The document discusses blood transfusion, including its history, processes, components, indications, and complications. It describes how blood is collected, tested, separated into components like red blood cells, plasma, and platelets. It provides details on appropriate storage, administration, and indications for transfusion of each component. Complications of transfusion and the importance of blood typing and cross-matching to prevent transfusion reactions are also summarized.

![The first successful transfusion of human blood was performed in 1818 by a British
obstetrician, Dr James Blundell, who drew blood from the patient’s husband and, to prevent
the blood from coagulating ex vivo, infused it directly into the patient
BLOOD TRANSFUSION
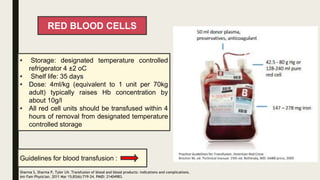
Blood transfusion begins with
collecting whole blood from a donor using a
preservative-anticoagulant combination
(typically citrate phosphate dextrose or
citrate phosphate dextrose adenine-1).
The blood is then tested for transfusion-
transmitted diseases, separated into its
specific components, and then transfused
as clinically indicated.
Zomorrodi A, Picciola EA, Hotwagner DT. Determining the Need for Blood Transfusion. [Updated 2023 Jun 8]. In: StatPearls
[Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from:](https://image.slidesharecdn.com/crossmatchtransfusi-240403154046-bc14c5ff/85/Crossmatch-Transfusion-Procedure-Hospital-2-320.jpg)


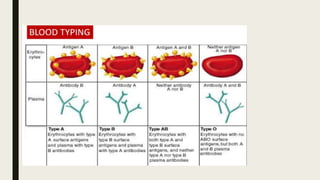
![BLOOD TYPE AND CROSS MATCH
To avoid a transfusion reaction, donated blood must be compatible with the blood of the
patient who is receiving the transfusion. The donated RBCs must lack the same ABO and Rh
D antigens that the patient's RBCs lack
Before transfusion
Blood type Cross match
the recipient's blood type is determined,
their ABO type and Rh D status
may still be incompatible because it contains other
antigens that are not routinely typed but may still
cause a problem if the recipient's serum contains
antibodies that will target them
to ensure that the donor RBCs actually
do match against the recipient's serum
Dean L. Blood Groups and Red Cell Antigens
[Internet]. Bethesda (MD): National Center for
Biotechnology Information (US); 2005. Chapter 3,
Blood transfusions and the immune system.
Available from:
https://www.ncbi.nlm.nih.gov/books/NBK2265/](https://image.slidesharecdn.com/crossmatchtransfusi-240403154046-bc14c5ff/85/Crossmatch-Transfusion-Procedure-Hospital-5-320.jpg)
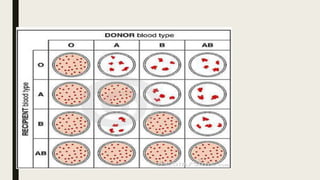
![CROSS MATCH
To perform a cross match, a small
amount of the recipient's serum is mixed
with a small amount of the donor RBCs
Dean L. Blood Groups and Red Cell Antigens [Internet]. Bethesda (MD): National Center for
Biotechnology Information (US); 2005. Chapter 3, Blood transfusions and the immune
system. Available from: https://www.ncbi.nlm.nih.gov/books/NBK2265/
If the proposed transfusion is
incompatible, the donor RBCs are
agglutinated by antibodies in the
recipient's serum.](https://image.slidesharecdn.com/crossmatchtransfusi-240403154046-bc14c5ff/85/Crossmatch-Transfusion-Procedure-Hospital-7-320.jpg)







![WHOLE BLOOD
There has been increasing interest in using low titer group O whole blood (LTOWB) in military and
civilian trauma, and there is evidence to show that it saves lives.
LTOWB provides all of the components of blood (RBCs, platelets, and plasma with fibrinogen) and
provides a balanced resuscitation addressing oxygen needs and coagulopathy in a single bag of
blood.
The transfusion of up to 4 units of whole blood has been shown to be safe.
Zomorrodi A, Picciola EA, Hotwagner DT. Determining the Need for Blood Transfusion. [Updated 2023 Jun 8]. In:
StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from:
The whole blood has a critical titer of anti-A and anti-B of less than 50 to 200](https://image.slidesharecdn.com/crossmatchtransfusi-240403154046-bc14c5ff/85/Crossmatch-Transfusion-Procedure-Hospital-19-320.jpg)