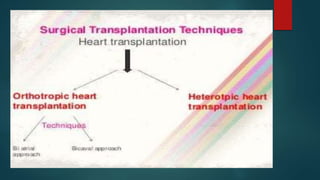
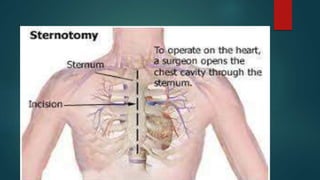
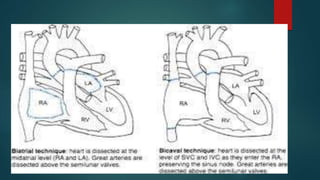
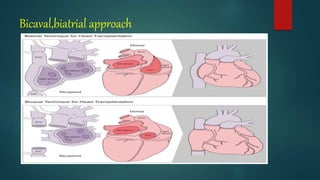
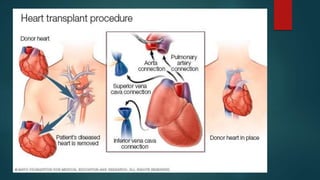
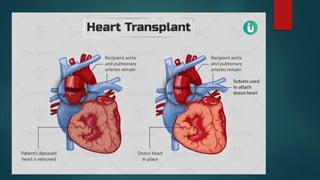
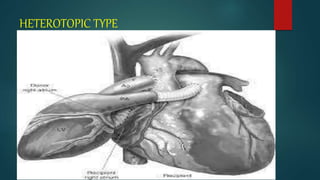
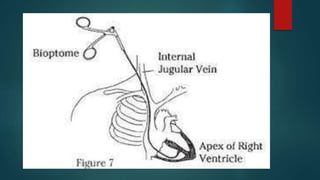
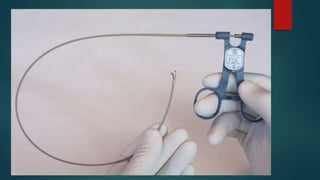
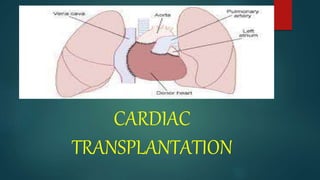
This document provides information about cardiac transplantation:
- Cardiac transplantation involves removing a diseased heart and replacing it with a healthy donor heart. It has improved survival rates but also risks that require careful monitoring and treatment.
- The history of cardiac transplantation is reviewed, including early experiments in dogs in the 1900s, the first human-to-human transplant in 1967, and the progression of the field since then globally and in India.
- Patient evaluation criteria and listing for transplantation are outlined, as well as management of patients awaiting transplant. The donor selection process and criteria are also described.

![HISTORY[global]
Carrel and Guthrie first reported successful heterotopic cardiac
transplantation in dogs in 1905 .
1913 Mann and colleagues at Mayo Clinic reported successful
transplantation of the heart into the neck of dogs.
Medawar was the first to develop concepts of immunology applicable to
Transplantation.
Lower and Shumway first reported successful experimental orthotopic
cardiac transplantation in 1960.
Orman Shumway is widely regarded as the father of heart
transplantation although the world's first adult human heart transplant
was performed by a South African cardiac surgeon,Christiaan Barnard,
utilizing the techniques developed and perfected by Shumway and
Richard Lower.
In 1964, Hardy and colleagues performed the first heart transplant into
a human, using a chimpanzee heart.
The first human-to-human heart transplant (allograft) was performed in
Cape Town, South Africa, by on December 3, 1967.
The recipient was Louis Washkansky, a 53-yearChristiaan Barnard -old](https://image.slidesharecdn.com/cardiactransplantation-240319181429-6358c342/85/cardiac-transplantation-pptx-surgeries-4-320.jpg)

![History [india]
At KEM Hospital, Mumbai, Dr PK Sen and his team performed
the first heart transplant in India in February 1968.
The Organ Transplant Bill 1994 was passed in the Indian
Parliament in May 1994 which cleared the way for organ harvest
from brain-dead patients.
Successively, Dr P Venugopal and his team performed the first
successful heart transplant on August 3, 1994. Hyderabad first
transplant was done in global hospital.
The full extent of the law and notification happened in 1995 after
which other centres in India performed the surgery successfully.
6 heart transplants have been done in NIMS ,INDIA .
Collectively, 129 heart transplants have been performed in India
since 1994 with 82 in Chennai, 33 at AIIMS and 14 at other
centres in India.](https://image.slidesharecdn.com/cardiactransplantation-240319181429-6358c342/85/cardiac-transplantation-pptx-surgeries-6-320.jpg)
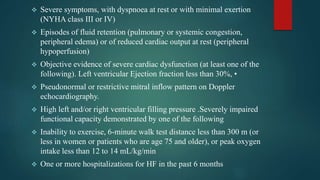









![Criteria For Determining Brain Death Clinical
Evaluation
Mechanism of brain injury is sufficient to account for irreversible loss of
brain function
Absence of reversible causes of CNS depression
CNS depressant drugs
Hypothermia (<32°C [85°F]) ,Hypotension (MAP <55 mmHg)
Absence of neuromuscular blocking drugs that may confound the results
of the neurologic exam
No spontaneous movements, motor responses, or posturing](https://image.slidesharecdn.com/cardiactransplantation-240319181429-6358c342/85/cardiac-transplantation-pptx-surgeries-19-320.jpg)






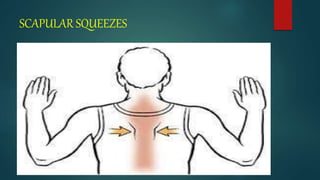
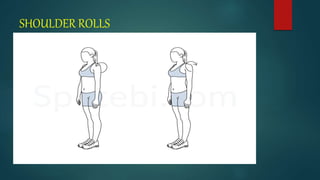
![PREOPERATIVE TEACHING
We should teach the patient to increase his health by giving advices like
(a) stop smoking,
(b) maintain personal hygiene,
(c) deep breathing and coughing exercises
[d]active and passive exercise
SURGICAL PREPARATION OF THE SKIN
Skin preparation helps reduce the number of microorganism present on skin
and thus reduce the possibility of wound infection.
Shave the area and clean the area with antiseptic lotion/swab](https://image.slidesharecdn.com/cardiactransplantation-240319181429-6358c342/85/cardiac-transplantation-pptx-surgeries-27-320.jpg)