Finding the Path to Improved Recognition and Management of Tardive Dyskinesia
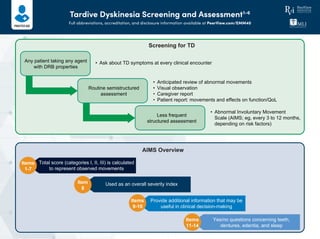
- 1. Tardive Dyskinesia Screening and Assessment1-6 Full abbreviations, accreditation, and disclosure information available at PeerView.com/EMM40 Any patient taking any agent with DRB properties • Ask about TD symptoms at every clinical encounter • Anticipated review of abnormal movements • Visual observation • Caregiver report • Patient report: movements and effects on function/QoL • Abnormal Involuntary Movement Scale (AIMS; eg, every 3 to 12 months, depending on risk factors) Routine semistructured assessment Less frequent structured assessment Total score (categories I, II, III) is calculated to represent observed movements Items 1-7 Used as an overall severity indexItem 8 Provide additional information that may be useful in clinical decision-making Items 9-10 Yes/no questions concerning teeth, dentures, edentia, and sleep Items 11-14 AIMS Overview Screening for TD
- 2. Tardive Dyskinesia Screening and Assessment1-6 Full abbreviations, accreditation, and disclosure information available at PeerView.com/EMM40 1. Johnson BG. Nursing. 2017;47:56-60. 2. Caroff SN et al. J Clin Psychiatry. 2020;81:19cs12983. 3. Caroff SN et al. Psychiatr Clin North Am. 2016;39:391-411. 4. Correll CU et al. J Clin Psychiatry. 2017:78:1136-1147. 5. Citrome L at al. Am J Manage Care. 2007;13(Suppl):1-12. 6. Kane JM et al. J Clin Psychiatry. 2018;79:17cs11959. Criteria • Involuntary movements during exposure to or after withdrawal from antipsychotic or other dopamine-blocking agent • Differential diagnosis • Patient history • Cumulative exposure to antipsychotic medication • Current medication • Duration and severity of involuntary movements Consider neurologic consultation in these cases • Atypical presentation or course • Family history of movement or neurodegenerative disorders • Presence of other signs/symptoms • Uncertainty in tardive dyskinesia diagnosis • Unexpected treatment response More Common • Akathisia • Drug-induced parkinsonism or tremor • Edentulous dyskinesia • Spontaneous dyskinesia • Drug-induced dyskinesia caused by other agents • Chronic motor tics • Autism • Blepharospasm • Huntington’s disease • Meige syndrome • Psychotic stereotypies • Restless leg syndrome • Senile chorea • Sydenham chorea • Tourette syndrome • Wilson disease • Rett syndrome Less Common Diagnosis of Tardive Dyskinesia Tardive Dyskinesia: Differential Diagnosis
- 3. Recommendations: Use of Adjunctive Therapies for TD1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/EMM40 1. Bhidayasiri R et al. J Neurol Sci. 2018;389:67-75. Troublesome symptoms Nontroublesome symptoms Nonapproved indications Level A: New VMAT2 inhibitors (or tetrabenazine) Level B: Clonazepam or Ginkgo biloba Level C: Amantadine Level C: GPi DBPS Determining lowest effective dosage Determining lowest effective dosage Review by specialist to identify suitable alternative Improved Improved Improved ImprovedImproved Improved Not improved and still troublesome Not improved and still troublesome Not improved and still troublesome Not improved and still troublesome Context for antipsychotic/ dopamine-blocking agent use TD assessment Dosage evaluation Consider suppressive agents Monitoring and follow-up assessment Close observation and regular follow-up Close observation and regular follow-up Approved indications Tardive dyskinesia
- 4. FDA-Approved VMAT2 Inhibitors for TD1-3 Full abbreviations, accreditation, and disclosure information available at PeerView.com/EMM40 a Also consider hypersensitivity to compounds related to valbenazine (ie, tetrabenazine, deutetrabenazine). Characteristics of VMAT2 Inhibitors Characteristic Valbenazine Deutetrabenazine Dosing frequency Once daily Twice daily Initial dose 40 mg/d 12 mg/d (divided doses) Recommended dose 80 mg/d 12-48 m/d (divided doses) Administer with or without food With or without With CYP interaction (substrate) CYP2D6, CYP3A4 CYP2D6 Contraindications Monoamine oxidase inhibitor, valbenazine hypersensitivitya MAOIs, valbenazine, reserpine, tetrabenazine, hepatic impairment Warnings/precautions Impaired driving/somnolence, QT prolongation, parkinsonism Impaired driving/somnolence, QT prolongation, parkinsonism, neuroleptic malignant syndrome Common adverse events Somnolence Nasopharyngitis, insomnia
- 5. FDA-Approved VMAT2 Inhibitors for TD1-3 Full abbreviations, accreditation, and disclosure information available at PeerView.com/EMM40 a Cases deemed not related to valbenazine; one case possibly related to deutetrabenazine (long-term trial in Huntington disease found no relationship). 1. McEvoy JP, Kremens DE. J Clin Psychiatry. 2020:81:1:NU18041AHSC. 2. Factor SA et al. J Clin Psychiatry. 2017;78:1344-1350. 3. Fernandez HH et al. J Neurol Neurosurg Psychiatry. 2019;90:1317-1323. Outcome Valbenazine (N = 189, 52 weeks) Deutetrabenazine (N = 343, 106 weeks) Any AE, n (%) 137 (69.2) 233 (67.9) Any AE leading to discontinuation, n (%) 31 (15.7) 26 (7.6) Any serious AE, n (%) 29 (14.6) 45 (13.1) Suicidal ideation, n (%) 10 (5.1)a 16/342 (5)a AEs with incidence ≥5% Headache, UTI, diarrhea, dizziness Headache, UTI, somnolence, depression, anxiety, diarrhea, nasopharyngitis Safety Outcomes in Long-Term Trials of Valbenazine and Deutetrabenazine Long-Term Efficacy of Valbenazine and Deutetrabenazine AIMS Mean Improvement With Deutetrabenazine Time, week MeanChangeFrom BaselineinAIMSScore -10 - 8 - 6 -4 -2 0 0 11040 50 60 70 80 90 100302010 AIMS Mean Improvement With Valbenazine Time, week -7 -6 -5 -4 -3 -1 -2 0 1 0 32 48 52166 8 AIMSScoreMeanChange FromBaseline(±SEM) End of DBPC End of washout End of extension
