Synapse Formation Between Newborn Granule Cells and Transplanted GABAergic Interneurons
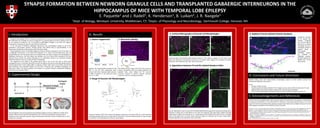
- 1. SYNAPSE FORMATION BETWEEN NEWBORN GRANULE CELLS AND TRANSPLANTED GABAERGIC INTERNEURONS IN THE HIPPOCAMPUS OF MICE WITH TEMPORAL LOBE EPILEPSY E. Paquette1 and J. Radell1, K. Henderson1, B. Luikart2, J. R. Naegele1 1Dept. of Biology, Wesleyan University, Middletown, CT; 2Depts. of Physiology and Neurobiology, Dartmouth College, Hanover, NH I. Introduction Temporal lobe epilepsy (TLE) is a disorder characterized by recurrent seizures persisting throughout life. Medications are available for seizure management, but approximately one third of patients with TLE do not respond to treatment. TLE is also linked to pathological changes in the brain and cognitive impairments; furthering the need for more effective treatments. In TLE, the death of inhibitory GABAergic interneurons and morphological changes to one of the population’s post-synaptic partners, dentate granule cells (DGCs) lead to hyperexcitability in the hippocampus (Kobayashi and Buckmaster 2003, Scharfman 2004). Neural progenitor cells in the Medial Ganglionic Eminence (MGE) of embryonic mice (E13.5) give rise to GABAergic interneurons in the adult brain (Maisano et al. 2012). By harvesting these cells and transplanting them into the hippocampus of an epileptic brain, we hope to replace the lost population of GABAergic interneurons. We have shown that epileptic mice that receive MGE transplants (TX) have reduced seizure number and severity (Henderson et al. Submitted). However, the mechanism through which this reduction occurs is in need of further investigation. The subgranular zone (SGZ) of the dentate gyrus (DG) is one of the few sites in which adult neurogenesis occurs in mammals. New DGCs are born throughout life and are continually being wired into existing hippocampal circuitry (Gage 2002). In epilepsy, newborn DGCs sprout mossy fibers, extend hilar basal dendrites, and ectopically migrate into the hilus, increasing their excitability (Kobayashi and Buckmaster 2003). This study examines the formation of synapses between transplanted GABAergic interneurons and newborn DGCs. II. Experimental Design Neural progenitor cells are harvested from the Medial Ganglionic Eminence (MGE) (C) of VGAT-Venus positive embryos (E13.5) ((A) GFP, (B) Brightfield, Scale 1mm) and transplanted into the DG (D) of epileptic mice via stereotaxic injection using a quintessential stereotaxic injector (100,000 cells in 1µl of media over 5 minutes). III. Results 1. Seizure Suppression Epileptic mice receiving MGE transplants in the hilus of the DG have significantly fewer seizures than those receiving media injections or MGE transplants in the entorhinal cortex (EC). (* p ≤ .001) B 2. Retroviral Labeling The pRubi retrovirus labels only cells in the process of mitosis. (A) Newborn DGCs are robustly labeled in the epileptic brain. (Scale 100µm) (B) The retroviral construct contains an mCherry reporter expressed under the control of a ubiquitin promoter. 3. Range of Granule Cell Morphologies Tracings of newborn DGCs from confocal micrographs containing a normotopic DGC (A), a DGC extending a hilar basal dendrite (B), and an ectopic DGC (C). (B) and (C) are known hyperexcitable cell types thought to contribute to the development of seizures. (Scale 20µm) 4. Confocal Micrographs of Granule Cell Morphologies Confocal images of the newborn DGCs depicted in figure 3. (RV: mCherry: magenta, TX: GFP: green, Scale 20µm) The proximity of transplanted interneurons to newborn DGCs suggests the possibility of synaptic interaction. (ML: Molecular layer, GCL: Granule Cell Layer) 5. Appositions between TX and RV Labeled Newborn DGCs (A) Low magnification view of a DG with retrovirally labeled DGCs (magenta) in the granule cell layer (GCL) (outlined in white) and TX cells (green) in the hilus and molecular layer (ML) (Scale 100µm). (B) High magnification image of the boxed area in A, containing several RV labeled DGCs (Scale 20 µm). (C) Digital zoom of the boxed area in B, which shows contacts between RV dendrites and TX axons. (Scale 20µm) Probable appositions are indicated in the Z-stack video. 6. Gephyrin Puncta Indicate Putative Synapses Appositions between an RV labeled DGC dendrite (magenta) and TX axons (green) depicted as a confocal micrograph (A) and a tracing (B). Clusters of gephyrin (blue), a post-synaptic scaffolding protein, are localized at GABAergic synapses. Probable synapses are labeled with arrows. (Scale 20µm) IV. Conclusions and Future Directions Newborn DGCs, some with hyperexcitable morphologies, appear to make synaptic contacts with transplanted GABAergic Interneurons Future: • Increase number of cases • Quantify synapse formation between TX and newborn versus mature DGCs to determine if TX cells preferentially contact hyperexcitable newborn DGCs with altered morphologies • Perform electrophysiological recordings of DGCs to confirm inhibitory synaptic input from TX V. Acknowledgements and References Thanks to the entire Naegele lab for their constant support and assistance on this project, to Nick Woods for laying the foundations, and to Jeff Gilarde for sharing his microscopy expertise with us. This work would not have been possible without generous support from Randy and Lisa Siegel and grants from the CURE Epilepsy Foundation and the National Institute of Health. Kobayashi, M. and P. S. Buckmaster (2003). Reduced inhibition of dentate granule cells in a model of temporal lobe epilepsy. J Neurosci 23(6): 2440-2452 Scharfman, H. E. (2004). Functional implications of seizure-induced neurogenesis. Advances in Experimental Medicine and Biology, 548: 192-212. Maisano, X., Litvina, E., Tagliatela, S., Aaron, G.B., Grabel, L.B., & Naegele, J.R. (2012). Differentiation and functional incorporation of embryonic stem cell derived GABAergic interneurons in the dentate gyrus of mice with temporal lobe epilepsy. The Journal of neuroscience: the official journal of the Society for Neuroscience, 32(1):46-61. Henderson, K. W., Gupta, J., Tagliatela, S., Litvina, E., Zheng, X. T., Van Zandt, M. A., Woods, N., Grund, E., Lin, D., Royston, S., Yanagawa, Y., Gloster, A. B., Naegele, J. R. (Submitted) Long-Term Seizure Supression and Optogenetic Analyses of Synaptic Connectivity in Epileptic Mice with Hippocampal Grafts of GABAergic Interneurons Gage F. H. Neurogenesis in the adult brain. J Neurosci 22: 612– 613, 2002. mCherry / GFP / Gephyrin