Med device processes
•Download as PPTX, PDF•
0 likes•222 views
The document discusses the medical product realization process and medical device quality system processes. It outlines the steps involved from concept development through design, manufacturing, and post-production activities like monitoring complaints and corrective actions. The overall goal is to design and produce safe and effective medical devices that meet regulatory requirements.
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (19)
Role of quality systems and audits in pharmaceutical manufacturing environment

Role of quality systems and audits in pharmaceutical manufacturing environment
White Paper - Application of FDA Current Good Manufacturing Practices

White Paper - Application of FDA Current Good Manufacturing Practices
New medical device regulation: implications for medical device manufacturers

New medical device regulation: implications for medical device manufacturers
EU Medical Device Regulation: Preparing for Disruptive (yet Incomplete) Regu...

EU Medical Device Regulation: Preparing for Disruptive (yet Incomplete) Regu...
Viewers also liked
Viewers also liked (20)
How to Start a Med Device Startup From Your Garage - Vancouver Edition

How to Start a Med Device Startup From Your Garage - Vancouver Edition
2015-CLSA-Report-Recent-Trends-in-FDA-Med-Device-Regulation-Final

2015-CLSA-Report-Recent-Trends-in-FDA-Med-Device-Regulation-Final
Healthcare & Med Device - LiveWorx Recommended Agenda

Healthcare & Med Device - LiveWorx Recommended Agenda
Modern SEO Techniques Part 1:New SEO Methods for Medical Device Marketers

Modern SEO Techniques Part 1:New SEO Methods for Medical Device Marketers
Med Device Vendors Have Big Opportunities in Health IT Software, Services, an...

Med Device Vendors Have Big Opportunities in Health IT Software, Services, an...
Life Science Fast Track Faculty and Mentor Directory

Life Science Fast Track Faculty and Mentor Directory
Swipes pitch deck for Beta Pitch 2013 Finals in Berlin

Swipes pitch deck for Beta Pitch 2013 Finals in Berlin
The deck we used to raise $270k for our startup Castle

The deck we used to raise $270k for our startup Castle
Med device processes
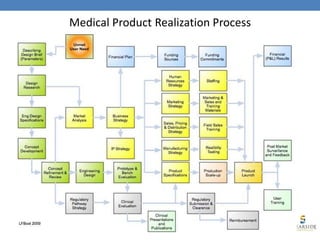
- 1. Medical Product Realization Process
- 2. Medical Device Quality System Processes