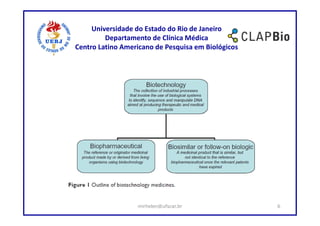
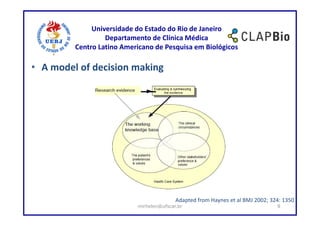
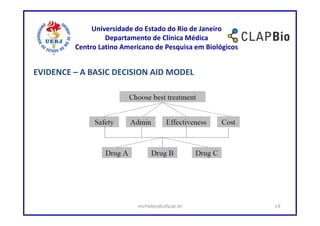
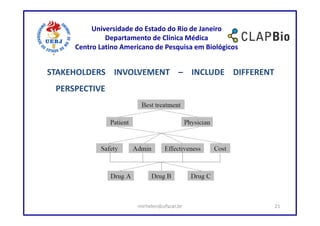
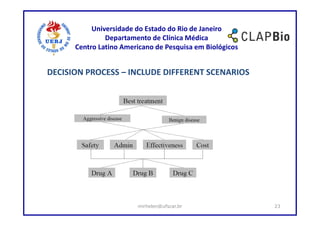
This document outlines a research project on using shared decision making (SDM) to guide discussions about biosimilars and biological agents in Latin America. The project will involve two stages: 1) an international survey to understand stakeholders' perspectives on SDM and its applicability to biosimilars, and 2) development and testing of a decision aid tool to facilitate SDM discussions on biosimilars while accounting for cultural factors. The goal is to explore barriers to SDM implementation and develop a validated decision aid to guide discussions and priority setting around biosimilar treatment options in Latin America.