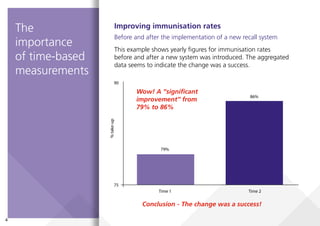
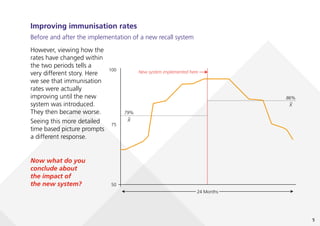
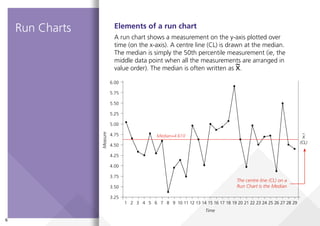
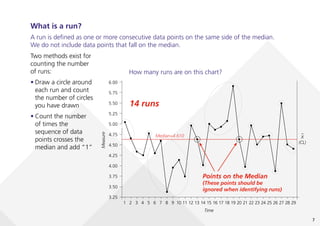
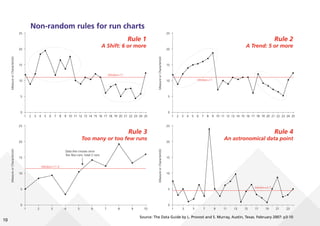
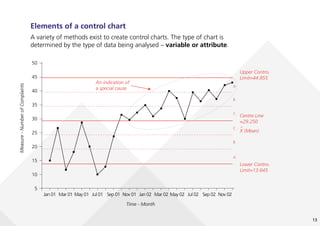
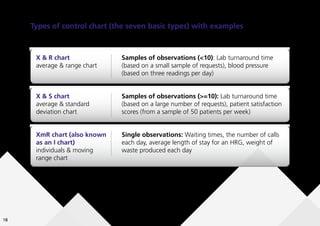
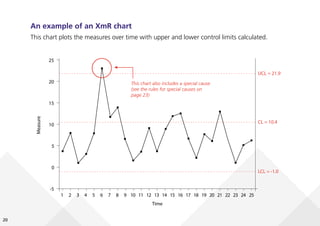
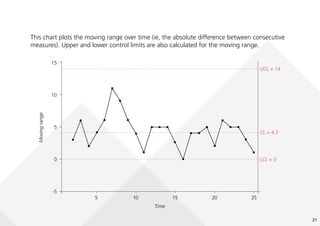
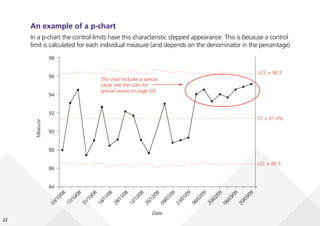
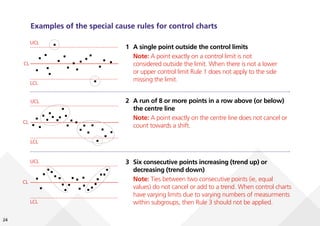
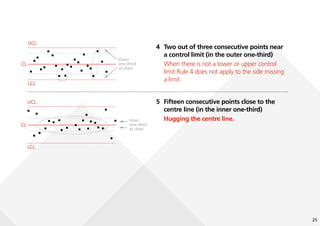
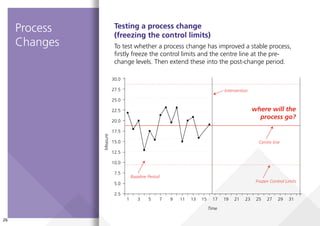
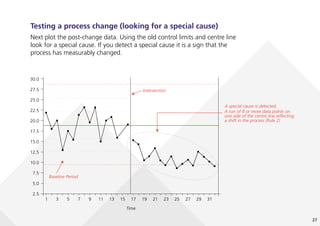
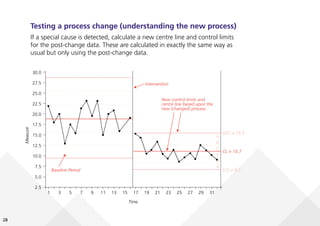
This document provides guidance on creating and interpreting run charts and control charts to analyze time-series data for quality improvement purposes. It explains that run charts show measurements over time with a center line at the median, and control charts show measurements over time with a center line at the mean and upper and lower control limits. Various rules are provided to identify special cause variation indicating a process change may be needed. The document also distinguishes between attribute and variable data, and provides examples of each along with a decision tree to determine which type of control chart is most appropriate.