The Quest to Win Through Science in Pancreatic Cancer: From Sequencing Current Options to Testing Novel Approaches
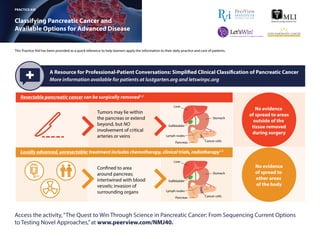
- 1. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Classifying Pancreatic Cancer and Available Options for Advanced Disease FOLFIRINOX: 5-FU, leucovorin, irinotecan, and oxaliplatin; RR: response rate. 1. https://www.lustgarten.org/get-informed/stages-of-pancreatic-cancer. Accessed December 21, 2017. 2. NCCN Clinical Practice Guidelines in Oncology. Pancreatic Adenocarcinoma. Version 3.2017. September 11, 2017. https://www.nccn.org/professionals/ physician_gls/pdf/pancreatic.pdf. Accessed December 21, 2017. 3. Conroy T et al. N Engl J Med. 2011;364:1817-1825. 4. Von Hoff DD et al. N Engl J Med. 2013;369:1691-1703. 5. Goldstein D et al. J Natl Cancer Inst. 2015;107(2). 6. Wang-Gillam A et al. Lancet. 2016;387:545-557. PRACTICE AID Access the activity,“The Quest to Win Through Science in Pancreatic Cancer: From Sequencing Current Options to Testing Novel Approaches,”at www.peerview.com/NMJ40. A Resource for Professional-Patient Conversations: Simplified Clinical Classification of Pancreatic Cancer More information available for patients at lustgarten.org and letswinpc.org Resectable pancreatic cancer can be surgically removed1,2 Confined to area around pancreas; intertwined with blood vessels; invasion of surrounding organs Tumors may lie within the pancreas or extend beyond, but NO involvement of critical arteries or veins Liver Stomach Gallbladder Lymph nodes Pancreas Cancer cells Locally advanced, unresectable: treatment includes chemotherapy, clinical trials, radiotherapy1,2 Liver Stomach Gallbladder Lymph nodes Pancreas Cancer cells No evidence of spread to areas outside of the tissue removed during surgery No evidence of spread to other areas of the body
- 2. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Classifying Pancreatic Cancer and Available Options for Advanced Disease FOLFIRINOX: 5-FU, leucovorin, irinotecan, and oxaliplatin; RR: response rate. 1. https://www.lustgarten.org/get-informed/stages-of-pancreatic-cancer. Accessed December 21, 2017. 2. NCCN Clinical Practice Guidelines in Oncology. Pancreatic Adenocarcinoma. Version 3.2017. September 11, 2017. https://www.nccn.org/professionals/ physician_gls/pdf/pancreatic.pdf. Accessed December 21, 2017. 3. Conroy T et al. N Engl J Med. 2011;364:1817-1825. 4. Von Hoff DD et al. N Engl J Med. 2013;369:1691-1703. 5. Goldstein D et al. J Natl Cancer Inst. 2015;107(2). 6. Wang-Gillam A et al. Lancet. 2016;387:545-557. PRACTICE AID Access the activity,“The Quest to Win Through Science in Pancreatic Cancer: From Sequencing Current Options to Testing Novel Approaches,”at www.peerview.com/NMJ40. Metastatic pancreatic cancer: chemotherapy can be used across multiple lines of therapy; clinical trials are an important treatment option1,2 Modern chemotherapy options in advanced pancreatic cancer Has spread beyond the area of the pancreas and involves other organs • Improves RR, PFS, OS • Cytopenias, fatigue, peripheral sensory neuropathy; consider primary growth factor prophylaxis • Biweekly dosing; requires infusion pump, central IV access • Improves RR, PFS, OS • Cytopenias, fatigue, alopecia, peripheral sensory neuropathy (usually reversible) • 3 of 4 weeks dosing schedule • Improves objective response, PFS, OS • Neutropenia, fatigue, diarrhea, vomiting • IV infusion every 2 weeks FOLFIRINOX3 Gemcitabine + nab-paclitaxel4,5 Nanoliposomal irinotecan (nal-IRI)6 Liver Stomach Gallbladder Lymph nodes Pancreas Cancer cells Involved organs may include the liver and lungs; other areas of the abdomen may be affected
- 3. Access the activity,“The Quest to Win Through Science in Pancreatic Cancer: From Sequencing Current Options to Testing Novel Approaches,”at www.peerview.com/NMJ40. AGuidetoClinicalTrialsinPancreatic Cancer:FocusonModernTreatment PlatformsandNovelTherapeutics PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. 5-FU: 5 fluorouracil; CT: chemotherapy; FOLFIRINOX: 5-FU, leucovorin, irinotecan, and oxaliplatin; FOLFOX: folinic acid, fluorouracil, and oxaliplatin; IDO: indoleamine 2,3 dioxygenase; LV: leucovorin; nab-paclitaxel: nanoparticle albumin–bound paclitaxel; nal-IRI: nanoliposomal irinotecan; SBRT: stereotactic body radiation therapy; TTFields: tumor-treating fields. HALO-109-301: PEGPH20 ± gemcitabine/ nab-paclitaxel in hyaluronan-high disease (NCT02715804) CARRIE: MM-141 (IGF-1R and ErbB3 antibody [NCT02399137]) NLG2104: Indoximod (IDO inhibitor [NCT02077881]) Nivolumab + cisplatin + paricalcitol (NCT02754726) ± olaratumab (NCT03086369) 144525: ibrutinib (NCT02562898) Nal-IRI + 5-FU/LV + oxaliplatin vs nal-IRI + 5-FU/LV vs nab-paclitaxel + gemcitabine (NCT02551991) POLO: olaparib in gBRCA-mutated disease that has not progressed on first-line CT (NCT02184195) CanStem111P: napabucasin (cancer stemness inhibitor ± gemcitabine/nab-paclitaxel) (NCT02993731) Clinical trials can be an important therapeutic tool for the treatment of pancreatic cancer These selected trials and other resources provided can be shared with your patients Frontline Metastatic Phase3 Combinations With Gemcitabine + Nab-Paclitaxel Other Frontline/ Maintenance Trials Phase 2 Previously Treated Metastatic Olaparib BRCAness phenotype in pancreatic cancer (NCT02677038) PembroPlus: pembrolizumab + nab-paclitaxel/ gemcitabine (phase 1/2; NCT02331251) NivoPlus: + CT (phase 1/2; NCT02423954) Sequoia: AM0010+FOLFOXvsFOLFOX (NCT02923921) Olaparib + cediranib (NCT02498613) Phase2
- 4. Access the activity,“The Quest to Win Through Science in Pancreatic Cancer: From Sequencing Current Options to Testing Novel Approaches,”at www.peerview.com/NMJ40. AGuidetoClinicalTrialsinPancreatic Cancer:FocusonModernTreatment PlatformsandNovelTherapeutics PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. 5-FU: 5 fluorouracil; CT: chemotherapy; FOLFIRINOX: 5-FU, leucovorin, irinotecan, and oxaliplatin; FOLFOX: folinic acid, fluorouracil, and oxaliplatin; IDO: indoleamine 2,3 dioxygenase; LV: leucovorin; nab-paclitaxel: nanoparticle albumin–bound paclitaxel; nal-IRI: nanoliposomal irinotecan; SBRT: stereotactic body radiation therapy; TTFields: tumor-treating fields. Let’sWin offers information for patients on therapies being tested in clinical trials Pancreatic Cancer Action Network offers several clinical trial resources Other Settings Clinical trial search for patients and professionals Clinical trial FAQ Free education packet Informational video clinicaltrials.pancan.org Neoadjuvant therapy Experimental drug combinations Immunotherapy Maintenance therapy Radiation … and much more letswinpc.org/clinical-trials Second-line nab-paclitaxel + gemcitabine vs FOLFOX for post-resection gemcitabine-refractory disease (NCT02506842) RTOG 0848: gemcitabine ± erlotinib → CT ± radiation (NCT01013649) Phase3PostResection PANOVA-3: TTFields with concomitant gemcitabine and nab-paclitaxel (NCT03377491) PANC0015/27492: FOLFIRINOX +/- SBRT (NCT01926197) Phase3 LocallyAdvanced S1505: nab-paclitaxel/gemcitabine or FOLFIRINOX (NCT02562716) PEGPH20, gemcitabine, and nab-paclitaxel; run-in followed by neoadjuvant treatment (NCT02487277) Gemcitabine/nab-paclitaxel + radiation followed by surgery (NCT02481635) UVA-PC-PD101: chemoradiotherapy ± pembrolizumab (NCT02305186) Phase2 Neoadjuvant
