Crisaborole slide
- 2. INTRODUCTION Crisaborole is a novel oxaborole approved for topical treatment of mild to moderate atopic dermatitis in patients 2 years old and older.
- 3. • Efficacious in- –Improving disease severity –Reducing the risk of infection –Reducing the signs and symptoms –Reducing the local inflammation in the skin –Preventing further exacerbation of the disease with a good safety profile.
- 4. • Currently under development as topical treatment of psoriasis. • Shown efficacy in treating inflammatory linear verrucous epidermal nevus
- 5. Why crisaborole? • Although chronic inflammatory skin diseases such as atopic dermatitis and psoriasis can be effectively treated with topical treatments, a number of safety concerns warrant special consideration.
- 6. • Topical corticosteroids (TCSs) and topical calcineurin inhibitors (TCIs) are effective treatment options for AD- – Use of TCS on thin-skinned areas such as the face, intertriginous areas, and genitals are limited by potential adverse effects. – TCIs are associated with application-site reactions and contain a boxed warning citing cases of malignancy
- 7. Therapy Adverse Event Malignancy Risk Topical Corticosteroi ds Epidermal atrophy; telangiectasia; tachyphylaxis; systemic absorption leading to HPA axis suppression Evidence-based: No evidence of immunosuppressive malignancy Topical Calcineurin Inhibitors Irritation at application site Theoretical: topical dosing can induce cancer in mice at >40x maximum human recommended dose. Evidenced-based: No evidence of immunosuppressive malignancy Skin
- 8. Therapy Adverse Event Malignancy Risk Phototherapy UVA, UVB, PUVA(topical) Erythema,pruritus; nausea, headache, chronic actinic skin damage, dyskeratotic and precancerous skin conditions Increased risk of squamous cell carcinoma, basal cell carcinoma and malignant melanoma
- 9. • In psoriasis, cutaneous irritation may develop following treatment with topical retinoids and vitamin D analogs • Therefore, effective and well-tolerated topical treatment alternatives that can be safely applied to the face and other thin and sensitive skin areas are needed for inflammatory skin diseases such as atopic dermatitis and psoriasis.
- 11. • This compound 3 is a benzoxaborole which was called AN2728 and PF-06930164 during preclinical and clinical development. • And now it is called crisaborole.
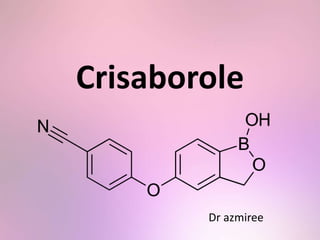
- 12. Structure • A member of the class of benzoxaboroles that is 5-hydroxy-1,3-dihydro-2,1-benzoxaborole in which the phenolic hydrogen has been replaced by a 4-cyanophenyl group.
- 13. • This lead to formation of Crisaborole, 5-(4- cyanophenoy)-1,3-dihydro-1- hydroxy-[2,1]- benzoxaborole
- 14. • The empirical formula is C14H10BNO3 and the molecular weight is 251.1 g/mol. • Crisaborole drug substance is freely soluble in common organic solvents such as isopropyl alcohol and propylene glycol, and insoluble in water.
- 15. Mechanism of action • Boron-based small molecules are potent inhibitors of phosphodiesterase 4 (PDE4) exerting broad-spectrum anti-inflammatory activity. • Intracellular PDE4 is an enzyme that converts the intracellular second messenger 3'5'-cyclic adenosine monophosphate (cAMP) into the active metabolite 5’-AMP
- 16. • PDE4 predominantly found in keratinocyte and immune cells, including— – B cells, – T cells, – Neutrophils, – Monocytes, – Macrophages, and – Eosinophils.
- 18. • The inclusion of boron mimics the phosphate in cAMP, allowing the drug to effectively target the PDE4 and inhibit cAMP breakdown
- 19. • The resulting increase in cAMP activates protein kinase A. • This protein kinase negatively modulates signaling pathways – Nuclear factor-kB and –T-cell signalling pathways
- 20. • This promote release of Th1 and Th2 cytokines involved in initiating and sustaining –Inflammation, –Pruritus and –Epidermal hyperplasia.
- 22. • Benzoxaborole PDE4 inhibitors have been shown in cell culture to suppress release of tumor necrosis factor-alpha (TNFα), interleukin23 (IL-23), IL-22, IL-17, IL-14, IL-13, IL-4, and interferon-ɤ.
- 23. Pharmacokinetics: Absorption • Topical application of this drug is useful as it potentiates the localization of this drug in the skin • The use of boron chemistry enabled synthesis of a low-molecular-weight compound (251 daltons), thereby facilitating effective penetration of crisaborole through human skin
- 24. • Topical administration of PDE4 inhibitors allows rapid absorption into the skin, but limits systemic exposure. • Systemic concentrations of crisaborole were reached by 8 days of twice-daily topical administration.
- 25. • The small fraction of a crisaborole 2% ointment dose that is systemically absorbed is low , thus posses less risk for developing systemic side effects.
- 26. Pharmacokinetics: Distribution • Protein binding –Crisaborole is highly bound (97%) to plasma proteins.
- 27. Pharmacokinetics: Metabolism • The small fraction absorbed systemically undergoes extensive hepatic metabolism via hydrolysis and subsequent oxidation to two inactive metabolites. • The major metabolite 5-(4-cyanophenoxy)-2- hydroxyl benzylalcohol (metabolite 1), is formed via hydrolysis.
- 28. • This metabolite is further metabolized into downstream metabolites, among which 5-(4- cyanophenoxy)-2-hydroxyl benzoic acid (metabolite 2), formed via oxidation, is also a major metabolite. • Crisaborole does not produce clinically significant induction or inhibition of cytochrome P450 enzymes
- 29. Pharmacokinetics: Elimination • The major route of elimination of the metabolites: Renal excretion
- 30. Indication • Mild to moderate atopic dermatitis in patients 2 years of age and older.
- 31. Contraindication • Known hypersensitivity to crisaborole or any component of the formulation
- 32. Availability • Crisaborole 2% ointment (EucrisaTM) is available in 60 gram and 100 gram tubes.
- 33. Content of formulation • Active ingredient: crisaborole(each gram contains 20 mg crisaborole ) • Inactive ingredients: white petrolatum, propylene glycol, mono- and di-glycerides, paraffin, butylated hydroxytoluene, and edetate calcium disodium.
- 34. Dosing • Applied in a thin layer twice daily to affected areas of the skin only • Not for ophthalmic, oral, or intravaginal use.
- 35. Side effects • Common : – Application site pain(1% to 10%) present as burning or stinging • Less frequent: – Contact urticaria ( <1%) – Hypersensitivity reaction: Hypersensitivity should be suspected in the event of severe pruritus, swelling and erythema at the application site or at a distant site.
- 36. • No evidence of mutagenic or clastrogenic potential as well as altered effects on fertility.
- 37. • No drug-related neoplastic findings were noted at topical doses up to 7% crisaborole ointment (2 times the Maximum recommended human dose)
- 38. Drug Interaction Studies • In vitro studies using human liver microsomes indicated that under the conditions of clinical use, crisaborole and metabolite 1 are not expected to inhibit cytochrome P450 (CYP) 1A2, 2B6, 2C8, 2C9, 2C19, 2D6, and 3A4.
- 39. • In vitro human liver microsomes studies for metabolite 2 showed that it did not inhibit activities of CYP2C19, 2D6, and 3A4; was a weak inhibitor of CYP1A2 and 2B6; and a moderate inhibitor of CYP2C8 and 2C9
- 40. • The most sensitive enzyme, CYP2C9, was further investigated in a clinical trial using warfarin as a CYP2C9 substrate. The results of this study showed no drug interaction potential.
- 41. Crisaborole in Pregnancy • There is no available data in pregnant women to inform the drug-associated risk for major birth defects and miscarriage. • No adverse developmental effects were observed in pregnant rabbits with oral administration of crisaborole.
- 42. Crisaborole in Breastfeeding • There is no information available on –the presence of crisaborole in human milk, – the effects of the drug on the breastfed infant, or –the effects of the drug on milk production after topical application of crisaborole to women who are breastfeeding.
- 43. • Absorbed systemically • Therefore, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for crisaborole
- 44. Pediatric Use • The safety and effectiveness of crisaborole have been established in pediatric patients age 2 years and older for topical treatment of mild to moderate atopic dermatitis.
- 45. • The safety and effectiveness of EUCRISA in pediatric patients below the age of 2 years have not been established yet.
- 46. CONCLUSION • The novel nonsteroidal anti-inflammatory topical agent, crisaborole, can herald an new era not only in treatment of atopic dermatitis but also in treatment of psoriasis, vitiligo, Inflammatory linear verrucous epidermal nevus.
- 47. THANK YOU