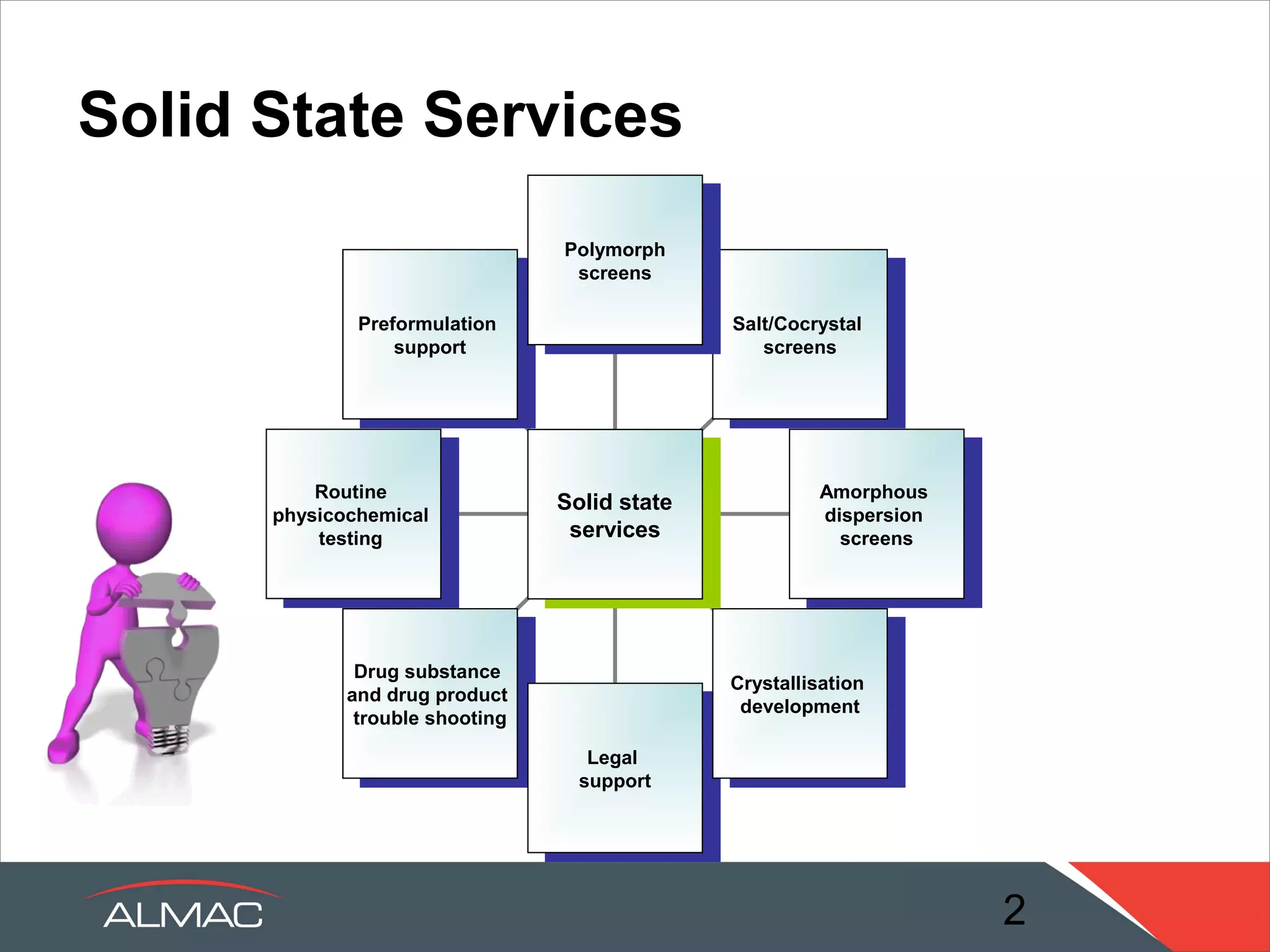
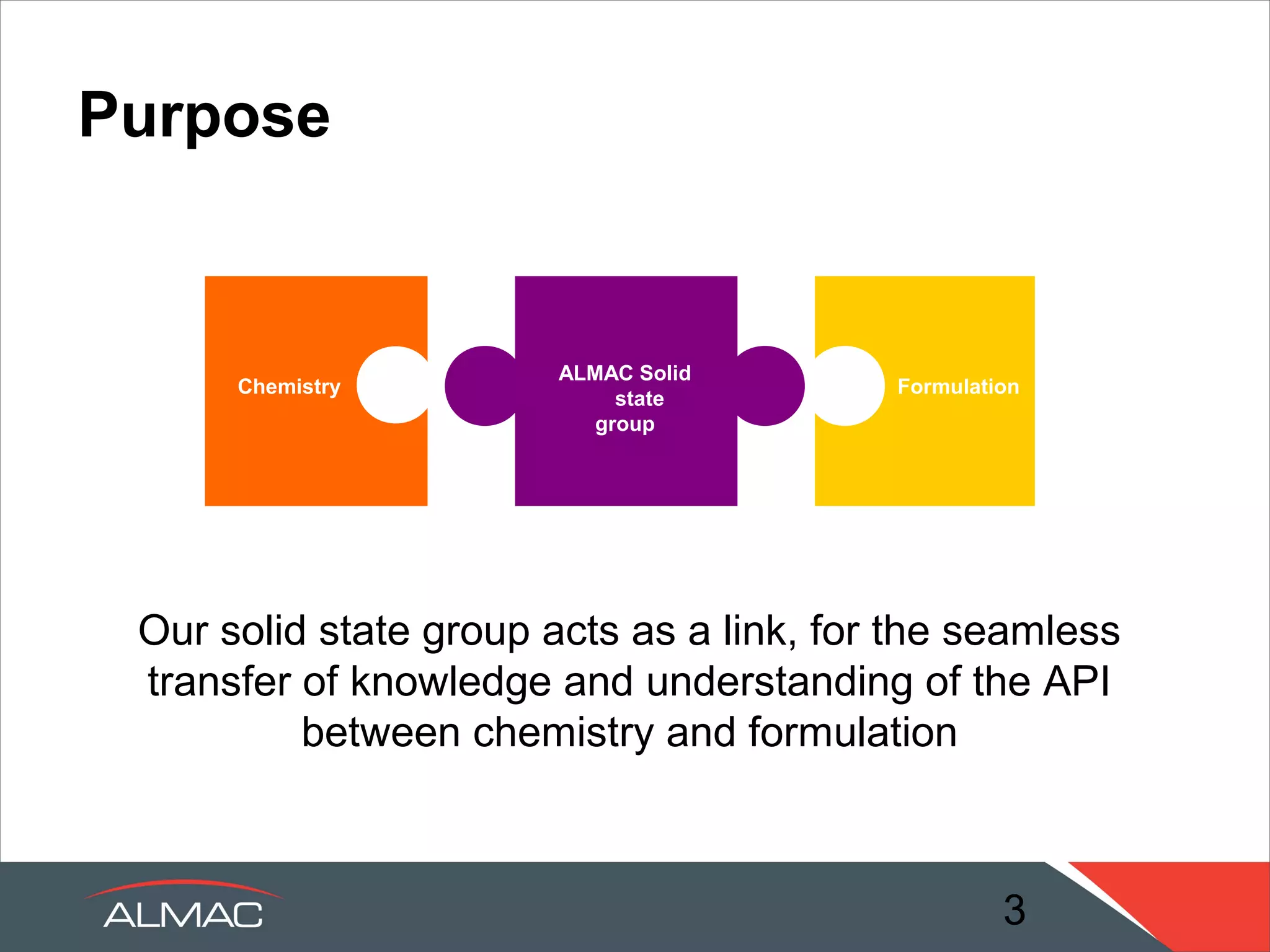
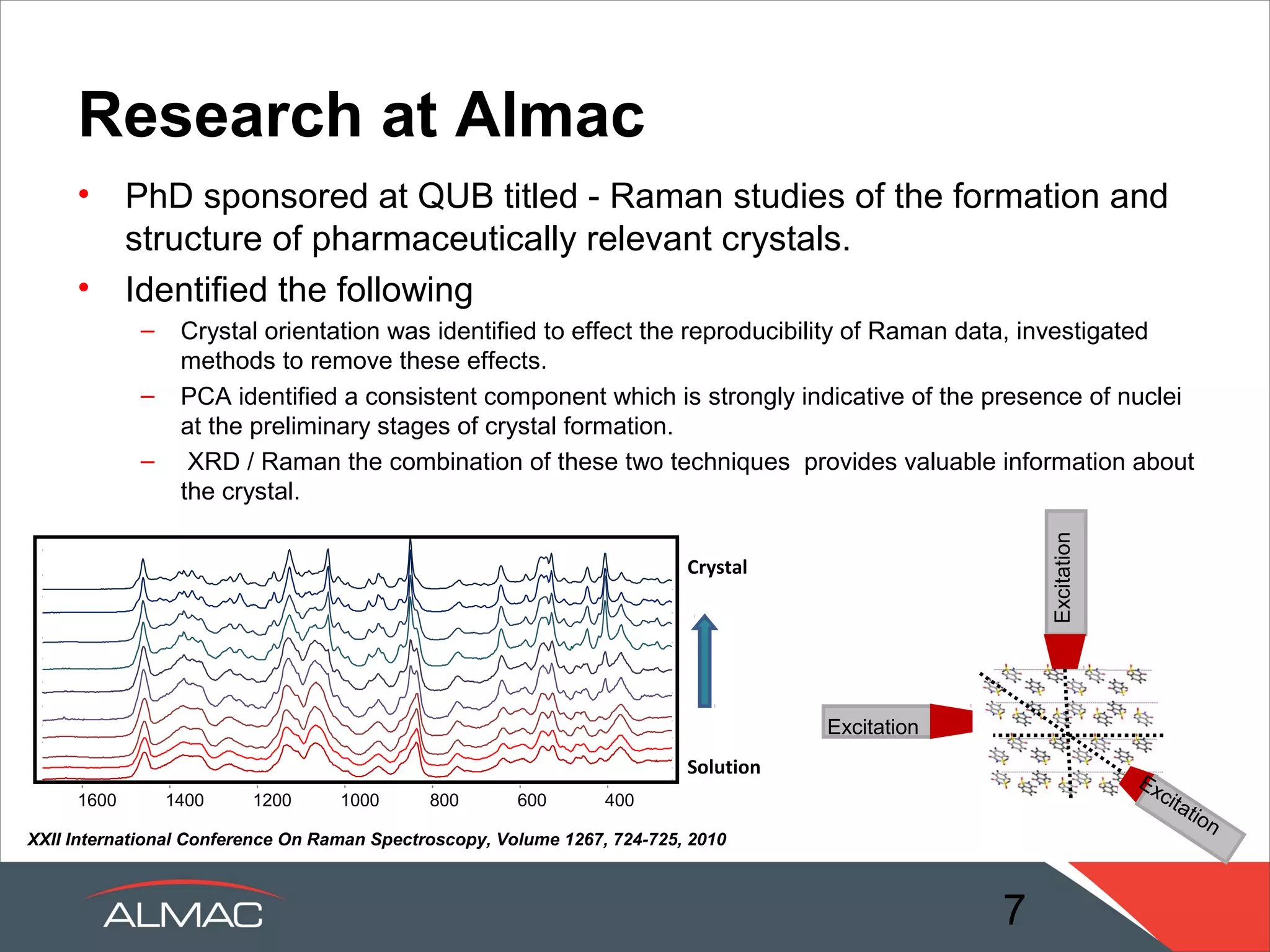
The document describes the solid state services provided by ALMAC including polymorph screens, salt/cocrystal screens, amorphous dispersion screens, and crystallization development. It discusses how the solid state group acts as a link between chemistry and formulation to transfer knowledge about active pharmaceutical ingredients. A range of expert services and in-house technologies are available to support drug development including rapid formulation screening, analytical support, and pre-formulation studies.