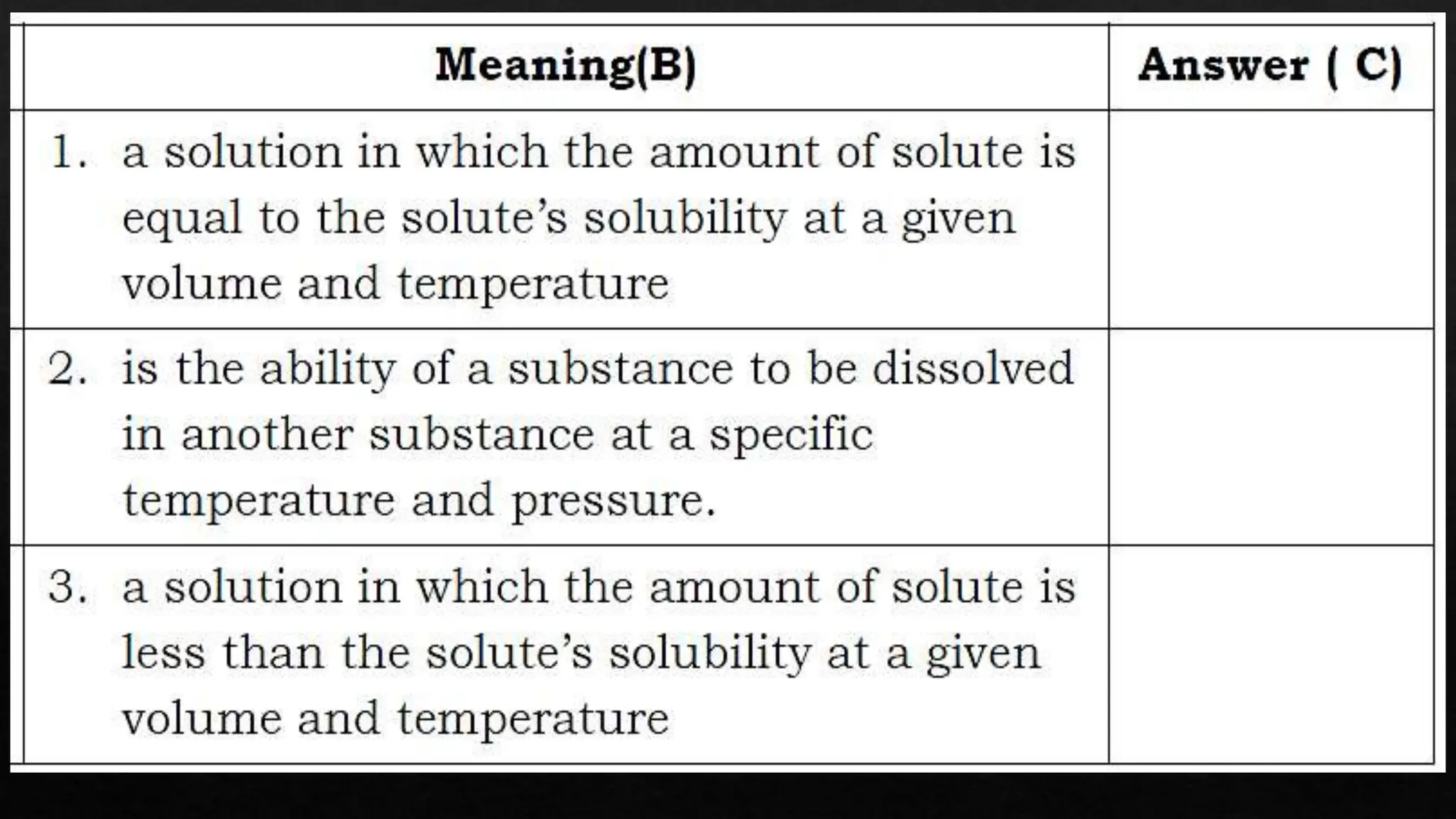
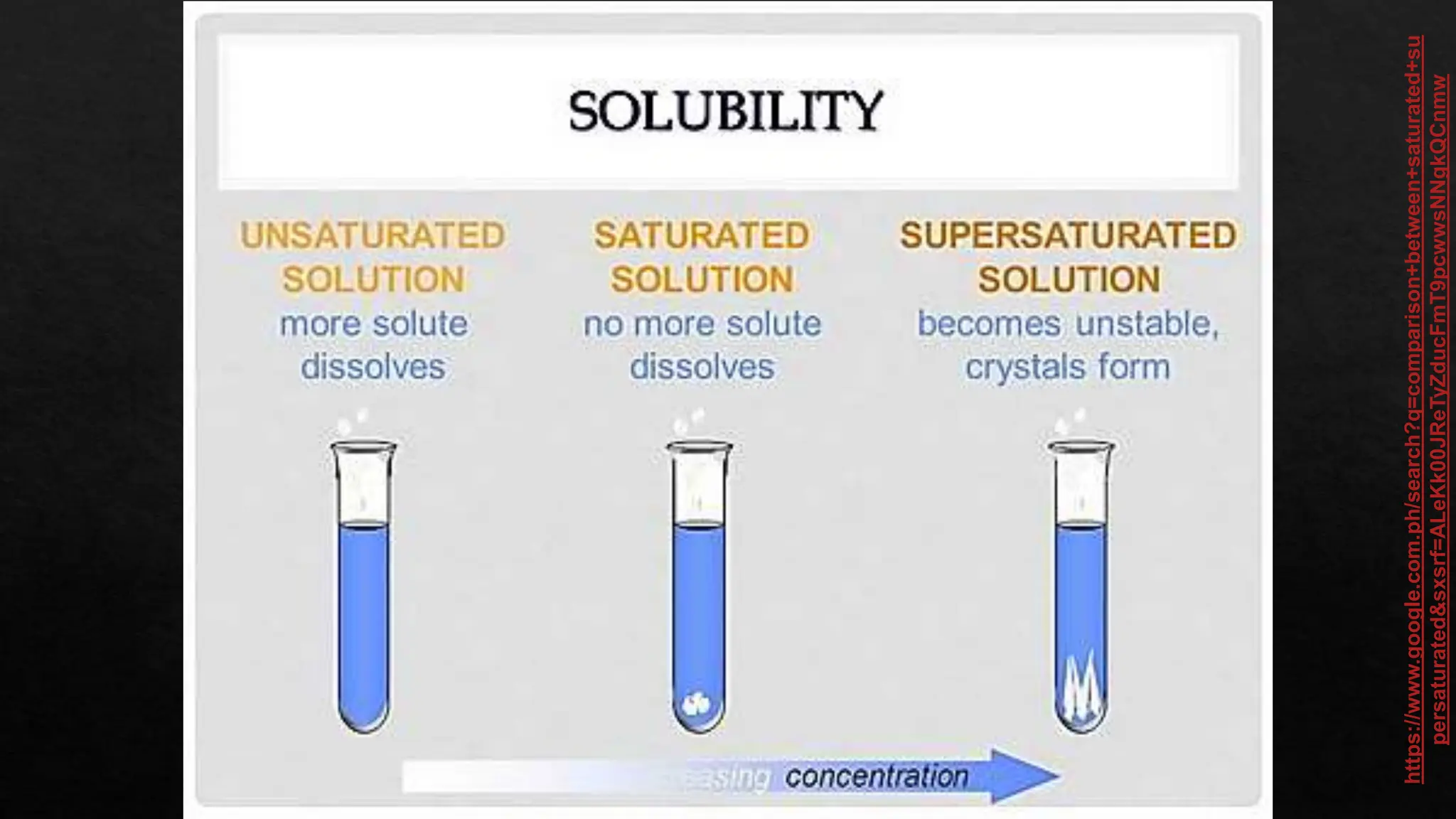
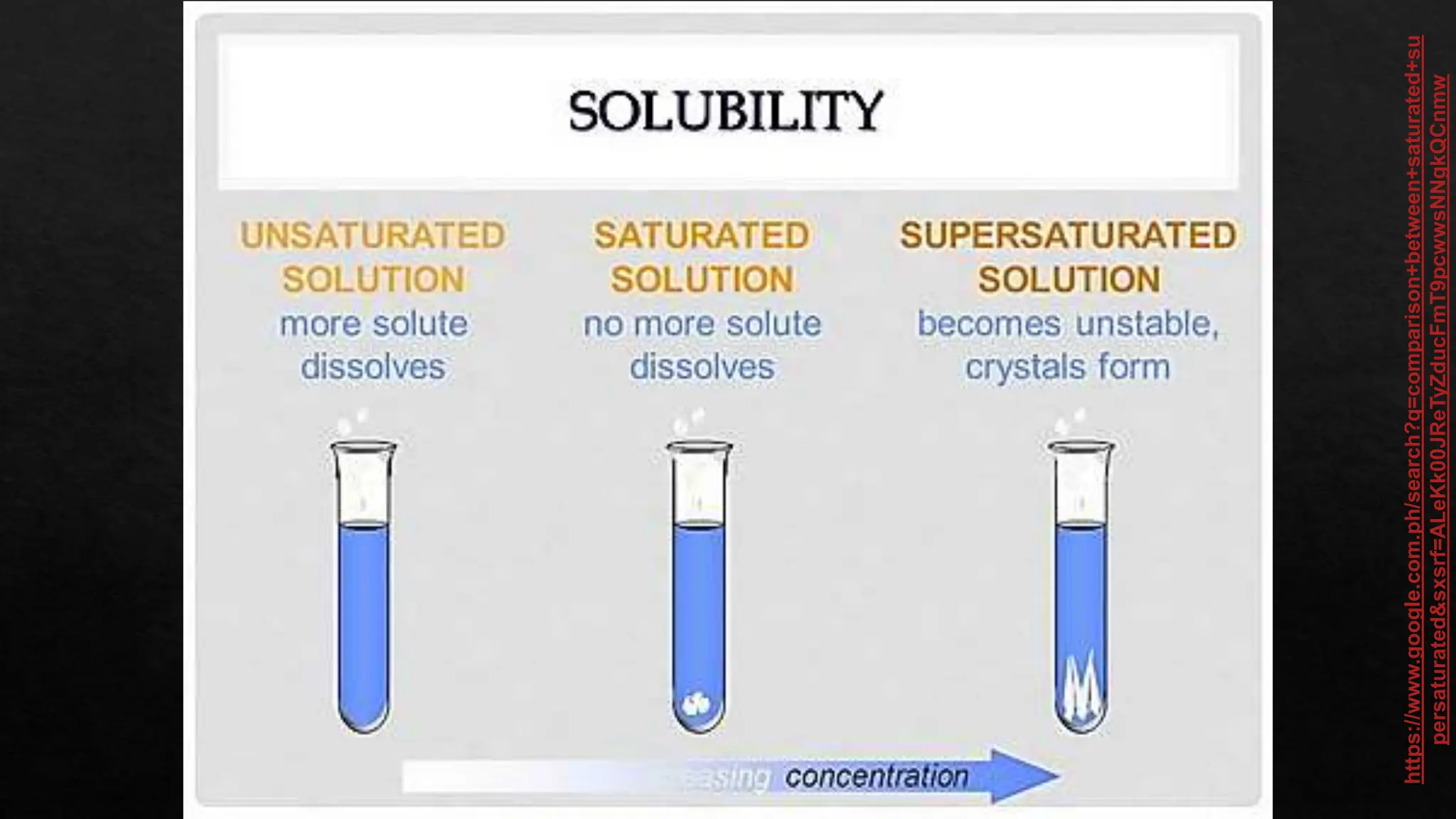
The document provides instructions for unscrambling words related to solubility concepts and matching them with definitions. It includes repeated directions for arranging scrambled words and a reference to online resources. Additionally, it features placeholder text that seems unrelated to the main task.