1.Unit processes used in pharmacy- INTRODUCTION (1).pdf
- 1. Associate Professor & Research coordinator, Department of Pharmaceutics, KIU-WC.
- 2. Pharmaceutical Processing is the process of drug manufacturing and can be broken down into a range of unit operations, such as blending, granulation, milling, coating, tablet pressing, filling, and others. Unit Operations: Unit operation is a basic step in a process. Unit operations involve a physical change or chemical transformation such as separation, crystallization, evaporation, filtration, polymerization, iso-merization, and other reactions. For example, in milk processing, homogenization, pasteurization, chilling, and packaging are each unit operations which are connected to create the overall process. A process may require many unit operations to obtain the desired product from the starting materials. 1. Fluid flow processes: including fluids transportation, filtration, and solids fluidization. 2. Heat transfer processes: including evaporation, condensation, and heat exchange. 3. Mass transfer processes: including gas absorption, distillation, extraction, adsorption, and drying. 4. Thermodynamic processes: including gas liquefaction, and refrigeration. 5. Mechanical processes: including solids transportation, crushing and pulverization, and screening and sieving. Unit Operations- Classifications INTRODUCTION
- 3. •A ''unit process'' is one or more grouped operations in a manufacturing system that can be defined and separated from others. •Unit process involves a chemical change or sometimes it is referred as chemical changes along with Physical Change leading to the synthesis of various useful product. Example: Hydrogenation, oxidation, nitration etc. Unit process
- 4. History and Background 1) Arthur Dehon Little propounded the concept of "unit operations" to explain industrial chemistry processes in 1916. 2) In 1923, William H. Walker, Warren K. Lewis and William H. McAdams wrote the book “The Principles of Chemical Engineering” and explained that the variety of chemical industries have processes which follow the same physical laws. Importance of unit operation
- 5. Drug: A drug product consists of therapeutics (API) and excipients combined in a delivery system. A drug product’s success lies in its ability to deliver the drug at a certain rate in a certain environment in the body.
- 7. Chapter -1 Flow of Fluid Fluids - The substances which have ability to flow. Solid –movement but size, shape does not charge, Liquid –movement and deformation in shape. Gas – Same So, liquid & gas are fluids. Fluid pressure: The pressure which apply by the fluid. Also known as “Hydrostatic pressure”. Pressure = Force /Area = F/A = mass (m) × gravitational (g) / Area (A) The solution to a fluid dynamics problem typically involves the calculation of various properties of the fluid, such as flow velocity, pressure, density, and temperature, as functions of space and time. Thermodynamic equation: P= where p is pressure, ρ is density, T the absolute temperature, while Ru is the gas constant and M is molar mass for a particular gas. ρRuT M Reynolds number: It is used for measurement and type of flow determination. Re = D× u × density of liquid/Viscosity of fluid D = diameter of pipe, u = Average velocity When Re<2000 then flow is laminar or viscous or streamline Re>4000 then flow is turbulent Re is 2000–4000 then flow is laminar or turbulent Reynolds number is used to predict the nature of the flow. Bernoulli's equation: Used in the measurement of rate of fluid flow. It applied in the working of the centrifugal pump, in this “ kinetic energy is converted in to pressure”.
- 8. Chapter 2 Heat and Mass Transfer Heat transfer is the movement of heat energy from one substance to another. • Heat will continue to move until both substances are the same temperature. • The greater the difference in temperature between the substances, the faster the heat will transfer.
- 9. Chapter 3 Filtration Filtration, the process in which solid particles in a liquid or gaseous fluid are removed by the use of a filter medium that permits the fluid to pass through but retains the solid particles. Either the clarified fluid or the solid particles removed from the fluid may be the desired product. 1. Surface filtration (Screen filtration): E.g., Membrane filtration 2. Depth filtration: E.g., Ceramic filtration, Sintered filtration 3. Cake filtration: E.g., Filter cake made from diatomite Types of Filtration
- 10. 3
- 13. Chapter 4 Centrifugation Centrifugation is a method of separating molecules having different densities by spinning them in solution around an axis (in a centrifuge rotor) at high speed. It is one of the most useful and frequently employed techniques in the molecular biology laboratory. Centrifugation
- 16. Chapter 5 Size Reduction Size reduction is a process in which the particle size of a solid is made smaller. The term size reduction is applied to ways in which particles of solids are cut or broken into smaller pieces. 1. Hardness: Harder the material, more difficult to reduce its size. 2. Toughness: Soft but tough material creates problem in size reduction and its toughness is reduced by decrease temperature. 3. Stickness: Gum and resinoussubstancescause problemin size reduction. 4. Moisture content: <5% moisture suitable for dry grindingand >50% for wet grinding. Factors affecting size reduction
- 18. Chapter 6 Mixing of Solids Definition: It is a unit operation in which two or more than two components in separately or roughly mixed. Mixing Equipments 1. Liquid mixers 3. Semi-solid mixers A. Agitator mixers B. Shear mixers
- 19. Chapter 7 Extraction Extraction is the method of removing active constituents from a solid or liquid by means of liquid solvent. The separation of medicinally active portions of plant or animal tissues from the inactive or inert components by using selective solvents. Extracts are prepared by using ethanol or other suitable solvent. Extract: Extracts can be defined as preparations of crude drugs which contain all the constituents which are soluble in the solvent. Marc: Solid residue obtain after extraction. Menstrum: Solvent used for extraction. In this method the wanted components are dissolved by the use of selective solvents known as menstrum & undissolved part is a marc. After the extraction unwanted matter is removed.
- 20. Chapter 8 DISTILLATION It is defined as the separation of the components of the liquid mixture by process involving vaporization and subsequent condensation at another place. Psolution = Xsolvent . Psolvent Raoult’s law: It states that a solvent's partial vapour pressure in a solution (or mixture) is equal or identical to the vapour pressure of the pure solvent multiplied by its mole fraction in the at given temperature. Psolution = Vapor pressure of the solution Xsolvent = mole fraction of the solvent Psolvent = vapor pressure of the pure solvent. Ideal solution obeys (behaves) Raoult’s law. Raoult’s law is obeyed by only a few solution of liquid in liquids. Examples are benzene, toluene, n-hexane, n-heptane, ethyl bromide, ethyl iodide. Dalton’s law: It states that the total pressure p of moist air is the sum of partial pressures of dry air pa and water vapor pv: p = pa + pv
- 21. Chapter 9 Evaporation Defined as a process by which a liquid is transformed into vapour.
- 22. Chapter 10 Crystallization Crystallization is the physical transformation (phase transition) of a liquid, solution, or gas to a crystal, which is a solid with an ordered internal arrangement of molecules, ions, or atoms.
- 23. Chapter 11 Drying Drying is the process of using evaporation to remove water from a solution, suspension, or other solid-liquid mixture. In addition to solids, the process can also be used to remove water from liquids or gases.
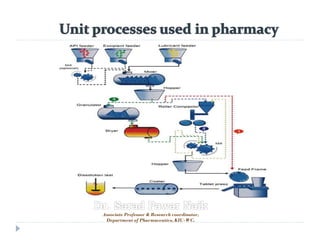
- 24. Formulation and pre-formulation development Before a drug can be manufactured at any scale, much work goes into the actual formulation of the drug. Formulation development scientists must evaluate a compound for uniformity, stability and many other factors. After the evaluation phase, a solution must be developed to deliver the drug in its required form such as solid, semi-solid, immediate or controlled release, tablet, capsule.
- 25. Powder blending In the pharmaceutical industry, a wide range of excipients may be blended together to create the final blend used to manufacture the solid dosage form. The range of materials that may be blended (excipients, API), presents a number of variables which must be addressed to achieve products of acceptable blend uniformity. These variables may include the particle size distribution (including aggregates or lumps of material), particle shape (spheres, rods, cubes, plates, and irregular), presence of moisture (or other volatile compounds), and particle surface properties (roughness, cohesivity).
- 26. Milling During the drug manufacturing process, milling is often required in order to reduce the average particle size in a drug powder. There are a number of reasons for this, including Increasing homogeneity and dosage uniformity, Increasing bioavailability, and Increasing the solubility of the drug compound.
- 27. Granulation Granulation can be thought of as the opposite of milling; It is the process by which small particles are bound together to form larger particles, called “granules”. Granulation is used for several reasons. Granulation prevents the "de-mixing" of components in the mixture, by creating a granule which contains all of the components in their required proportions, Improves flow characteristics of powders (because small particles do not flow well), and Improves compaction properties for tablet formation.
- 28. Hot melt extrusion Hot melt extrusion is utilized in pharmaceutical solid oral dose processing to enable delivery of drugs with poor solubility and bioavailability. Hot melt extrusion has been shown to molecularly disperse poorly soluble drugs in a polymer carrier increasing dissolution rates and bioavailability. The process involves the application of heat, pressure and agitation to mix materials together and 'extrude' them through a die. Twin-screw high shear extruders blend materials and simultaneously break up particles. The resulting particles can be blended and compressed into tablets or filled into capsules.