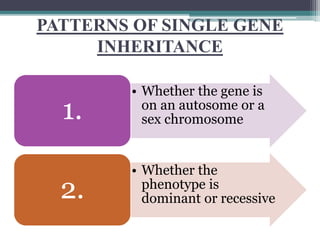
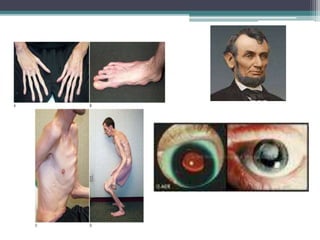
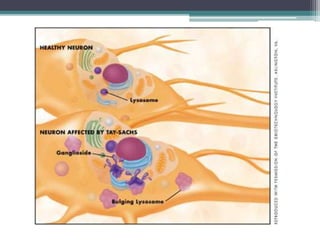
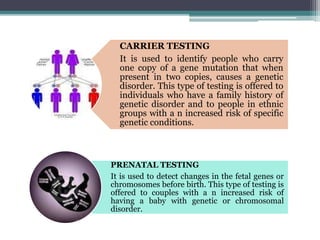
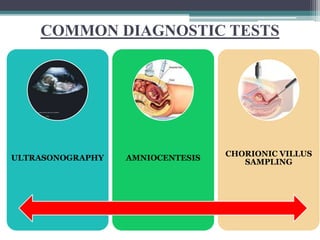
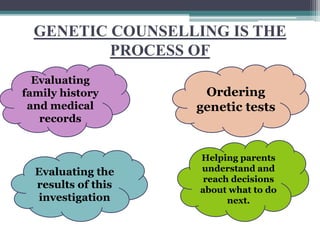
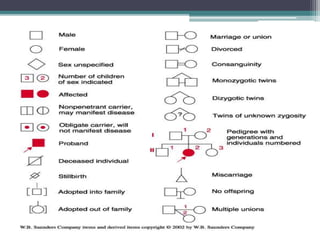
This document provides information on various genetics concepts including genes, chromosomes, inheritance patterns, and examples of genetic disorders. It discusses the basics of genetics including definitions of key terms like gene, chromosome, karyotype, genotype, phenotype, alleles, mutation, and inheritance patterns such as autosomal recessive, autosomal dominant, X-linked recessive, and examples for each. Specific genetic disorders discussed include cystic fibrosis, mucopolysaccharidoses, phenylketonuria, sickle cell disease, Tay-Sachs disease, and Duchenne muscular dystrophy.