Advances in Tardive Dyskinesia: A Patient's Journey From Diagnosis to Management and the Role of New Therapies for Improved Outcomes
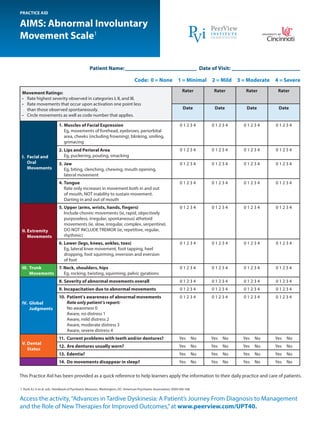
- 1. Access the activity,“Advances in Tardive Dyskinesia: A Patient’s Journey From Diagnosis to Management and the Role of New Therapies for Improved Outcomes,”at www.peerview.com/UPT40. AIMS: Abnormal Involuntary Movement Scale1 PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. 1. Rush AJ Jr et al, eds. Handbook of Psychiatric Measures. Washington, DC: American Psychiatric Association; 2000:166-168. Movement Ratings: • Rate highest severity observed in categories I, II, and III. • Rate movements that occur upon activation one point less than those observed spontaneously. • Circle movements as well as code number that applies. Rater Rater Rater Rater Date Date Date Date I. Facial and Oral Movements 1. Muscles of Facial Expression Eg, movements of forehead, eyebrows, periorbital area, cheeks (including frowning), blinking, smiling, grimacing 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 2. Lips and Perioral Area Eg, puckering, pouting, smacking 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 3. Jaw Eg, biting, clenching, chewing, mouth opening, lateral movement 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 4. Tongue Rate only increases in movement both in and out of mouth, NOT inability to sustain movement. Darting in and out of mouth 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 II. Extremity Movements 5. Upper (arms, wrists, hands, fingers) Include choreic movements (ie, rapid, objectively purposeless, irregular, spontaneous) athetoid movements (ie, slow, irregular, complex, serpentine). DO NOT INCLUDE TREMOR (ie, repetitive, regular, rhythmic) 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 6. Lower (legs, knees, ankles, toes) Eg, lateral knee movement, foot tapping, heel dropping, foot squirming, inversion and eversion of foot 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 III. Trunk Movements 7. Neck, shoulders, hips Eg, rocking, twisting, squirming, pelvic gyrations 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 IV. Global Judgments 8. Severity of abnormal movements overall 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 9. Incapacitation due to abnormal movements 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 10. Patient’s awareness of abnormal movements Rate only patient’s report: No awareness 0 Aware, no distress 1 Aware, mild distress 2 Aware, moderate distress 3 Aware, severe distress 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 0 1 2 3 4 V. Dental Status 11. Current problems with teeth and/or dentures? Yes No Yes No Yes No Yes No 12. Are dentures usually worn? Yes No Yes No Yes No Yes No 13. Edentia? Yes No Yes No Yes No Yes No 14. Do movements disappear in sleep? Yes No Yes No Yes No Yes No Patient Name: Date of Visit: Code: 0 = None 1 = Minimal 2 = Mild 3 = Moderate 4 = Severe
- 2. Access the activity,“Advances in Tardive Dyskinesia: A Patient’s Journey From Diagnosis to Management and the Role of New Therapies for Improved Outcomes,”at www.peerview.com/UPT40. Practical Considerations for Use of Approved VMAT2 Inhibitors for Tardive Dyskinesia Management This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. VMAT2: vesicular monoamine transporter 2. 1. Austedo (deutetrabenazine) Prescribing Information. https://www.austedo.com/hd/pi. Accessed February 16, 2018. 2. Ingrezza (valbenazine) Prescribing Information. https://ingrezzahcp.com/PI. Accessed February 16, 2018. PRACTICE AID Therapy Dosing Safety • Initial dose: 12 mg/d • Titrate at weekly intervals by 6 mg/d based on reduction of tardive dyskinesia and on tolerability • Administer total daily dosage of 12 mg or above in two divided doses • Maximum dose: 48 mg/d • Maximum recommended dosage in poor CYP2D6 metabolizers: 36 mg/d • Common adverse events (in 4% of deutetrabenazine-treated patients and greater than placebo): nasopharyngitis and insomnia • For patients at risk for QT prolongation, assess the QT interval before and after increasing the total dosage above 24 mg/d • If switching patients from tetrabenazine, discontinue and initiate deutetrabenazine the following daya • Initial dose: 40 mg/d • After 1 week, increase to recommended dose of 80 mg/d • Consider dose reduction based on tolerability in known CYP2D6 poor metabolizers • Recommended dose for patients with moderate or severe hepatic impairment: 40 mg/d • Most common adverse reaction (≥5% and twice the rate of placebo): somnolence • Avoid use in patients with congenital long QT syndrome or with arrhythmias associated with prolonged QT interval a See full prescribing information for recommended conversion table. O O O O H H2 N N Valbenazine2 Deutetrabenazine1 CD3 O O O H N CD3
