Yale Tulane ESF-8 Special Report COVID-19 04-07-2020
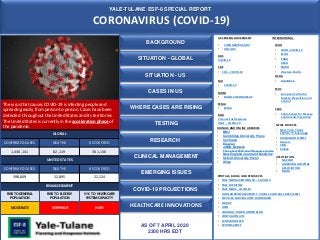
- 1. YALE-TULANE ESF-8 SPECIAL REPORT CORONAVIRUS (COVID-19) AS OF 7 APRIL 2020 2300 HRS EDT US FEDERAL GOVERMENT • CORONAVIRUS.GOV • USA.GOV HHS COVID-19 CDC • CDC – COVID-19 NIH • COVID-19 NIOSH • NIOSH CORONAVISUS FEMA • FEMA DOD Coronavirus Response USAF _ COVID-19 NEWS SOURCES • New York Times COVID-19 Coverage • WASHINGTON POST • Reuters • CNN • Xinhua ASSOCIATION • NACCHO • AMERICAN HOSPITAL ASSOCIATION • NRHA SITUATION - GLOBAL PORTALS, BLOGS, AND RESOURCES • YALE NEWHAVEN HEALTH – COVID-19 • YALE MEDICINE • YALE NEWS _COVID 19 • JOHN HOPKINS UNIVERSITY COVID-19 GLOBAL CASES (CSSE) • COVID-19 SURVEILLANCE DASHBOARD • CIDRAP • H5N1 • VIROLOGY DOWN UNDER BLOG • CONTAGION LIVE • WORLDOMETER • 1POINT3ACRES BACKGROUND WHO • WHO –COVID-19 • ECHO • PAHO AFRO • EMRO • Western Pacific OCHA • ReliefWeb ECDC • European Centre for Disease Prevention and Control CCDC • China Center for Disease Control and Prevention INTERNATIONAL JOUNALS AND ONLINE LIBRARIES • BMJ • Cambridge University Press • Cochrane • Elsevier • JAMA Network • The Lancet 2019-nCoV Resource Centre • New England Journal of Medicine • Oxford University Press • Wiley RESEARCH SITUATION - US The virus that causes COVID-19 is infecting people and spreading easily from person-to-person. Cases have been detected throughout the United States and its territories . The United States is currently in the acceleration phase of the pandemic. RISK ASSESSMENT RISK TO GENERAL POPULATION RISK TO ELDERLY POPULATION RISK TO HEATHCARE SYSTEM CAPACITY MODERATE VERY HIGH HIGH GLOBAL CONFIRMED CASES DEATHS RECOVERED 1,430,141 82,119 301,130 UNITED STATES CONFIRMED CASES DEATHS RECOVERED 398,809 12,895 22,224 HEALTHCARE INNOVATIONS COVID-19 PROJECTIONS CASES IN US WHERE CASES ARE RISING TESTING CLINICAL MANAGEMENT EMERGING ISSUES
- 2. BACKGROUND WHERE: WORLDWIDE WHEN: DECEMBER 2019 - CURRENT SITUATION PANDEMIC OUTBREAK – COVID-19 BACKGROUND: At the end of December 2019, Chinese public health authorities reported several cases of acute respiratory syndrome in Wuhan City, Hubei province, China. Chinese scientists soon identified a novel coronavirus as the main causative agent. The disease is now referred to as coronavirus disease 2019 (COVID-19), and the causative virus is called severe acute respiratory syndrome coronavirus 2 (SARS- CoV-2). It is a new strain of coronavirus that has not been previously identified in humans. The initial outbreak in Wuhan spread rapidly, affecting other parts of China. The International Health Regulations (IHR) (2005) Emergency Committee on the outbreak of COVID-19 was first convened on 22‒23 January 2020, and subsequently reconvened on 30 January 2020. PUBLIC HEALTH EMERGENCY OF INTERNATIONAL CONCERN (PHEIC) The WHO Director General declared the COVID-19 outbreak to be a public health emergency of international concern (PHEIC) on 30 January 2020. The Emergency Committee provided recommendations to WHO, to China, to all countries and to the global community, on measures to control the outbreak. PANDEMIC On 11 March 2020. WHO declared COVID-19 a pandemic because of the “alarming levels of spread and severity, and by the alarming levels of inaction”. At that time, there were large outbreaks of the virus in Italy, South Korea, and the United States. In the US, the slow rollout of testing and limited testing capacity has crippled response to the disease. The declaration came after a 13-fold rise in the number of cases outside China in the two weeks prior to the declaration. SOURCE: NATURE WHAT IS COVID-19 Coronavirus disease (COVID-19) is a new strain that was discovered in 2019 and has not been previously identified in humans. Coronaviruses are zoonotic, meaning they are transmitted between animals and people. HOW IS IT SPREAD? People can catch COVID-19 from others who have the virus. The disease spreads from person to person through small droplets from the nose or mouth when a person with COVID-19 coughs or exhales. Droplets can land surfaces around the infected person and other can catch COVID-19 by touching these objects or surfaces, then touching their eyes, nose or mouth. People can also catch COVID- 19 if they breathe in droplets from the infected person. This is why it is important to stay more than 3 feet away from a person who is sick. (WHO) Time from exposure to onset of symptoms is generally between 2 and 14 days. (CDC) WHAT ARE THE SYMPTONS? Common symptoms include fever, cough, and shortness of breath (CDC). Complications may include pneumonia and acute respiratory distress syndrome. PREVENTIVE MEASURES Recommended preventive measures: hand washing, covering the mouth when coughing, maintaining distance from other people, and monitoring and self- isolation for people who suspect they are infected. NOTE: Anecdotal evidence is rapidly accumulating from sites around the world that anosmia (lack of smell) and dysgeusia (lack of taste) are significant symptoms associated with the COVID-19 pandemic. Anosmia, in particular, has been seen in patients ultimately testing positive for the coronavirus with no other symptoms.
- 3. RISK ASSESSMENT RISK TO GENERAL POPULATION RISK TO ELDERLY POPULATION RISK TO HEATHCARE SYSTEM CAPACITY MODERATE VERY HIGH VERY HIGH SITUATION - GLOBAL On 31 March 2020, WHO published a Medical Product Alert (Number 3/2020) regarding falsified medical products, including in vitro diagnostics, that claim to prevent, detect, treat or cure COVID-19. WHO has received reports regarding falsified in vitro diagnostics and laboratory reagents for the detection of SARS- CoV-2. Due diligence is required from all actors in the procurement, use and administration of medical products, in particular those affected by the current crisis of, or related to, COVID-19. MEDICAL PRODUCT ALERT AFFECTED COUNTRIES/AREAS /TERRITORIES WITH CASES : 211 CONFIRMED CASES: 1,430,141 DEATHS: 82,119 CURRENT EPI CENTER OF THE VIRUS: EUROPE (686,338) COUNTRY WITH MOST CASES: UNITED STATES Pandemic is continuing to spread globally and is accelerating. Cases are now being reported in almost every county/territory. There is evidence that countries are having success flattening the epi curve where stay-at-home policies have been in place for two weeks or more. NOTE: Flattening the curve does not necessarily mean seeing a decrease in total cases right away; it first produces a decline in the number of new cases, which result in fewer hospitalizations and death in the weeks that follow. Testing remains problematic. There are not sufficient test kits to meet demand. This makes interpretation of new case statistics problematic. though death/mortality data may be most usable information available. Almost every country in the world facing a shortages in personnel protective gear Emerging evidence suggest that asymptomatic cases could be drivers of transmission https://who.sprinklr.com/ OFF-LABEL USE OF MEDICINES FOR COVID-19 No pharmaceutical products have yet been shown to be safe and effective for the treatment of COVID-19. However, a number of medicines have been suggested as potential investigational therapies, many of which are now being or will soon be studied in clinical trials, including the SOLIDARITY trial co-sponsored by WHO and participating countries.(WHO). The study treatments are remdesivir, chloroquine or hydroxychloroquine, lopinavir plus ritonavir, and interferon-beta.(ISRCTN Registry) The risk of the virus continuing to spread globally remains high. Insufficient number of test kits, Personal Protective Equipment (PPE), and the lack of discipline in maintaining social distancing will only make matters worse in the coming days. Globally, national health security is weak. Most countries are not fully prepared to deal with a pandemic. Even the US, which is ranked first in the world, has shown it does not have the capacity yet to do wide spread testing or produce sufficient PPE.
- 4. DISTRIBUTION OF COVID-19 CASES WORLDWIDE (AS OF 7 APRIL 2020) SOURCE: WHO SITREP #77
- 5. SITUATION - UNITED STATES • All 50 states have reported cases of COVID-19 (CDC) • Case numbers are expected to continue rising through the coming weeks, as more cases are identified through testing. • Different parts of the country are seeing different levels of COVID-19 activity. The acceleration of the spread of the diseases, however, is slowing down. • In response, many states have closed schools and businesses; banned gatherings, meetings and sporting events; and, in some places, residents have been ordered to stay inside. Currently, the majority of cases have been recorded in New York, New Jersey, Michigan, California, and Louisiana. • All states except for North Dakota, South Dakota, Nebraska, and Arkansas. have implemented statewide curfews; directed the closure of non essential business and food operations restricted to carry out/delivery only. • Louisiana announced 35 new deaths on Monday, for a total of 512, a number that had more than doubled in a week., however, the curve is flattening. • All state Emergency Operations Centers are activated for COVID-19 RISK ASSESSMENT RISK TO GENERAL POPULATION RISK TO ELDERLY POPULATION RISK TO HEATHCARE SYSTEM CAPACITY MODERATE VERY HIGH HIGH UNITED STATES CONFIRMED CASES DEATHS RECOVERED 398,809 12,895 22,224 HOSPITALIZATIONS Cumulative COVID-19-associated hospitalization rates since March 1, 2020, will be updated weekly. The overall cumulative hospitalization rate is 4.6 per 100,000, with the highest rates in persons 65 years and older (13.8 per 100,000) and 50-64 years (7.4 per 100,000). (CDC) The densely populated New York metro area has been hit the hardest so far, in terms of a total count. And the area accounts for more than 40 percent of total U.S. deaths from the virus, which had infected more than 365,000 U.S. residents, as of Monday night, 7 April 2020. (NYT) Although many European countries have more coronavirus deaths per capita, the United States has the third-highest death toll in the world. DEATH
- 6. CASES IN THE US SOURCE: NYT SOURCE:JOHN HOPKINS UNIVERSITY COVID-19 GLOBAL CASES (CSSE) SOURCE: NYT The number of known cases of the coronavirus in the United States has surged. As 07 April, 1000 hrs. EST, at least 368,449 people across every state, plus Washington, D.C., and three U.S. territories, have tested positive for coronavirus, and at least 10,959 patients with the virus have died and 19,843 have recovered.
- 7. WHERE CASES ARE RISING
- 8. NYT STATES WITH A LOCKDOWN ORDER OR ADVISORY STATE DATE ENACTED Alabama April 4, 2020 Alaska March 28, 2020 Arizona March 31, 2020 California March 19, 2020 Colorado March 26, 2020 Connecticut March 23, 2020 Delaware March 24, 2020 Florida April 2, 2020 Hawaii March 25, 2020 Idaho March 25, 2020 Illinois March 21, 2020 Indiana March 24, 2020 Kansas March 30, 2020 Kentucky March 26, 2020 Louisiana March 23, 2020 Maine April 2, 2020 Maryland March 30, 2020 Massachusetts March 24, 2020 Michigan March 24, 2020 Minnesota March 27, 2020 Mississippi April 3, 2020 Missouri April 6, 2020 Montana March 28, 2020 Nevada April 2, 2020 New Hampshire March 27, 2020 New Jersey March 21, 2020 New Mexico March 24, 2020 New York March 22, 2020 North Carolina March 30, 2020 Ohio March 23, 2020 Oregon March 23, 2020 Pennsylvania April 2, 2020 Rhode Island March 28, 2020 Tennessee March 31, 2020 Texas April 2, 2020 Vermont March 25, 2020 Virginia March 30, 2020 Washington March 23, 2020 Washington, D.C. April 1, 2020 West Virginia March 24, 2020 Wisconsin March 25, 2020 The directives to keep people at home, which began in California in mid-March, have quickly swept the nation. Today, a vast majority of states, the Navajo Nation and many cities and counties have instructed residents to stay at home in a desperate race to stunt the spread of the coronavirus. This means at least 316 million people in at least 42 states, three counties, nine cities, the District of Columbia and Puerto Rico are being urged to stay home. A handful of more rural states — Arkansas, Iowa, Nebraska, North Dakota, and South Dakota — did not have statewide stay-at-home orders in place as of 7 April 2020. A few others had only partial orders, issued locally by cities or counties.
- 9. PROGRESS ON TESTING IN US NOTE: Data during the gray period of graph are incomplete because of the lag in time between when specimens are accessioned, testing is performed, and results are reported. The range was extended from 4 days to 7 days on March 26. (SOURCE: CDC) As of April 6th 2020, there have been approximately 1,917,095 COVID-19 tests administered in the United States. • 361,331 were positive • 1,555,764 were negative • 17,292 are pending CURRENT CDC TESTING PRIORITIES PRIORITY 1: Hospitalized Patients and Symptomatic Healthcare Workers PRIORITY 2: Patients in long-term care facilities with symptoms, patients 65+ with symptoms, patients with comorbidities with symptoms, first responders with symptoms PRIORITY 3: critical infrastructure workers with symptoms, any other individual with symptoms, health care workers and first responders, individuals with mild symptoms in communities experiencing high COVID-19 hospitalizations Non-Priority: Individuals without symptoms (CDC; COVID TRACKING) NUMBERS OF SPECIMENS TESTED FOR COVID-19 BY CDC LABS (N=4,904) AND U.S. PUBLIC HEALTH LABORATORIES*(N=207,446)
- 10. TESTING Doctors rely on a test called quantitative reverse-transcription polymerase chain reaction (qRT-PCR) to determine whether a person is infected with SARS-CoV-2. A team led by Nathan Grubaugh at Yale School of Public Health in New Haven, Connecticut, compared nine widely used versions of the test and found that all of them reliably detect the virus (C. B. F. Vogels et al. Preprint at medRxiv https://www.medrxiv.org/content/10.1101/2020.03.30.200481 08v1; 2020). But the researchers also found that some tests — including one made by the US Centers for Disease Control and Prevention, another developed at Hong Kong University, and a third from Charité–Universitätsmedizin Berlin — performed best when it came to detecting low levels of the virus in samples. Surgical face masks effectively block the spread of seasonal coronaviruses in respiratory droplets, suggesting that masks could prevent transmission of SARS-CoV-2. Seasonal coronaviruses are one cause of the common cold. Benjamin Cowling at the University of Hong Kong and his colleagues had ill volunteers who were infected with seasonal coronaviruses sit in an enclosed booth and place their faces in a sampling device, called the Gesundheit-II, that captures airborne particles (N. H. L. Leung et al. Nat. Med. https://doi.org/10.1038/s41591-020-0843-2; 2020). MASKS COULD CUT SPREAD OF COVID-19 VIRUS RESEARCH INTERSTATE TRANSMISSION OF COVID-19 IS NOW MOST URGENT THREAT Interstate transmission of the novel coronavirus that causes COVID-19 is now a much greater public health threat in the United States than cases coming into the country via international travel, a new study led by researchers at the Yale School of Public Health finds. The findings underscore a critical need for more widespread diagnostic testing for COVID-19 at the state level as well as more intense tracking of individuals who may have been exposed to SARS-CoV-2, the highly infectious virus that causes COVID-19. The researchers based their findings on a genomic analysis of nine virus samples collected from some of Connecticut’s first COVID-19 patients in mid-March. Using genomic epidemiology, they sequenced the unique genetic code or ‘signature’ for each sample. They then compared that Connecticut data to the known signatures of 168 sequenced genomes for SARS-CoV-2 from the United States and around the world. The study found that most of Connecticut’s virus samples were more closely linked to outbreaks in other states than international locations such as Europe and China. (Joseph R Fauver et al. Preprint at medRxiv https://www.medrxiv.org/content/10.1101/2020.03.25.20043828v1; 2020
- 11. UPDATES ON CLINICAL MANAGEMENT WITH CO-MORBIDITY Preexisting heart conditions (hypertension and coronary heart disease) have been the most common category of comorbidity. While the focus of the COVID-19 pandemic has been on respiratory problems and securing enough ventilators, an initial study found cardiac damage in 1 in 5 patients, leading to heart failure and death even among those who show no signs of respiratory distress. This will require a need for new precautions in people with preexisting heart problems, new demands for equipment and, new treatment plans for damaged hearts among those who survive. The question of whether the emerging heart problems are caused by the virus itself or are a by-product of the body’s reaction to it has become one of the critical unknowns facing doctors. If doctors in New York, Washington state and other hot spots can start to tease out how the virus is affecting the heart, they may be able to provide a risk score or other guidance to help clinicians manage COVID-19 patients in other parts of the country. RISK FACTORS Source: NYT, NCBI PREEXISTING HEART CONDITIONS Comorbidities that have been associated with severe illness and mortality include • Cardiovascular disease • Diabetes mellitus • Hypertension • Chronic lung disease • Cancer • Chronic kidney disease In a subset of 355 patients who died with COVID-19 in Italy, the mean number of pre-existing comorbidities was 2.7, and only 3 patients had no underlying conditions. Source: NCBI Hazard ratio for risk factors associated with admission to ICU, ventilation or death:
- 12. EMERGING ISSUES VENTILATORS Somewhere between 10% and 25% of patients sick with COVID-19 eventually require assistance to breathe. Roughly 5% of patients will develop acute respiratory distress syndrome, at which point only a mechanical ventilator can drive oxygen into their lungs and push fluid out. Recognizing the difference between those whose survival depends on access to a ventilator and those who can recover with less aggressive breathing assistance has become a vital skill for doctors. The United States has roughly 173,000 ventilators scattered across the country, according to the Center for Health Security at Johns Hopkins University. It may sound like a lot, but there could be 31 times as many patients who need one, experts from Harvard Medical School predict. Acute respiratory distress syndrome, or ARDS, is the endgame for the unluckiest COVID-19 patients. By that point, the virus has begun to destroy the tiny compartments in the lungs where blood normally collects oxygen. Roaring inflammation, a response to infection, further deteriorates the lung’s ability to draw in air. Without help, these patients could drown. More than a dozen medications that were developed to treat other diseases are now being tested on COVID-19 patients. Ideally, several of them would allow patients with mild to moderate symptoms recover before their illnesses reach the severe or critical stage. REMDESIVIR Remdesivir is an investigational intravenous drug with broad antiviral activity that inhibits viral replication through premature termination of RNA transcription and has in-vitro activity against SARS-CoV-2 and in-vitro and in-vivo activity against related betacoronaviruses. Information about clinical trials of remdesivir is available at ClinicalTrials.govexternal icon. Remdesivir is also available through an expanded access programexternal icon from the manufacturer, Gilead Sciences. HYDROXYCHLOROQUINE AND CHLOROQUINE Hydroxychloroquine and chloroquine are oral prescription drugs that have been used for treatment of malaria and certain inflammatory conditions. Hydroxychloroquine and chloroquine are under investigation in clinical trials for pre-exposure or post-exposure prophylaxis of SARS-CoV-2 infection, and treatment of patients with mild, moderate, and severe COVID-19. More information on clinical trials can be found at ClinicalTrials.govexternal icon. FDA issued an Emergency Use Authorization (EUA) to authorize use of chloroquine and hydroxychloroquineexternal icon from the Strategic National Stockpile for treatment of hospitalized adults and adolescents (weight ≥50 kg) with COVID-19 for whom a clinical trial is not available or participation is not feasible. OTHER DRUGS Several other drugs (e.g., investigational antivirals, immunotherapeutic, host- directed therapies) are under investigation in clinical trials or are being considered for clinical trials of pre-exposure prophylaxis, post-exposure prophylaxis, or treatment of COVID-19 in the United States and worldwide. Information on registered clinical trials for COVID-19 in the United States is available at ClinicalTrials.govexternal icon. FDA has issued guidance for administering or studying use of convalescent plasma for treatmentexternal icon of patients with COVID-19. DRUGS
- 13. COVID-19 PROJECTIONS HOSPITAL RESOURCE - US 8 DAYS UNTIL PEAK RESOURCE USE ON APRIL 15, 2020 RESOURCES NEEDED FOR COVID-19 PATIENTS ON PEAK DATE All beds needed: 140,823bed Bed Shortage: 36,654beds ICU beds needed: 140,823bed ICU Bed Shortage: 36,654beds Invasive ventilators needed: 24,828 SOURCE: IHME COVID-19 projections assuming full social distancing through May 2020
- 14. VID-19 projections assuming full social distancing through May 2020 SOURCE: IHME
- 15. HEALTHCARE INNOVATIONS TO FIGHT COVID-19 (DIAGNOSTICS) DIAGNOSTICS SOURCE : https://www.av.co/covid-diagnostics @vasudevbailey @zoeguttendorf
- 16. HEALTHCARE INNOVATIONS TO FIGHT THE PANDEMIC (TREATMENTS) SOURCE : https://www.av.co/covid-diagnostics @vasudevbailey @zoeguttendorf
- 17. HEALTHCARE INNOVATIONS TO FIGHT THE PANDEMIC (VACCINES) SOURCE : https://www.av.co/covid-diagnostics 6 @vasudevbailey @zoeguttendorf
