Dendrimers.pptx
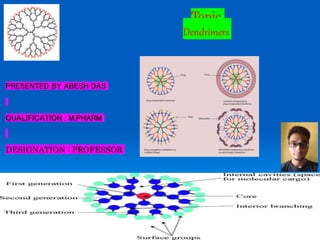
- 1. PRESENTED BY ABESH DAS QUALIFICATION : M.PHARM DESIGNATION : PROFESSOR Topic Dendrimers
- 2. CONTENTS • Introduction • Structure and chemistry • Classification of Dendrimer • Approaches for dendrimer • Convergent Method • Divergent Method • Application of dendrimer in biomedical field • Drugs used with PAMAM Dendrimer
- 3. Dendrimers are nano-sized, radially symmetric molecules with well-defined, homogeneous, and monodisperse structure consisting of tree-like arms or branches. The second group called synthesized macromolecules ‘arborols’ means, in Latin, ‘trees’. Dendrimers might also be called ‘cascade molecules’ Dendrimers are nearly monodisperse macromolecules that contain symmetric branching units built around a small molecule or a linear polymer core ‘Dendrimer’ is only an architectural motif and not a compound. Dendrimers have gained a broad range of applications in supra molecular chemistry, particularly in host-guest reactions and self-assembly processes. Fig 1 : Dendrimer
- 4. Dendrimers are highly defined artificial macromolecules, which are characterized by a combination of a high number of functional groups and a compact molecular structure. Dendritic macromolecules tend to linearly increase in diameter and adopt a more globular shape with increasing dendrimer generation. Therefore, dendrimers have become an ideal delivery vehicle candidate for explicit study of the effects of polymer size, charge, and composition on biologically relevant properties such as lipid bilayer interactions, cytotoxicity, internalization, blood plasma retention time, biodistribution, and filtration. Dendrimer Molecule Fig 2 : Dendrimer
- 5. The structure of dendrimer molecules begins with a central atom or group of atoms labeled as the core. From this central structure, the branches of other atoms called ‘dendrons’ grow through a variety of chemical reactions. There continues to be a debate about the exact structure of dendrimers, in particular whether they are fully extended with maximum density at the surface or whether the end-groups fold back into a densely packed interior. Dendrimers can be prepared with a level of control not attainable with most linear polymers, leading to nearly monodisperse, globular macromolecules with a large number of peripheral groups Fig 3 : Dendrimer Architecture
- 6. CLASSIFICATION OF DENDRIMERS ON THE BASIS OF STRUCTURE Simple Dendrimers. These types of dendrimers consist of simple monomeric units which are based upon symmetrical substitution of benzene tricarboxylic acid ester. They have 4, 10, 22, and 46 benzene rings linked symmetrically and molecular diameters of 45Å. Crystalline Dendrimers. These types of dendrimers are formed by mesogenic monomers which are produced by the functionalization of carbosylane. Chiral Dendrimers. In these types of dendrimers, the chirality depends on the building of 4 constitutionally different but chemically similar branches to a chiral core, for example, chiral dendrimers obtained from pentaerythritol Micellar Dendrimers. These types of dendrimers are fully aromatic, water-soluble hyperbranched polypropylene dendrimers generating a cluster of aromatic polymeric chain that is capable to create a milieu that resembles some micellar structures which results in complex with small organic molecules in water
- 7. Hybrid Dendrimers. These dendrimers are formed by the changes in the functionalization of peripheral amines of zero generation polyethyleneimine which results in the formation of structural diverse columnar and cubic-like organized structures which were significantly transformed to produce dendritic structures, for example, hybrid dendritic linearpolymers Amphiphilic Dendrimers. Amphiphilic dendrimers aremainly prepared by the segregation of the two sides of the chain with one having electron-withdrawing and the other part electron-donating, for example, superfect, hydraamphiphiles,and bolaamphiphiles Metallo dendrimers. Metallo dendrimers are formed by acomplex formation method which takes place either at the peripheral surface or in the interior of the molecule. The dendrimers formed by this method were found to possess both electrochemical and luminescence properties, for example,ruthenium bipyridine Multilingual Dendrimers. VivaGel is a commercially available multilingual dendrimer. This contains multiplecopies of a specific group of functions on the surface Multiple Antigen Peptide Dendrimers. Multiple antigenpeptide (MAP) dendrimers have a dendron-like structure formed using the poylysine skelton. Lysine helps in the conjugationof the alkyloamine side chain which is a monomer for the various branching units. These types of dendrimers were formed and found to have numerous biological applications such as in vaccine formation and for diagnostipurposes
- 9. Fig 4: Convergent Method
- 10. Fig 5 : Divergent Method
- 11. APPLICATION OF DENDRIMERS IN BIOMEDICAL FIELD • Perhaps to improve pharmacokinetic properties of drugs for cancer the most promising potential of dendrimers is in their possibility to perform controlled and specified drug delivery, which regards the topic of nanomedicine. • Drugs conjugated with polymers are characterized by lengthened half-life and antigenicity. Unique pathophysiological traits of tumors such as extensive angiogenesis resulting in hypervascularization, the increased permeability of tumor vasculature, and limited lymphatic drainage enable passive targeting, and as a result, selective accumulation of macromolecules in tumor tissue. • This phenomenon is known as ‘enhanced permeation and retention Anticancer Drugs Fig 6: Dendrimers as drug delivery agents for the treatment of cancer
- 12. Transdermal Drug Delivery : Clinical use of NSAIDs is limited due to adverse reactions such as GI side effects and renal side effects when given orally. Transdermal delivery suffers poor rates of transcutaneous delivery due to barrier function of the skin. Dendrimers have found applications in transdermal drug delivery systems. Generally, in bioactive drugs having hydrophobic moieties in their structure and low water solubility, dendrimers are a good choice in the field of efficient delivery system Dendritic sensors : Dendrimers, although are single molecules, can contain high numbers of functional groups on their surfaces. This makes them striking for applications where the covalent connection or close proximity of a high number of species is important. Dendrimers as magnetic resonance : imaging contrast agents Dendrimer- based metal chelates act as magnetic resonance imaging contrast agents. Dendrimers are extremely appropriate and used as image contrast media because of their properties
- 13. Gene Delivery • The ability to deliver pieces of DNA to the required parts of a cell includes many challenges. Current research is being performed to find ways to use dendrimers to traffic genes into cells without damaging or deactivating the DNA. • To maintain the activity of DNA during dehydration, the dendrimer/ DNA complexes were encapsulated in a water soluble polymer and then deposited on or sandwiched in functional polymer films with a fast degradation rate to mediate gene transfection. • Based on this method, PAMAM dendrimer/DNA complexes were used to encapsulate functional biodegradable polymer films for substrate-mediated gene delivery. Research has shown that the fast- degrading functional polymer has great potential for localized transfection Fig 7: Gene Delivery
- 14. Dendrimers used for enhancing solubility PAMAM dendrimers are expected to have potential applications in enhancing solubility for drug delivery systems. Dendrimers have hydrophilic exteriors and interiors, which are responsible for its unimolecular micelle nature. Dendrimer-based carriers offer the opportunity to enhance the oral bioavailability of problematic drugs. Thus, dendrimer nano carriers offer the potential to enhance the bioavailability of drugs that are poorly soluble and/or substrates for efflux transporters Figure 8: Poly (propylene amine) dendrimer, containing 32 dansyl units at its periphery
- 15. DENDRIMERS IN ANTINEOPLASTIC THERAPY An increasingly addressed strategy is the use of active molecule carriers, belonging to nanomaterial technology, which aims to improve the target selectivity of drugs in neoplastic cells. Due to their physicochemical properties, nanoparticles pass biobarriers more easily. The carrier capacity of dendrimers offers an advantage and constitutes an important strategy in cancer therapy, dendrimers having the role of useful ligands in transporting the drug molecule to tumor tissue through various biological compartments, while maximizing the pharmacodynamic activity to the targeted site. Drug release from the dendrimer complex is controlled by different mechanisms: degradable spacers at specific sites, and the numeric variation of terminal groups. The structure and specific functionality of dendrimer surfaces, correlated with special properties of these materials, such as high hydrosolubility, allows the encapsulation/conjugation of several entities, either in the central structure or on the surface, which makes dendrimers ideal carriers for various
- 16. Doxorubicin (DOX)—used in lung cancer and brain tumors, and has been conjugated to PAMAM dendrimers of the fifth generation (G4). The conjugation was performed via acylhydrazone bonds on the dendrimer surface, with the advantage of increasing the therapeutic efficiency and specificity of action in the lung neoplasm, by directing the pH-controlled DOX-PEG-PAMAM dendrimer Paclitaxel (PTX)—this was conjugated with a PAMAM G4 dendrimer through a glycine–phenylalanine–leucine–glycine peptide linker for the indication of breast cancer. The dendrimeric conjugate increases specificity and cytotoxicity compared to the PTX molecule alone Docetaxel (DTX)—the surface modification of PAMAM dendrimers is a strategy employed to lower systemic toxicity and to increase tumor targeting . An example of this improvement in efficacy is the case of DTX, indicated in breast cancer:
- 17. Imatinib (IMT)—the PAMAM dendrimer complex of IMT is formed via electrostatic interactions, as in the case of other hydrophobic acid molecules, while also non-polar groups of dendrimer ramifications can act as micelles, increasing solubility.Thus, a PEGylated PAMAM G5 dendrimer conjugate of IMT, a drug used in lung cancer, showed increased water solubility, and improved targeting and release in neoplastic cells 5-fluorouracil (5-FU)—used in gastric neoplasm as an aptamer conjugate with PAMAM-PEG, it is specifically targeted to MKN45-type neoplastic cells, with increased uptake by neoplastic cells. Sunitinib—used in renal neoplasm, is conjugated with an NH2-PAMAM- G3 dendrimer through the platinum (II)-based binding system, obtaining the targeting of the active molecule at the neoplastic renal tissue
- 18. The combination of high surface area and high solubility makes dendrimers useful as nanoscale catalysts. Dendrimers have a multifunctional surface and all catalytic sites are always exposed towards the reaction mixture. They can be recovered from the reaction mixture by easy ultra filtration methods. Fig 9: Dendrimer as catalyst Dendritic shells can be used to create a microenvironment favorable for catalysis or provide shielding for functional groups at the dendritic core. the metal sites in these polymeric catalysts should be easily accessible for substrate molecules and reagents, and therefore exhibit characteristics- fast kinetics and solubility
- 19. Dendritic shells can be used to create a microenvironment favorable for catalysis or provide shielding for functional groups at the dendritic core. the metal sites in these well- deûned polymeric catalysts should be easily accessible for substrate molecules and reagents, and therefore exhibit characteristics- fast kinetics and solubility 1. Metallodendritic catalysts 2. Catalysis with phosphine-based dendrimers 3. Catalysis with (metallo)dendrimers containing chiral ligands 4. Non-metal containing dendrimers Fig 10 : Internal Structure of Dendritic shell
- 20. 1. Dendrimers: Synthetic Strategies, Properties and Applications urvashi singh, mohammad maqbool dar and athar adil hashmi*; doi.org/10.13005/ojc/300301 2. Review Article Dendrimers: A New Race of Pharmaceutical Nanocarriers Pooja Mittal; doi.org/10.1155/2021/8844030 3. Applications and Limitations of Dendrimers in Biomedicine; Adriana Aurelia Chis, Carmen Dobrea *, Claudiu Morgovan *; DOI: 10.3390/molecules25173982 4. www.ncbi.nlm.nih.gov/pmc/articles/PMC4074873/
