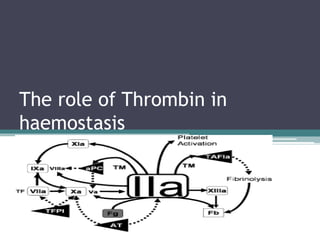
The role of thrombin in coagulation
- 1. The role of Thrombin in haemostasis
- 2. Alexander Schmidt • Described that the conversion of fibrinogen to fibrin is the result of an enzymatic process • Named the hypothetical enzyme “thrombin” and it’s precursor “pre-thrombin”
- 3. In this presentation: • Haemostasis –a brief overview • Basic biochemistry • The moves • The dance
- 4. A brief overview of a complex system Essential haematology 5th edition
- 6. Prothrombin • Zymogen ▫ Inactive precursor enzyme ▫ 72 kDa single chain protein mainly produced in liver • Serine protease ▫ Enzyme that depends on the presence of serine in it’s active center to hidrolyse peptide bonds ▫ Active site Carboxyl terminal Catalytic triad of serine, aspartic acid and histidine • Vitamin K dependant • t1/2 = 60 hours
- 7. Thrombin: ▫ 36 kDa molecule formed when prothrombin is cleaved by prothrombinase complex ▫ Sodium activated allosteric enzyme ▫ Inactivated by antithrombin and removed from the circulation by the liver
- 8. Thrombin: Structure • Two polypeptide chains • B-chain ▫ Active site ▫ Functional epitopes • Sodium binds between the two chains • Exosite I • Exosite II Di Cera E. Thrombin as proccoagulant and anticoagulant.J Thromb Hemost 2007;5(Suppl. 1):196-202
- 9. Thrombin structure: Exosite I • Integrity required by fibrinogen for docking onto the thrombin surface • Contains: ▫ Numerous hydrophobic patches and charged residues on it’s surface • Provides electrostatic steering to fibrinogen on it’s approach to the active site ▫ Fast rate of complex formation • Provides the locale for docking to TMDi Cera E. Thrombin as proccoagulant and anticoagulant.J Thromb Hemost 2007;5(Suppl. 1):196-202
- 10. Thrombin structure: Exosite II • Locale for interaction with polyanionic ligands ▫ Heparin ▫ Glucoseaminoglycans Di Cera E. Thrombin as proccoagulant and anticoagulant.J Thromb Hemost 2007;5(Suppl. 1):196-202
- 12. Essential haematology 5th edition
- 13. Thrombin generation in vivo • Network of amplification and negative feedback loops ▫ Localised and limited production • Two waves of different magnitude ▫ Initiation phase Picomolar [] Preparation for second larger burst ▫ Amplification phase Micromolar [] • Three procoagulant enzyme complexes: ▫ Consisting of: Protease, cofactor, PL and calcium Tissue factor complex, tenase, prothrombinase
- 14. Coagulation in vivo: Initiation phase • Initial generation of thrombin • TF factor bearing cells: ▫ Mononuclear cells ▫ Endothelial cells ▫ Stromal fibroblasts • TF bearing cell surface: ▫ Only displays TF once activated ▫ Factors V, IX, X activated • Small amount of thrombin produced
- 15. Coagulation in vivo: Amplification phase • The first thrombin generated prepares for the thrombin burst that occurs on the surface of activated platelets
- 16. Coagulation in vivo: Propagation phase • Propagation occurs on the surface of activated platelets, leading to the formation of large amounts of thrombin
- 17. Clot stabilization • Fibrinogen ▫ Produced in the liver ▫ T1/2 of 4/7 ▫ Dimer consisting of three paired chains • Fibrin monomer ▫ Formed after T cleaves fibtinopeptides from a and b chains • Fibrin polymer ▫ Self assembly of monomers, end to end association of D domains • Crosslinked fibrin ▫ Covalent bonds between chains ▫ Resistant to fibrinolysis Essential haematology 5th edition
- 18. Clot stabilization Factor XIII • Converts loose fibrin into a firm organized structure • Transglutaminase ▫ Catalyses crosslinking through the formation of gluatmine-lysine covalent bonds between chains • Ciculates as a hetrotetramer • Activation ▫ Requires T and Ca++ ▫ Two steps Limited proteolysis by T Dissociation of B subunits in the presence of Ca++
- 20. Cells responding to thrombin stimulation • Cellular effects mediated by protease activated receptors (PARs) • PAR ▫ G-protein ▫ Highly expressed in platelets ▫ Also found EC, Monocytes, fibroblasts, T-lymphocytes, neurons, smooth muscle cells ▫ 1,3,4 activated by thrombin
- 21. Cells responding to thrombin stimulation • PAR ▫ Carry their own ligands ▫ Ligands remain tethered to receptor after the cleaving of the receptor by thrombin ▫ Activation irreversible ▫ Duration of activity limited by rapid internalization of receptor ▫ Used once and then discarded
- 22. Essential haematology 5th edition
- 23. Thrombin -effects on endothelial cells • Signals EC and vascular smooth muscle cells to: ▫ Control vascular resistance • Amplifies the inflammatory response by modulating transendothelial movement of neutrophils and plasma proteins
- 24. Essential haematology 5th edition
- 25. Thrombin -effects on platelets • Most potent platelet activator • Shape change • Aggregation b.m.o integrin binding to vWF and fibrinogen • Induces synthesis and sectretion of: ▫ ADP ▫ 5-HT ▫ Thromboxane A2 ▫ EDPGF
- 26. Thrombin -effects on platelets • PAR-1 ▫ Increased intracellular [Ca2+] ▫ Upregulation GP IIb/IIIa ▫ Mobilization of P-selectin and CD40-L to platelet surface increased binding of platelets to fibrinogen and an increase in cross linking • PAR-4 ▫ Modulate and stabilize interplatelet binding
- 27. Thrombin and physiological limitation of blood coagulation • Important that the effect of thrombin is limited to the site of injury ▫ By –direct inhibition indirect inhibition
- 28. Thrombin and physiological limitation of blood coagulation: Tissue factor pathway inhibitor • Synthesized in endothelial cells • Present: ▫ plasma ▫ platelets • Accumulates at the site of injury –platelet activation • Inhibits: ▫ Xa ▫ VIIa via formation of the quaternary complex ▫ TF
- 29. Thrombin and physiological limitation of blood coagulation Antithrombin • Serpin • Direct inhibitor of thrombin • Inactivates serine proteases by combining with them by peptide bonding –forming high mw stable complexes • Stable complexes rapidly removed from the circulation in the liver
- 30. Thrombin and physiological limitation of blood coagulation Antithrombin • Neutralization of thrombin enhanced when T is bound to TM • Antithrombin activity enhanced by binding to heparans
- 31. Thrombin and physiological limitation of blood coagulation The protein C pathway
- 32. Thrombin and physiological limitation of blood coagulation The Protein C Anticoagulation Pathway • Complex interaction of multiple proteins on cell surfaces • Major players ▫ Thrombin, thrombomodulin, proteins C & S
- 33. Thrombin and physiological limitation of blood coagulation • Thrombomodulin ▫ Presented by EC ▫ Binds to Thrombin at exosite I Inhibits fibrinogen binding at exosite I Slows fibrin generation Decreases the level of free thrombin available for procoagulant activity. Contributes to the activation of Prot C Promotes stereo chemical association between the active site of thrombin with the cleavage site on proteinC
- 34. Thrombin and physiological limitation of blood coagulation • APC ▫ Inactivates Factor Va and VIIIa ▫ Promotes fibrinolysis by complexing and inactivating PAI-1
- 36. Fibrinolysis Thrombin • Inhibits fibrinolysis by activating TAFI via the T, TM complex ▫ TAFI acts by removing the terminal lysine residues of fibrin that normally facilitate the binding action of Plasmin and tPA • Promotes fibrinolysis via the protein C pathway
- 37. Just a reminder
- 38. Dancing • Na+ activated allosteric enzyme • Slow Fast
- 39. Dancing: The role of Na+ • Activity • Physiological [Na+] not sufficient for saturation • System optimally poised for allosteric regulation • Na+ binding required for ▫ Optimal cleaving of fibrinogen ▫ Activation of: Factor 5, 8, 9 necessary for the explosive generation of thrombin
- 40. Dancing: The role of Na+ • Promotes the prothrombotic and signalling functions by enhacing cleavage of ▫ PAR1, PAR3 and PAR4 • destabilized Na+ binding anticoagulant effect
- 44. Sources • Essential Haematology • Mechanisms in haematology • Di Cera E. Thrombin as procoacgulant and anticoagulant. Journal of Thrombosis and Haemostasis, 5(Suppl. 1):196-202 • Di Cera E. Thrombin; Molec Aspects of Medicine 2008;29(4):203-254 • Huntingdon. Slow thrombin is zymogen-like. Journal of Thrombosis and haemostasis 2009; 7(S1STATE): 159-164
