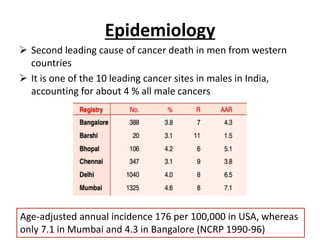
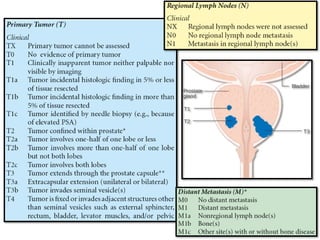
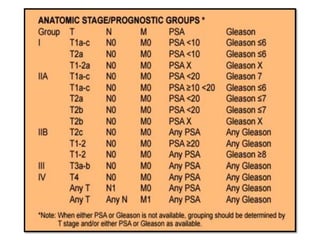
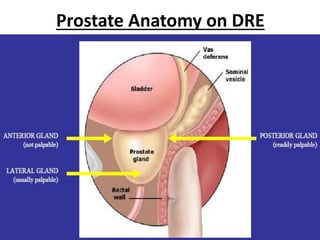
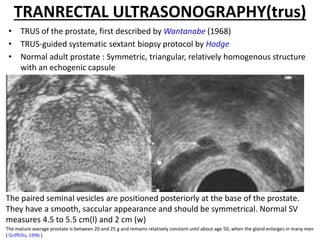
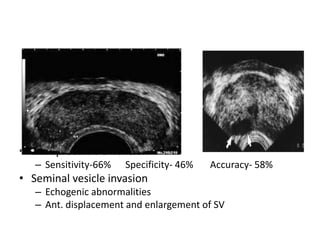
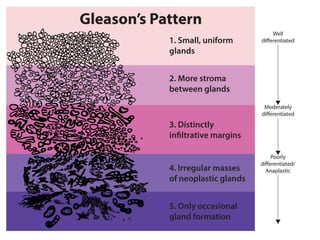
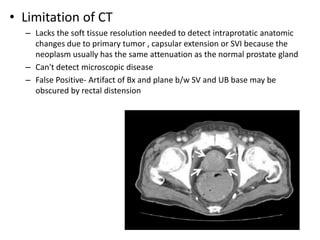
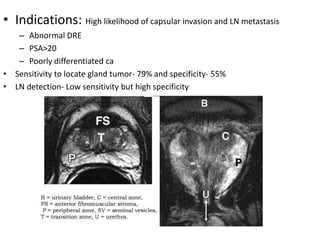
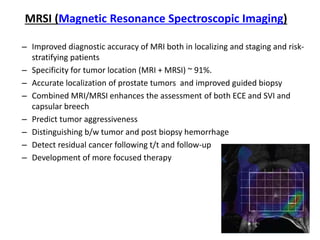
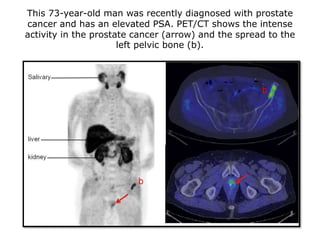
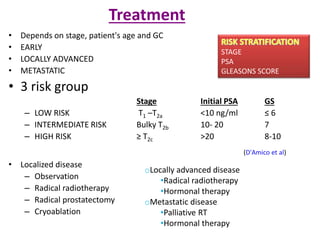
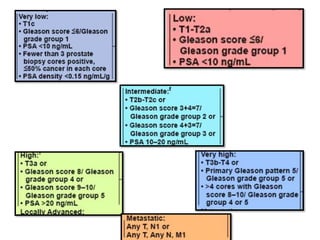
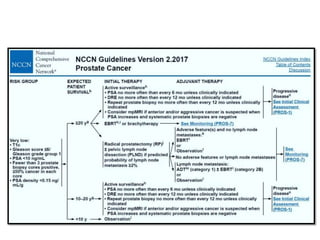
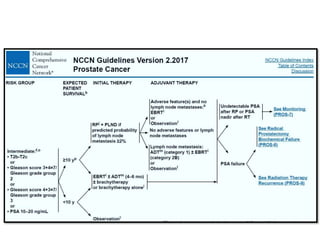
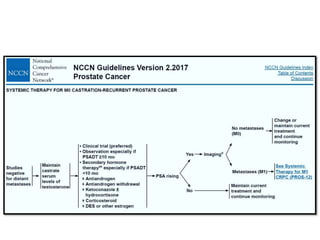
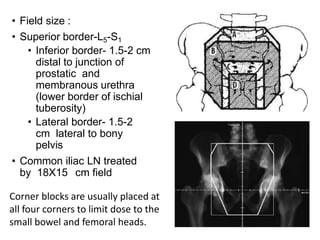
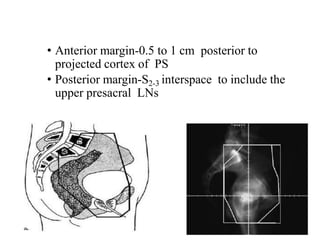
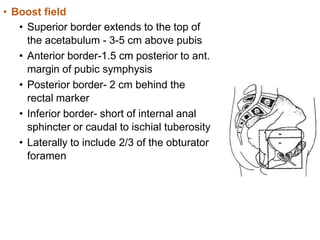
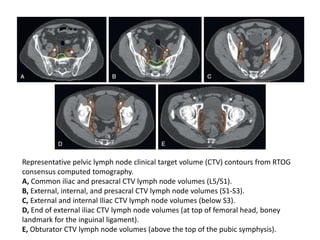
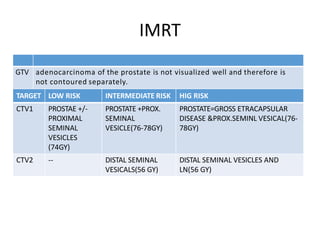
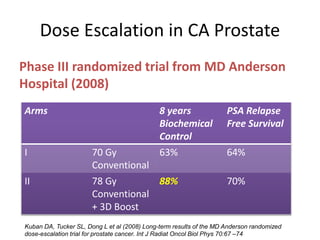
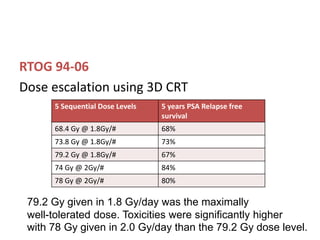
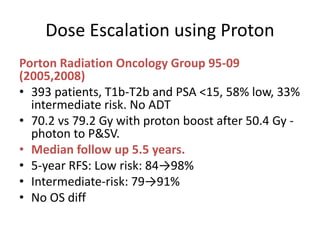
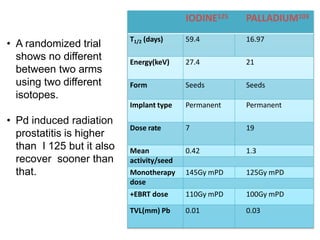
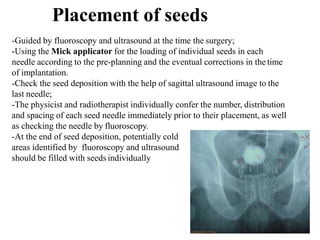
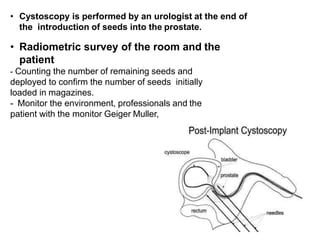
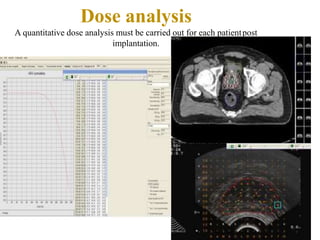
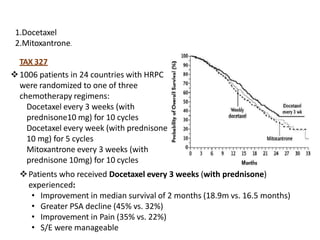
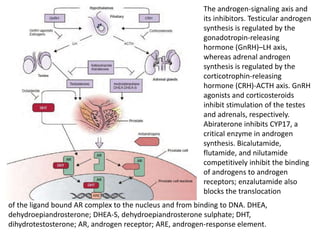
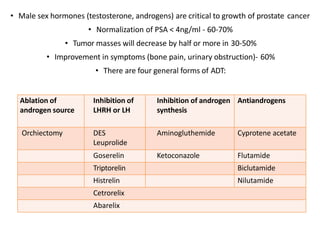
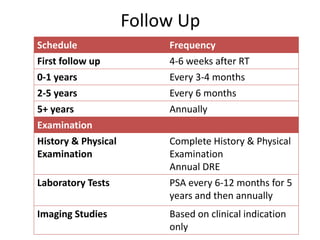
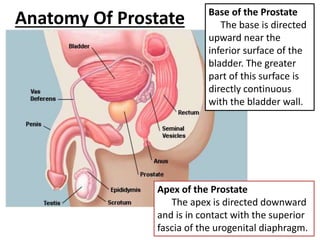
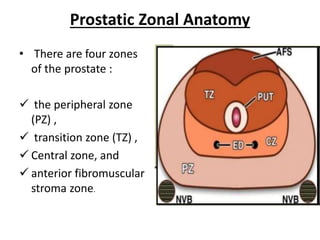
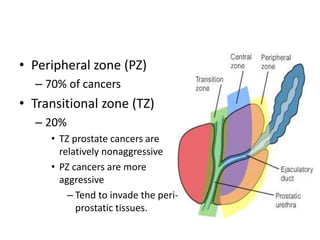
The document discusses the anatomy and diagnostic evaluation of prostate cancer. It describes the prostate as a walnut-sized gland located below the bladder and surrounding the urethra. The primary function is to produce seminal fluid. Diagnostic workup involves PSA levels, digital rectal exam, prostate biopsy and various imaging modalities like CT, MRI, bone scan and PSMA PET/CT to stage disease extent and metastasis. Gleason scoring is used to grade prostate cancer based on architectural patterns seen on biopsy.



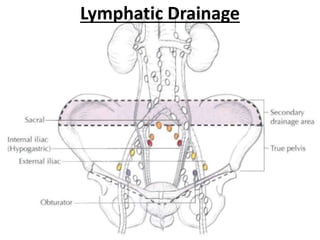
![Regional Lymph Nodes
• Pelvic
• Hypogastric
• Obturator
• Iliac (internal, external)
• Sacral (lateral, presacral, promontory
[Gerota's])](https://image.slidesharecdn.com/finalcaprostate-180114163813/85/CA-Prostate-8-320.jpg)