6_Poster_Bassam_Hb
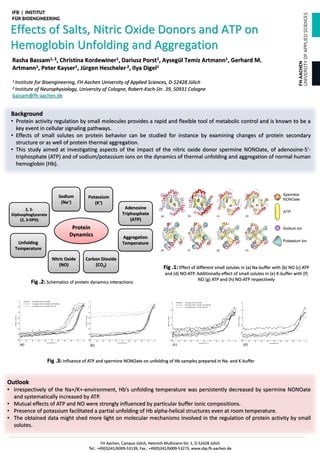
- 1. Effects of Salts, Nitric Oxide Donors and ATP on Hemoglobin Unfolding and Aggregation Rasha Bassam1, 2, Christina Kordewiner1, Dariusz Porst1, Aysegül Temiz Artmann1, Gerhard M. Artmann1, Peter Kayser1, Jürgen Hescheler 2, Ilya Digel1 1 Institute for Bioengineering, FH Aachen University of Applied Sciences, D-52428 Jülich 2 Institute of Neurophysiology, University of Cologne, Robert-Koch-Str. 39, 50931 Cologne bassam@fh-aachen.de Fig .1: Effect of different small solutes in (a) Na-buffer with (b) NO (c) ATP and (d) NO-ATP. Additionally effect of small solutes in (e) K-buffer with (f) NO (g) ATP and (h) NO-ATP respectively Fig .3: Influence of ATP and spermine NONOate on unfolding of Hb samples prepared in Na- and K-buffer Fig .2: Schematics of protein dynamics interactions Background • Protein activity regulation by small molecules provides a rapid and flexible tool of metabolic control and is known to be a key event in cellular signaling pathways. • Effects of small solutes on protein behavior can be studied for instance by examining changes of protein secondary structure or as well of protein thermal aggregation. • This study aimed at investigating aspects of the impact of the nitric oxide donor spermine NONOate, of adenosine-5'- triphosphate (ATP) and of sodium/potassium ions on the dynamics of thermal unfolding and aggregation of normal human hemoglobin (Hb). Outlook • Irrespectively of the Na+/K+-environment, Hb’s unfolding temperature was persistently decreased by spermine NONOate and systematically increased by ATP. • Mutual effects of ATP and NO were strongly influenced by particular buffer ionic compositions. • Presence of potassium facilitated a partial unfolding of Hb alpha-helical structures even at room temperature. • The obtained data might shed more light on molecular mechanisms involved in the regulation of protein activity by small solutes. Protein Dynamics Adenosine Triphosphate (ATP) 2, 3- Diphosphoglycerate (2, 3-DPG) Sodium (Na+) Potassium (K+) Nitric Oxide (NO) Carbon Dioxide (CO2) Aggregation TemperatureUnfolding Temperature FH Aachen, Campus Jülich, Heinrich-Mußmann-Str. 1, D-52428 Jülich Tel.: +49(0)241/6009-53139, Fax.: +49(0)241/6009-53273, www.zbp.fh-aachen.de