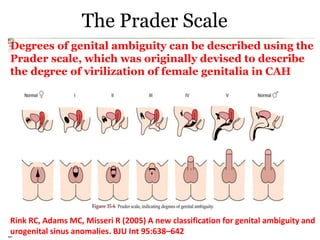
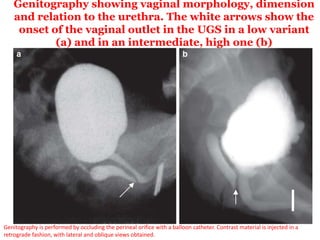
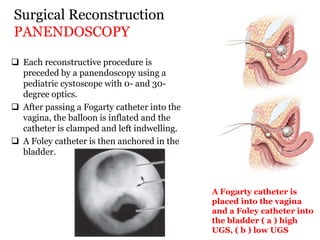
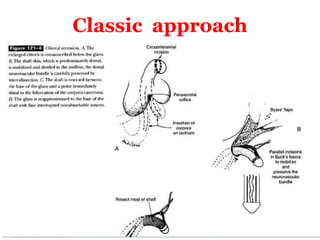
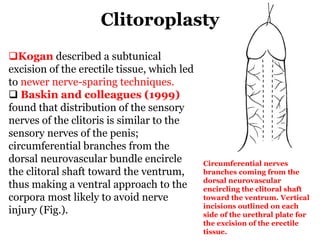
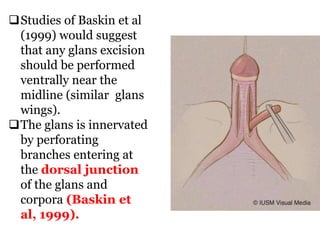
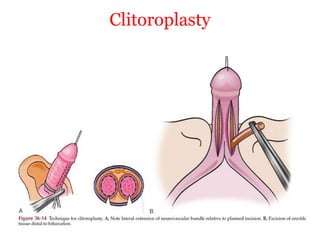
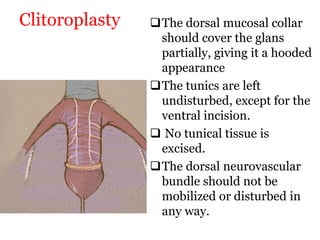
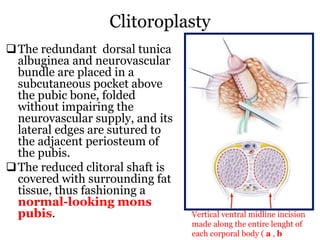
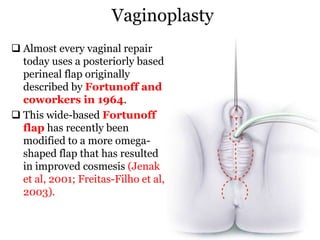
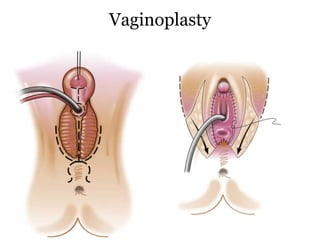
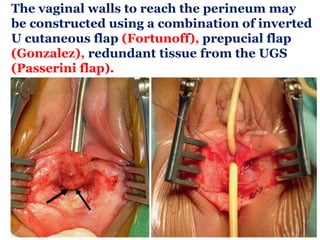
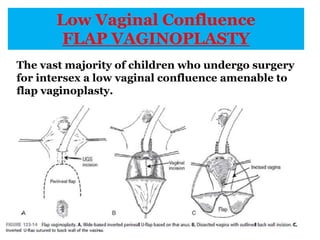
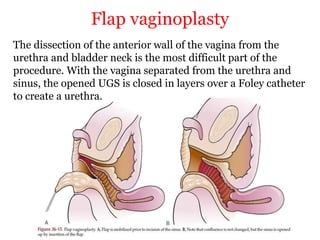
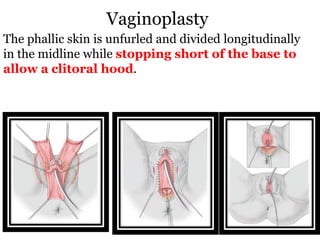
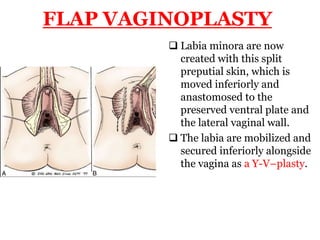
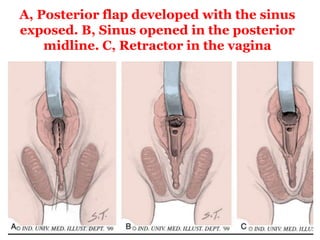
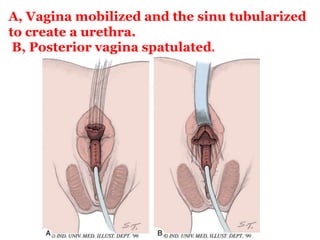
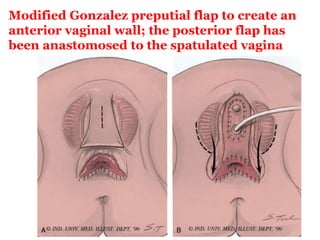
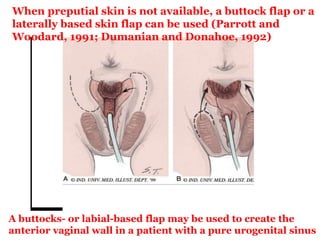
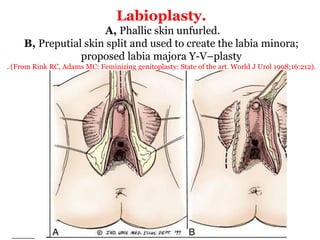
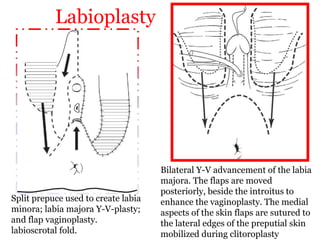
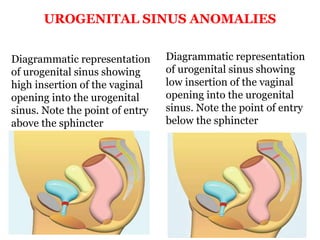
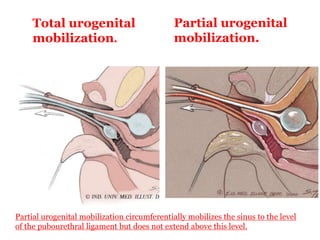
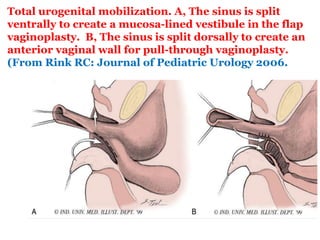
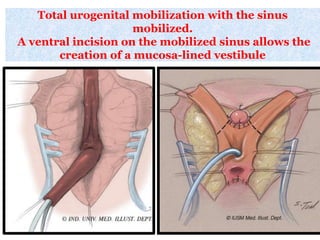
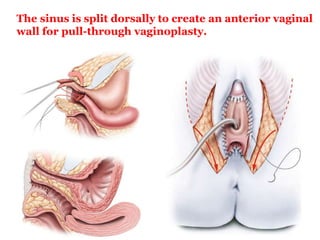
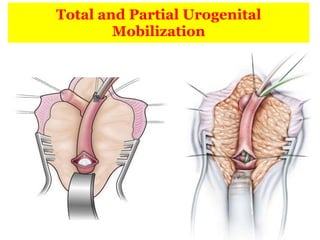
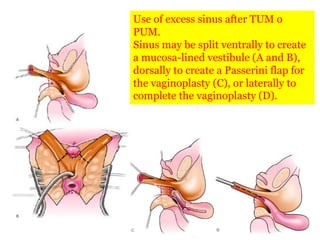
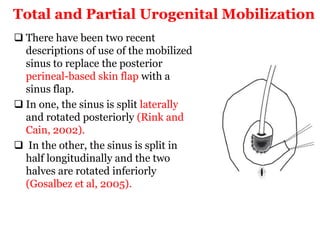
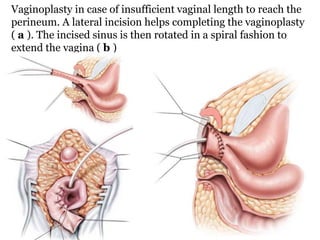
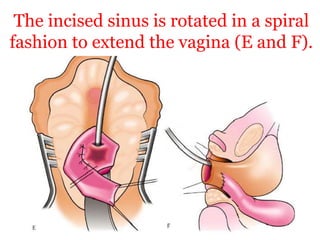
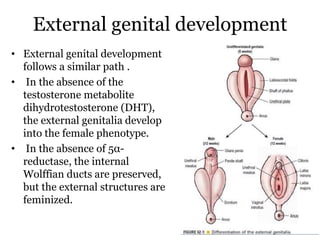
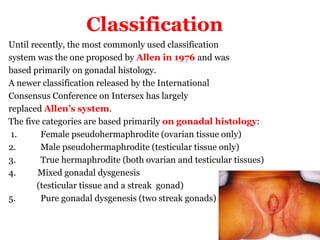
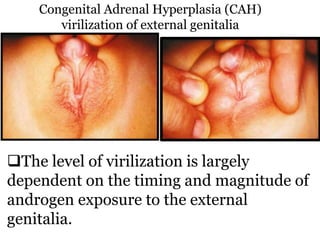
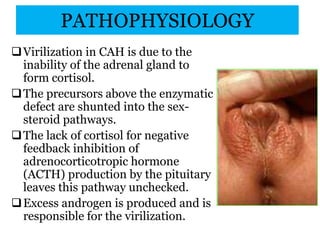
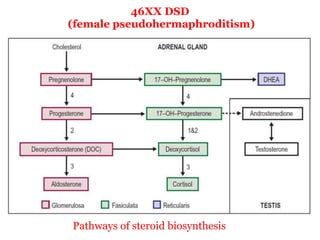
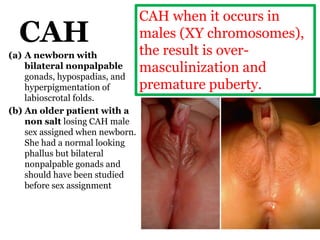
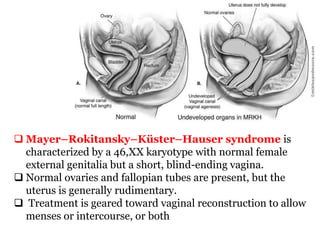
The document discusses disorders of sexual differentiation (DSD) characterized by atypical chromosomal, gonadal, and phenotypic sex leading to variations in the development of the urogenital tract and reproductive system. It describes embryological development, incidence rates, classification systems, and key genetic factors involved in DSD, along with diagnostic approaches and clinical evaluation of affected newborns. The document emphasizes the need for a multidisciplinary team approach for evaluation and management of these conditions.

![الرحيم الرحمن هللا بسم
ذَيَف ُدَبَّالز اَّمَأَفَّمَأ َو ًءاَفُج ُبَهاَم ا
مَيَف َاسَّنال ُعَفنَيِضرَألا يِف ُثُك
ُ َّاّلل ُب ِرضَي َكِلَذَكَلاَثمَألا
(17﴾[الرعد]
Wednesday, February 14, 2018 DR. ASHRAF HAMED 2](https://image.slidesharecdn.com/dsdupdate-180214055404/85/Dsd-update-2-320.jpg)


















![Rapid Diagnostic Algorithm
These two diagnostic criteria—presence or absence of Y
chromosome and gonadal symmetry or asymmetry—
allow the rapid, accurate assignment of a patient into
one of the four diagnostic categories with
approximately 90% accuracy.
Y Chromosome PresentY Chromosome Absent or
Abnormal
AsymmetrySymmetryAsymmetrySymmetry
46,XX/46,XY
MGD
[Mixed
gonadal
dysgenesis]
46,XY
DSD
Ovotesticular
DSD
46,XX DSD
[congenital
adrenal
hyperplasia]](https://image.slidesharecdn.com/dsdupdate-180214055404/85/Dsd-update-29-320.jpg)