Pollutants Transport, Accumulation, Transformation and Biomagnification Processes
- 1. Transportation ,accumulation and transformation of pollutants, Biomagnification and Transformation Processes
- 2. Transport Two kinds of transport Mechanisms are distinguished Intermedia transport • intermedia transport, which is transport from one environmental medium to another. • Intermedia transport (air-water, water- sediment, etc.) also takes place by advective and dispersive mechanisms. Advective intermedia transport takes place if a chemical is transported from one environmental compartment to another by a physical carrier. • Intermedia dispersion, like intramedia dispersion, is diffusive in nature and follows concentration gradients. Intramedia transport • intramedia transport, which is transport away from a source in one environmental medium • Intramedia transport takes place through the mechanisms of advection and dispersion. Advection causes a chemical to travel from one place to another as a result of the flow of the medium in which it occurs; locally emitted packages or “puffs” of a chemical are carried as far as the wind or water current can take it during the residence time in that medium. Dispersion mechanisms (molecular diffusion, eddy diffusion) make the chemical move down concentration gradients until the concentration gradients disappear.
- 3. Wet and Dry Deposition Chemicals are transported from the atmosphere to water and soil by atmospheric deposition Wet and dry atmospheric deposition mechanism. • Wet deposition is the sum of rain-out (in-cloud processes) and wash-out (below-cloud processes) • The efficiency of the wet deposition process varies greatly. It depends on meteorological factors such as the duration, intensity and type of precipitation (snow, rain, hail), as well as on the size and the number of droplets. Other specific parameters, like solubility in rain and snow. • In the case of dry deposition, the main resistances occur at the air-surface interface: transport of the chemical from the air to the interface, diffusion across the interface and transport from the interface to the solid surface.
- 4. Bio accumulation An important process through which chemicals can affect living organisms is bioaccumulation. Bioaccumulation means an increase in the concentration of a chemical in a biological organism over time, compared to the chemical's concentration in the environment. Compounds accumulate in living things any time they are taken up and stored faster than they are broken down (metabolized) or excreted. UPTAKE Uptake is a complex process which is still not fully understood. chemicals tend to move, or diffuse, passively from a place of high concentration to one of low concentration. The force or pressure for diffusion is called the chemical potential, and it works to move a chemical from outside to inside an organism. STORAGE Some chemicals are attracted to certain sites, and by binding to proteins or dissolving in fats, they are temporarily stored. If uptake slows or is not continued, or if the chemical is not very tightly bound in the cell, the body can eventually eliminate the chemical.
- 5. ELIMINATION Chemicals that dissolve readily in fat but not in water tend to be more slowly eliminated by the body and thus have a greater potential to accumulate. Many metabolic reactions change a chemical into more water soluble forms called metabolites, that are readily excreted. Bioconcentration Bioconcentration is the specific bioaccumulation process by which the concentration of a chemical in an organism becomes higher than its concentration in the air or water around the organism.
- 6. Volatilizationo or Gas Sorbtion Volatilization of chemicals from soil is the transfer of the chemical as a gas through the soil-air interface under environmental conditions; volatilization from plants is the corresponding process. • volatilization of chemicals may contribute significantly to air pollution and, thus, to the long-range transport of chemicals by air. • The resistance to intermedia transfer is considered to be concentrated in two thin films on either side of the interface • Transport through this interfacial double layer has to take place by molecular diffusion and is, therefore, slow. • The direction of transport depends on the concentrations in air and water. • If the actual concentration of the chemical in water is higher than the equilibrium concentration in water, the chemical will volatilize from the water phase into the gas phase. Ammonia Volatilization Loss from a Paddy Soil
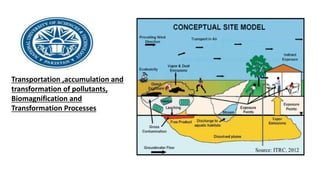
- 7. Conceptual model of vapors migration to outdoor air The case investigated is a contaminated area located in a industial area of Trento North district once occupied by Italian Carbochimica plant .Pollution in that area is due to contamination of soil and groundwater.
- 8. Biomagnification • When the concentration of a chemical becomes higher in the organism than in its food (and the major uptake route is food) this is called biomagnification. • Chemicals reaching relatively high concentrations in food • While low concentrations in other surrounding media, such as water for aquatic organisms, air for terrestrial organisms and soil and sediment for soil organisms.
- 9. Uptake from Food, Water & Sediment • Uptake from food occurs in the gastrointestinal tract (GIT). • After release of the contaminants in the GIT lumen, the chemicals may cross the lipid membranes • Xenobiotic compounds can be taken up by aquatic organisms from water, food or sediment • Methods for measuring Biomagnification: Dietary tests in fishes • The simplest measure is the Biomagnification Factor (BMF), which is described as the ratio of the chemical concentrations in the organism (CB) and the diet of the organism (CD), i.e., BMF = CB/CD, where the chemical are usually expressed in units of mass of chemical per kg of the organism (in wet weight or in a lipid basis) and mass chemical per kg of food (in wet weight or in a lipid basis) (Gobas and Morrison 2000)
- 10. Food as a major source of contaminants
- 11. Abiotic Transformation Processes • Degradation or transformation of a compound refers to the disappearance of the parent compound from the environment by a change in its chemical structure • Chemicals released into the environment gets transformed by abiotic and biotic processes • If the above mentioned change is brought by microorganism it is referred to as, biodegradation or bio transformation. • Transformation of chemicals in environment can occur by abiotic processes, 1- Hydrolysis: alteration of the chemical structure by direct reaction with water. 2- Oxidation: a transformation process in which electrons are transferred from the chemical to a species accepting the electrons; the oxidant. 3- Reduction: the reverse of oxidation; electron transfer takes place from a reductant to the chemical to be reduced. 4- Photochemical degradation: transformation due to interaction with sunlight
- 12. Abiotic Transformation Transport and transformation phenomena and processes in a water system. The transformation processes, including dissociation and degradation to form metabolites and degradation products (B, C, and D), simultaneously consist of both abiotic (e.g., hydrolysis and photolysis) and biotic (i.e., biodegradation). In addition to chemical reactions, Compound A, the parent, and its reaction products also undergo physical changes, e.g., scouring, molecular diffusion (usually only important in quiescent systems, e.g., sediment layers), and advective processes.
- 13. Biotic Transformation Processes • Organism eat chemical, they excrete or alter it • Biodegradation and biotransformation. • Biotransformation influences the fate of a compound by decreasing its amount due to conversion into a new xenobiotic compound, the metabolite • Biotransformation requires enzymes (Biological catalyst) • Formation of Hydrophilic compounds- Chemical structures altered then.. • Bio activation/ detoxification • There are two types of biotransformation reactions: Phase-I non-synthetic reactions and phase II synthetic reactions. • Phase-I reactions include hydrolysis, reduction, and oxidation. During Phase-I reactions the molecule is changed by the introduction of polar groups, such as hydroxy (-OH), carboxyl (-COOH) or amino (-NH2) group • Phase-II reactions are usually conjugation reactions. The products of Phase-I reactions are often reactive compounds which can be easily conjugated in Phase-II reactions. The conjugated products will then be excreted.
- 14. Biotransformation a) catabolism - degradation vs. mineralization (CO2, H2O) - energy for growth and motility (but not always!) b) Decomposition bacteria and fungi . Pseudomonas, Bacillus, Arthrobacter, Actinomycetes c) Methanogens - strict anaerobes - convert fermentation products (ethanol, acetate,...) to methane
- 15. Attenuation and degradation mechanisms in macrophytic plants.
Editor's Notes
- Biomagnification refers to the condition where the chemical concentration in an organism exceeds the concentration of its food when the major exposure route occurs from the organism’s diet. The industrial wastes polluting the ocean enter into the fish-food chains, and fishes are a staple diet in many parts of the world. West Bengal has a big fish cuisine Heavy metals like mercury, cadmium and chromium also get biomagnified through water/food resources and can affect the nervous systems of humans. This was found to be the case in a study in West Bengal. Biomagnification and Bioaccumulation: Biomagnification is related to Bioaccumulation, which is the increase in toxic substances in the body of organisms. These two terms re different, Accumulation pertains to harmful substances in a living organisms and its effects. Magnification deals with the substances being passed along the foodchain.
- Kf is the uptake rate constant from food Ke is an overall elimination rate constant Dietary Tests in fishes: exposes fish to chemicals via the diet, and measures both the uptake rate during exposure as well as depuration when fish are transferred to clean food. This approach allows for determination of the elimination half-life, dietary assimilation efficiency and biomagnification factor. It also allows for the determination of the bio concentration factor when it is assumed that the uptake rate can be derived from allometric relationships.
- Food as a major source of contaminants for mammals and birds in a simplified food web. 1=Application of spray, 2=Drinking from leaves/crop, 3=Ingestion of granules/treated seeds, 4=Bioconcentration soil-worm, 5=Drinking from surface water, 6=Bioconcentration water-fish, 7=Consumption.
- Biodegradation/ Biotransformation: In this process fractions of the chemical structure are incorporated into cellular material or used as an energy source by the organism. Often micro-organisms are capable of converting the chemical to simple molecules and ions, such as carbon dioxide, methane, water and chloride. This process is referred to as mineralization. Hydrolysis: The chemical reaction of organic compounds with water is called hydrolysis. In a typical hydrolysis reaction hydroxide replaces another chemical group. Oxidation: Oxidation is the chemical process in which an electron deficient particle (the oxidant) accepts electrons from the compound to be oxidized. Examples of oxidants that occur under environmental conditions in sufficiently high concentrations and also react quite quickly with organic compounds are: • Alkoxy radicals (RO⋅) • Peroxy radicals (RO2⋅) • Hydroxyl radicals (HO⋅) • Singlet oxygen (1O2) • Ozone (O3) Reduction: Reduction is the chemical process by which electrons are transferred from an electron donor (reductant) to the compound to be reduced.
- https://www.sciencedirect.com/topics/earth-and-planetary-sciences/biotic-transformation most environmental conditions ensure that both abiotic and biotic transformations occur together. In addition, physical processes, e.g., scouring, diffusion, and advection, accompany these chemical transformations [29]. At times, an abiotic reaction is the initiating event for subsequent biotic transformations. For example, adding heat and increasing water temperature can initiate microbial growth. As the aquatic microbial populations, the dissolved oxygen (DO) concentrations decline. The attendant oxygen concentration decrease, however, is a net decline between photosynthetic and nonphotosynthetic microbes. The growth and metabolism of the bacteria result in decreasing the DO levels, but the growth of the algae both consume DO for metabolism and produce DO by photosynthesis.
- Biodegradation: When a chemical is transformed by microorganisms it is called biodegradation Basic strategy degrade large contaminants outside the cells (by exoenzymes) . take the small molecules into the cell (passive diffusion, active transport) transform (mineralize) them to obtain energy Biochemical transformation reactions substitution, oxidation, reduction, dehydrohalogenation... Biotransformation: When a chemical is transformed by other organisms, it is called biotransformation Biotransformation can therefore be defined as an enzyme catalyzed conversion of one xenobiotic compound into another. Hydrophilic compounds : In general, biotransformation leads to the conversion of the parent compound into a more water soluble form. As a result these more hydrophilic compounds may be more easily excreted from the body than the parent compound Transformation into a more toxic compound is called bioactivation. Reduction of toxicity due to transformation to a less harmful product is called detoxification. Phase 1 and 2 Which type of reaction will occur depends on the chemical structure of the compound. Conjugation reactions Conjugation reactions, or phase II reactions, synthesize more water-soluble compounds by combining a substance with an endogenous molecule to enhance excretion of that substance. Glucuronide, sulfate, and glycine are the common endogenous molecules to which drugs are bound.
- Mineralization: Micro-organisms are capable of converting the chemicals to simple molecules and ions, such as carbon dioxide, methane, water and chloride. This process is referred to as mineralization. Transformation and mineralization processes can alter the physicochemical and toxicological properties and reduce exposure concentrations of chemicals released in the environment Degradation is the conversion of a chemical compound by splitting of one or more groups of atoms. Mineralisation refers to a process where an inorganic substance precipitates in an organic matrix or it is a conversion of organically bound form to an inorganic form.
- Attenuation and degradation mechanisms in macrophytic plants. A compound (xenobiotic) is stabilized or degraded in the rhizosphere, adsorbed or accumulated in to the roots and transported to the aerial parts, volatilized or degraded inside the plant tissue. Degradation generally involves enzymatic mediated metabolism (Phase I); conjugation (Phase II); and active sequestration (Phase III). Note: Active transporters are marked in the green box (GST = glutathione S-transferases; GT = glucosyltransferases; Mt = malonyltransferases; OA = organic acids). Conjugation: Conjugation reactions chemically link products of hydrolysis, oxidation, and reduction (i.e. phase I metabolites) to glutathione, sugars, or amino acids, so that the subsequent metabolites (i.e. phase II metabolites) have increased aqueous solubility and, hopefully, less toxicity than the parent compound.
