ACL healing presentation.pptx
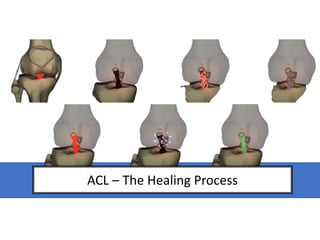
- 1. ACL – The Healing Process
- 2. Overview Native ACL rupture - reasons for reconstruction Post-operative healing: 1) Intra-articular remodelling: a) Early phase b) Proliferation - Recellularisation - Revascularisation c) Maturation/ Ligamentisation 2) Intra-tunnel incorporation
- 3. Why does the native ACL not heal? Inactive plasminogen Urokinase plasminogen activator (uPa) Plasmin Poor vascularisation Intra-articular joint Healthy Fibrinolysis Fibrinogen Natural, biological, fibrin-platelet scaffold (clot) cannot be formed Murray et al. (2000) Perrone et al.(2017) Smith & Noyes (2019)
- 4. Why does the native ACL not heal? No contact between “stumps” Poor vascularisation Murray et al. (2000) Smith & Noyes (2019) Intra-articular joint Retraction of the ligament remnant
- 5. Intra-articular remodelling & Intra-tunnel incorporation The Healing Process Janssen & Scheffler 2014
- 6. Intra-articular Remodelling a) Early phase b) Proliferation c) Maturation
- 7. Early phase (Week 1-4) Avascularisation (relative hypoxia) Partial Necrosis (*central)? (<30%) Inflammatory & fibroblastic responses: cytokines and growth factors (e.g. vascular endothelial growth factor) Initiates cascade of healing Johnson (1993); Amiel et al. (1986); Claes et al. (2011); Smith & Noyes (2019)
- 8. Proliferation (Week 4-12) (up to 10 months) Re-cellularisation ↑ fibroblastic density (*myofibroblasts) Mixed diameter fibrils to smaller diameter Increase in type III collagen/ fibronectin Become disorganised and lose crimp pattern ↑ collagen density Johnson (1993); Amiel et al. (1986); Grant & Mohtadi (2003); Scheffler et al. (2008); Claes et al. (2011); Smith & Noyes (2019)
- 10. Re-vascularisation 12 weeks post-op: >native ACL vascular density Hypothesised: angiogenesis enables maximal cellular activity?? From Early Phase ↑ expression of VEGF Neovascularisation Hoffa fat pad & synovium? External factors – smoking, drugs, diabetes Johnson (1993); Amiel et al. (1986); Grant & Mohtadi (2003); Scheffler et al. (2008); Claes et al. (2011); Smith & Noyes (2019); Proliferation (Week 4-12) (up to 10 months)
- 11. n = 18 Semitendinous autograft – VEGF (n = 9) 12 weeks post-op: ↑vascular density, fibroblastic content ↓ graft stiffness ↑ laxity Temporary or permanent?
- 12. Ligamentisation/ Maturation (week 12 +) (Up to 3 years) Graft cells to native-like cells: 1) ↑ glycoaminoglycans? (*myofibroblasts) 2) Collagen bundles attain a densely packed, parallel alignment 3) Crimp pattern re-develops 4) Collagen cross-linking progresses @ 2 years post: collagen content and density resembles but not same as native ACL (smaller diameter) Hydroxypyridium cross-link density (Ng et al. 1996): - Good correlation on regression analyses with Young’s Modulus - Comparable effects of increase of HP within ACL Johnson (1993); Ng et al. 1996; Amiel et al. (1986); Grant & Mohtadi (2003); Scheffler et al. (2008); Claes et al. (2011); Smith & Noyes (2019);
- 14. No real consensus on actual phase durations and limited human studies - Claes et al. (2011)
- 15. N = 67 Mid- substance biopsies Group 1 (6-12 months) Group 2 (13-24 months) Group 3 (>24 months) Myofibroblast density: ↑ Group 2 Collagen orientation: irregular in Group 1; “more regular” in 2 & 3 Group 1: ovoid; Group 2: spindle; Group 3:~narrow and long ↑ cellular & vascular density up to 24 months post-op Collagen orientation: not “native” by 117 months Hamstring remodelling: 12-24 months (Pauzenberger et al. 2013)
- 17. Incorporation • Systematic review (Lu et al. 2018) – not regenerated after aclr • 3 months (3-4 weeks): fibrovascular interface formed between tendon and bone • 6 months: fibrous interface throughout bone tunnel • 6-12 months: showed indirect insertion via Sharpey-like fibres intact native ACL – 4 distinct zones (Evans et al. 1990): 1) Ligament 2) Unmineralized fibrocartilage 3) Mineralisezed fibrocartilage 4) Bone Dissipation of forces Most findings from animal studies due to the invasive nature of biopsies – limited on humans and far from understood (Lu et al. 2018)
- 18. Adaptation NOT restoration Wilhelm Roux (1905) Self-differentiation vs Dependent differentiation
- 19. Jackson et al. (1993); Ng et al. (1995); Janssen & Scheffler (2013); Smith & Noyes (2019) Strongest: Wk 1
- 20. Zein et al. (2017)
- 21. Jackson et al. (1993); Ng et al. (1995); Janssen & Scheffler (2013); Smith & Noyes (2019) Strongest: Wk 1 Graft Failure: < Week 4: graft pull-out > Week 4: intra-articular (6-8 weeks post-op*) Full restoration of mechanical strength to intact ACL not observed in vivo: type III collagen? Most rehabilitative programmes are formed from animal progression
- 22. Graft loading: - Initial partial loading: maintain graft integrity; prevent stretching, laxity and future instability; optimise ingrowth and subsequent maturation of cellular and extracellular components Scheffler et al (2008); Smith & Noyes (201 No consensus on what optimal loading looks like throughout rehab – biological healing takes time
- 23. Summary A LOT OF DEBATE & HETEROGENEITY Hypoxia/Necrosis cell migration, proliferation, ECM synthesis, revascularisation Limited capacity for human in vivo studies Current RTS – modelled off animal biology? “Complete” healing: may take up to 3 years!!! Assisted by progressive loading
- 24. References See separate document for full list
Editor's Notes
- Focus on hamstring autograft
- GENERAL OVERVIEW – NOT REALLY SPECIFIC TO GRAFT TYPE EXCEPT IN INTRA-TUNNEL INCORPORTATION
- Injury to extra-articular ligaments, such as the MCL, lead to bleeding from surrounding tissues that delivers fibrinogen. This fibrinogen is cleaved by thrombin to fibrin – allowing for the formation of platelet-fibrin clot as these can coagulate. This clots acts as a scaffold for platelets, WBC, RBCs etc – which then allows a space for surrounding tissues to grow and connect. But in intra-articular ligaments, this cant happen. Synovial fluid surrounding the joint contacts plasminogen which is inactive in the healthy knee. When injured, a protease enzyme known as urokinase plasminogen activator is upregulated by synviocytes and this converts plasminogen to plasmin. Any fibrinogen in any bleeding is acted upon by plasmin by fibrinolysis which means that a natural and biological scaffold for ligament healing cannot occur.
- Movement of the knee and they position of the acl stumps left after rupture means that no consistent contact occurs to act as a structure to heal. A synovial tissue layer also forms around each stump end, encasing it and thickening and eventually contracting to restrict stump-to-stump healing. a) Disruption of the epiligament and synovial covering of the ligament (b) intimal hyperplasia of the vessels (c), and loss of the regular crimp structure near the site of injury (d). B: The epiligamentous regeneration phase, involving a gradual recovering of the ligament remnant by vascularized, epiligamentous tissue and synovial tissue (e). C: The proliferative phase, with revascularization of the remnant with groups of capillaries (f). D: The remodeling and maturation phase, characterized by a decrease in cell number density and blood vessel density (g) and by retraction of the ligament remanent
- - Partial / complete necrosis : complete in animals i.e. sheep A cascade of events here sets up for the latter phases as the cytokines and growth factors promote cell migration, proliferation, extracellular matrix synthesis (ie. Collagen), and the eventual revascularisation. Animal versus human – necrotic magnitude (3 and 6 weeks post-op), retained tissue from graft source, remodelling speed, degree of neovascularisation (dependent on necrosis?), quality of replacement tissue Within this stage there are no significant changes to collagen composition/ structure – retain
- Phenotype of the synovial fibroblasts that recolonize the graft likely differ from those of the native ACL – accounting for differences in ultra-structure and mechanical quality
- Cellular activity has slowed to native ACL levels – protein synthesis and growth factor release slow to native levels
- The higher the youngs modulus – the stiffer the material is stiffness is the resistance to stretch -> goats
- Sanchez – hamstring, falconiero mixed (*BPTB), abe BPTB, rougraff - BPTB No consensus – but gives indication of biological processes and timlines for return – where the biopsy is taken from, procedures used to determine remodelling status – optical microscrope versus electron microscope, thresholds utilised. Why its hard to draw on research for humans – animals: faster and more necrosis of which magnitude of neovascularisation is associated with – alongside differing weight-bearing, slight variations in surgical techniques, and mechanics – bipedal vs quadrupedal
- Biopsies from native HT (n = 17) and ACL (n = 8) served as controls. Strong increase in group 2 myofibroblast content, from 13 -24 months, indicated an activie remodelling process from 1 to 2 years. & Longer recovery – non-anatomic ACLR that used trans-tibial surgical techniques & No necrosis – too late Used factor VIII staining tehcniques tht can improve vessel detection possibility and special immunohistochemical staining techniques for mofibroblast density = not used by Sanchez.
- Sharpey’s-like fibres @ 12weeks for
- Tendon Healing in Bone Tunnel after Human Anterior Cruciate Ligament Reconstruction: A Systematic Review of Histological Results (Lu et al. 2018) Sharkey-like collagen fibres that connect the tendon graft to the bone has been described as is viewed as the earlier sign of osteointegration (Lui et al. 1997). 28) Evans et al 1990 - Fibrocartilage in the attachment zones of the quadriceps tendon and patellar ligament of man. Rossetti et al 2017 - The microstructure and micromechanics of the tendon-bone insertion.
- In 1905, Wilhem Roux stated the law of tissue adaptation (developmental mechancis as he labelled it) to which a tissue can adapt its structure to be better suited to an meet the demands of an alterative circumstance, possibly within the environment. With self-determinantion, the inherit structural and functional development of a specific organ or tissue is preprogrammed. Dependent differation, on the other hand, attempts to exmaplin how external stimuli can drive purposeful and biological adaptions to tissues to allow for improve function. As a graft integrates into its host, its intraarticular component experiences functional adaptions and reorgainziation of its internal structure to closely resemble a native acl. ( 1min 14)
- Strongest: Wk 1 Weakest: ~Wk 6-8 – revascularisation, cellular infiltration, loss of regular collagen oritentation and crimp pattern and the phases of collagen characteristisc (density, magnitude, size changes) Tendon healing in bone tunnel is influed by mechanical stress – it has been shown that the differentiation of mesenchymal stem cells is directly influend by pressure and tenson. Maximum cellular activity and remodelling (weeks 4 -10) - mechanical weakening graft (much weaker than native) Increase in type III collagen – mechanical strength of graft is not restored to native acl strength even after 2 – type III has lower mechanical strength maximum cellular activity and remodelling (weeks 4 -10) - mechanical weakening graft (much weaker than native) – Lowest mechanical strength at 6-8 weeks post-op Slow regain of strength – at 12 months mechanical strength is 50-60% of intact acl Graft loading: some authors suggest early loading should be avoid but tensile strength of patellar and HT ACLR decrease in stress-deprived as opposed to partially loaded early loading relies of stability of fixation of graft as adequate healing into tunnels hasn’t happened & too much loading can be deleterious for biological ingrowth and stability for required for healing
- The strength of all the available grafts is superior to that of the native ACL. All these tests were performed on the unimplanted graft, and therefore the subsequent weakening that takes place in the graft after implantation and during healing should be taken into consideration (From Zen et al. 2017 - Autogenous Hamstring-Bone Graft Preparation for Anterior Cruciate Ligament Reconstruction)
- Strongest: Wk 1 Weakest: ~Wk 6-8 – revascularisation, cellular infiltration, loss of regular collagen oritentation and crimp pattern and the phases of collagen characteristisc (density, magnitude, size changes) Tendon healing in bone tunnel is influed by mechanical stress – it has been shown that the differentiation of mesenchymal stem cells is directly influend by pressure and tenson. Maximum cellular activity and remodelling (weeks 4 -10) - mechanical weakening graft (much weaker than native) Increase in type III collagen – mechanical strength of graft is not restored to native acl strength even after 2 – type III has lower mechanical strength maximum cellular activity and remodelling (weeks 4 -10) - mechanical weakening graft (much weaker than native) – Lowest mechanical strength at 6-8 weeks post-op Slow regain of strength – at 12 months mechanical strength is 50-60% of intact acl Graft loading: some authors suggest early loading should be avoid but tensile strength of patellar and HT ACLR decrease in stress-deprived as opposed to partially loaded early loading relies of stability of fixation of graft as adequate healing into tunnels hasn’t happened & too much loading can be deleterious for biological ingrowth and stability for required for healing
- Scheffler et al. 2008 - Graft remodelling and ligamentization after cruciate ligament reconstruction.
- Human – periphery, sensitivity of staining techniques, rehabilitative programmes, surgical techniques, second-look or post mortem, endo-button or interference screws (tunnel widening)