CHEMISTRY
•
0 likes•318 views
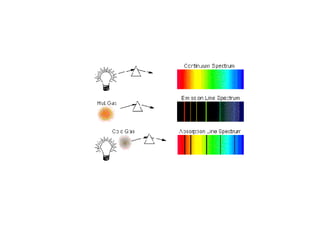
The hydrogen atom can only exist at certain quantized energy levels called stationary states. An atom does not emit energy when in one of these stationary states. An atom can change between stationary states by absorbing or emitting a photon with energy equal to the difference between the two states.
Report
Share
Report
Share
Recommended
More Related Content
Viewers also liked
Viewers also liked (10)
What's the Difference Between Publicity and Public Relations?

What's the Difference Between Publicity and Public Relations?
More from Mark Rabena
More from Mark Rabena (11)
Recently uploaded
Paddle, Float, and Explore The Ultimate River Tour Experience in Monitor, WA

Paddle, Float, and Explore The Ultimate River Tour Experience in Monitor, WARiver Recreation - Washington Whitewater Rafting
Recently uploaded (15)
Explore India Buddhist Pilgrimage Tour Packages with IRCTC Buddhist Circuit.pptx

Explore India Buddhist Pilgrimage Tour Packages with IRCTC Buddhist Circuit.pptx
Paddle, Float, and Explore The Ultimate River Tour Experience in Monitor, WA

Paddle, Float, and Explore The Ultimate River Tour Experience in Monitor, WA
Dummy Ticket l Dummy Flight Ticket l Get At $5/INR350

Dummy Ticket l Dummy Flight Ticket l Get At $5/INR350
Unveiling the National Logistics Policy (NLP) and navigating India's economic...

Unveiling the National Logistics Policy (NLP) and navigating India's economic...
Agence Régionale du Tourisme Grand Est - brochure MICE 2024.pdf

Agence Régionale du Tourisme Grand Est - brochure MICE 2024.pdf
TRAVEL TO MT. RWENZORI NATIONAL PARK WITH NILE ABENTEUER SAFARIS.docx

TRAVEL TO MT. RWENZORI NATIONAL PARK WITH NILE ABENTEUER SAFARIS.docx
Hunza Cherry Blossom tour 2025- Hunza Adventure Tours

Hunza Cherry Blossom tour 2025- Hunza Adventure Tours
Exploring Montreal's Artistic Heritage Top Art Galleries and Museums to Visit

Exploring Montreal's Artistic Heritage Top Art Galleries and Museums to Visit
Présentation About Algiers City (noureddine boureneb)

Présentation About Algiers City (noureddine boureneb)
Sinister History Exploring Austin’s Past Through a Night Walking Tour

Sinister History Exploring Austin’s Past Through a Night Walking Tour
Unveiling New Mexico Santa Fe and Taos Adventures Await.pptx

Unveiling New Mexico Santa Fe and Taos Adventures Await.pptx
Exploring Heritage The Ultimate Cultural Tour in Palmer, Puerto Rico

Exploring Heritage The Ultimate Cultural Tour in Palmer, Puerto Rico
CHEMISTRY
- 4. The hydrogen atom has only certain allowable energy levels which he called as stationary states.
- 5. The atom does not radiate energy while in one of its stationary states.
- 6. The atom changes to another stationary state only by absorbing or emitting a photon whose energy equals the difference in energy between the two states. It cannot lie in between two steps.
