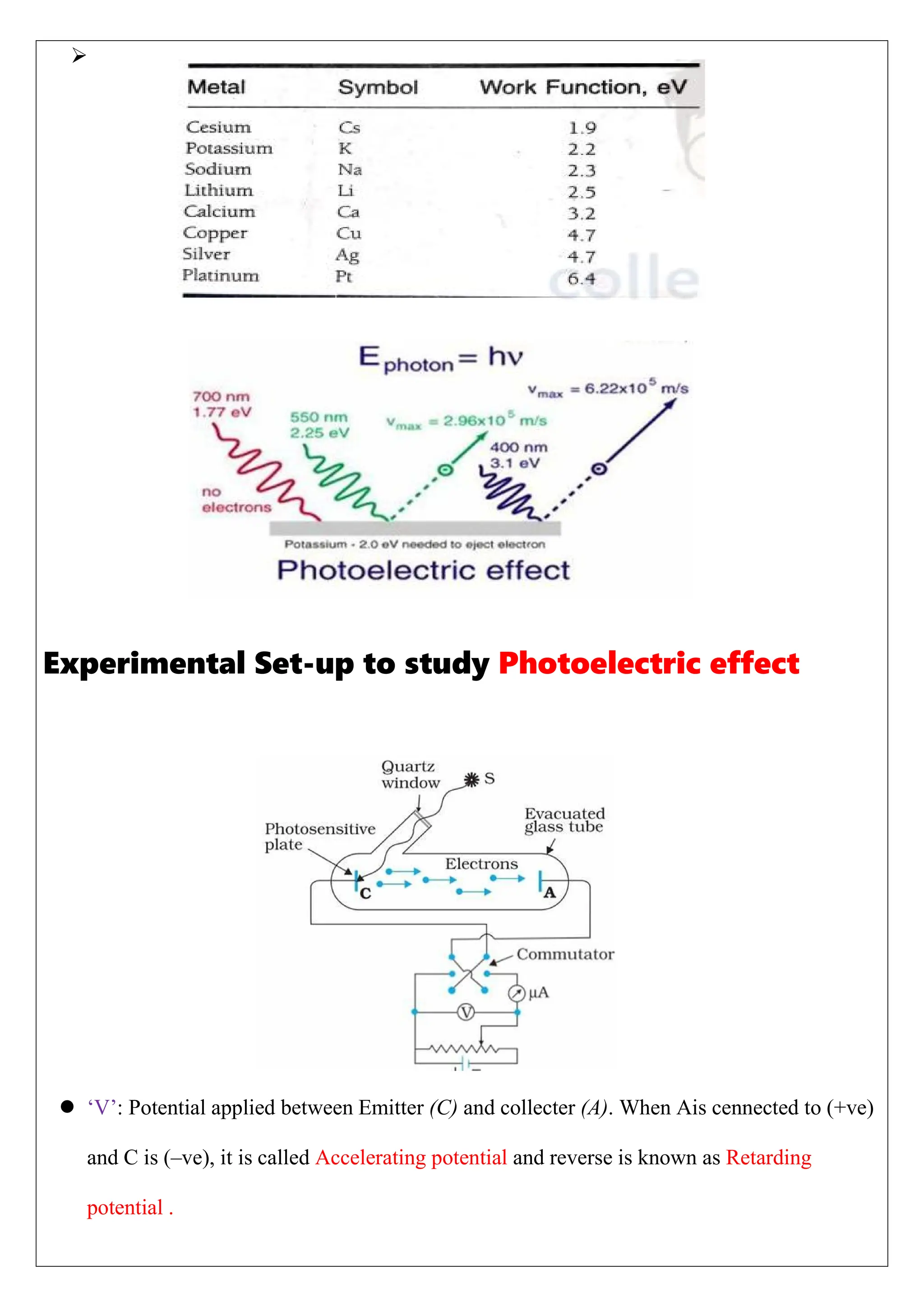
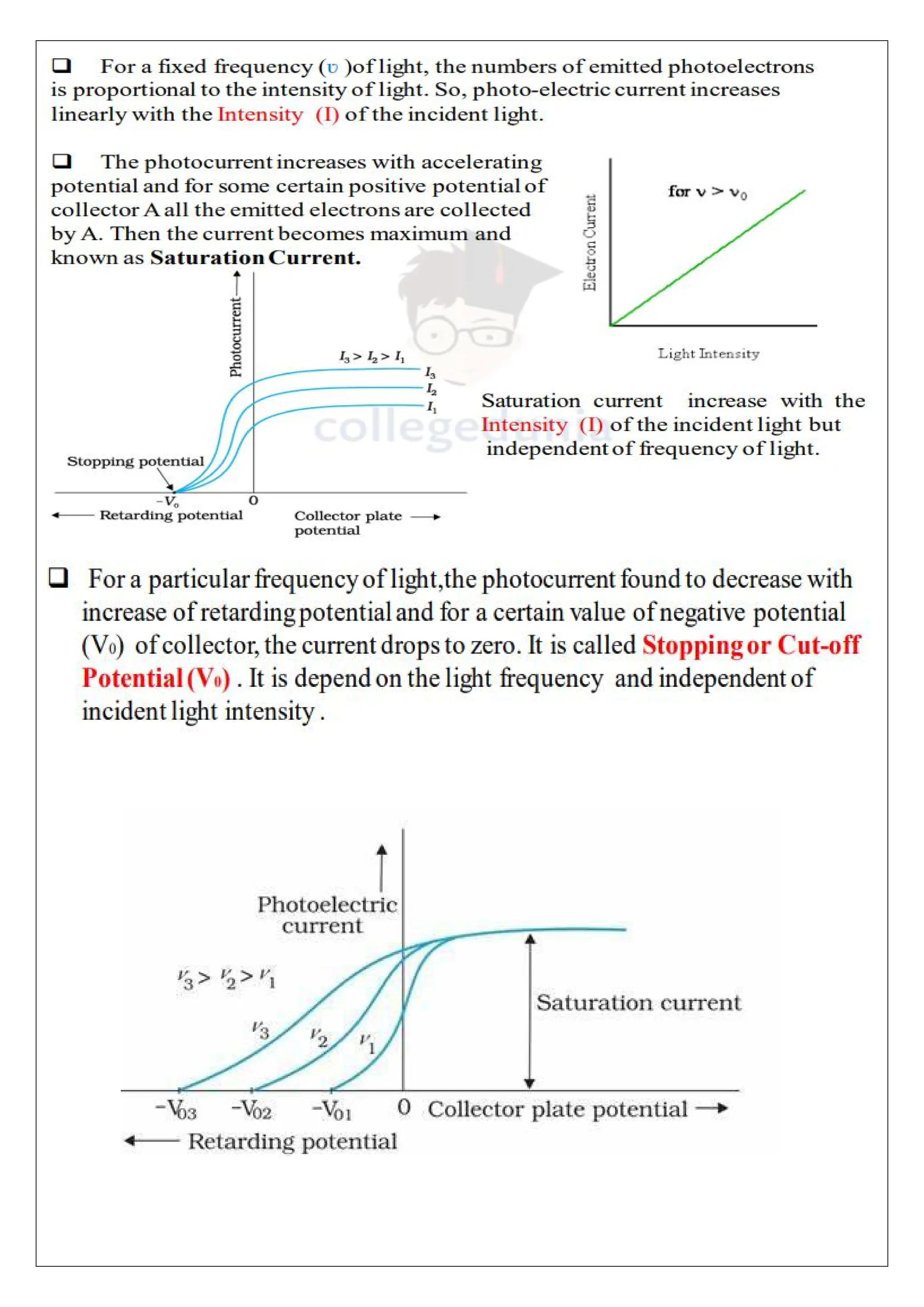
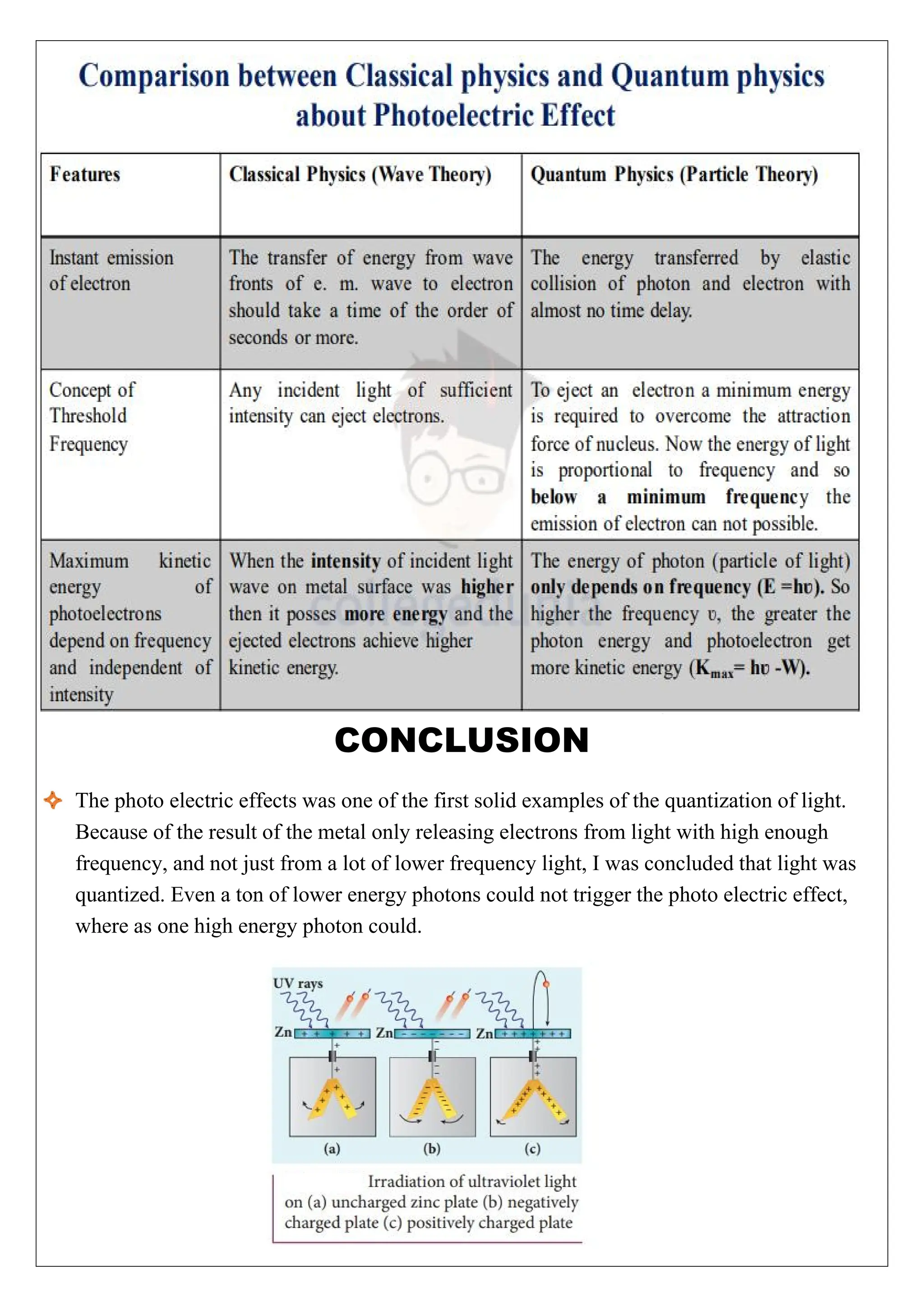
The photoelectric effect involves the emission of electrons from metal surfaces when exposed to light of suitable frequency, leading to the production of photoelectric current. First verified by Hertz in 1887, this phenomenon could not be explained by classical wave theory, prompting the development of quantum physics by Max Planck. Applications of the photoelectric effect include automatic alarms, light meters, and industrial measurements, with key laws detailing the relationship between light intensity, frequency, and electron emission energy.