IB Chemistry on Electromagnetic Spectrum and Wave particle duality
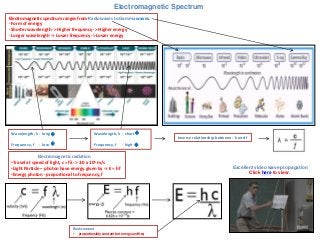
- 1. Electromagnetic Spectrum Electromagnetic spectrum ranges from Radiowaves to Gamma waves. - Form of energy - Shorter wavelength -> Higher frequency -> Higher energy - Longer wavelength -> Lower frequency -> Lower energy Wavelength, λ - long Frequency, f - low Wavelength, λ - short Frequency, f Inverse relationship between- λ and f - high Electromagnetic radiation •Travel at speed of light, c = fλ -> 3.0 x 108 m/s •Light Particle – photon have energy given by -> E = hf •Energy photon - proportional to frequency, f Plank constant • proportionality constant bet energy and freq Excellent video wave propagation Click here to view.
- 2. Electromagnetic Wave propagation. Electromagnetic radiation • • • Moving charges/particles through space Oscillating wave like property of electric and magnetic field Electric and magnetic field oscillate perpendicular to each other and perpendicular to direction of wave propagation. Electromagnetic radiation Electromagnetic wave propagation Click here to view video Violet λ = 410nm f = c/λ = 3 x 108/410 x 10-9 = 7.31 x 1014 Hz E = hf = 6.626 x 10-34 x 7.31 x 1014 = 4.84 x 10-19 J Wave Wave – wavelength and frequency - travel at speed of light Red λ = 700nm f = c/λ = 3 x 108/700 x 10-9 = 4.28 x 1014 Hz E = hf = 6.626 x 10-34 x 4.28 x 1014 = 2.83 x 10-19 J
- 3. Electromagnetic Wave propagation. • • • Electromagnetic radiation Moving charges/particles through space Oscillating wave like property of electric and magnetic field Electric and magnetic field oscillate perpendicular to each other and perpendicular to direction of wave propagation. Electromagnetic radiation Electromagnetic wave propagation Click here to view video Wave Wave – wavelength and frequency - travel at speed of light Simulation on Electromagnetic Propagation Click here to view simulation Click here to view simulation Click here to view simulation
- 4. Electromagnetic Wave Violet Red λ = 410nm λ = 700nm f = c/λ = 3 x 108/410 x 10-9 = 7.31 x 1014 Hz f = c/λ = 3 x 108/700 x 10-9 = 4.28 x 1014 Hz Wavelength – Distance bet two point with same phase, bet crest/troughs – unit nm Frequency – Number of cycle/repeat per unit time (cycles in 1 second) – unit Hz Which light wave have higher frequency, if both have same speed reaching Y same time? Click here on excellent video red /violet wave Violet light X Y Light travel same speed Red flippers – long λ - less frequent Violet shoes – short λ - more frequent Red light Click here to view video energy photon
- 5. Quantum Weirdness – Is Light/electron a Wave or Particle? Pass BIG particles through a 2 slit Pass waves through a 2 slit 2 bands Particle gun Interference patterns of many bands Slit Quantum particles – small particles, like electrons – Is light/electron a Wave or Particle? Particles Pass electron through a 2 slit Particles /photon •single slit – single band •double slit – double band Does electron behave as Click here to view simulation Interference patterns of many bands Waves Wave •Crest/trough appearing •Double slit – interference behind screen – bright/dark bands Conclusion Single electron behave like particles but travel like a wave Waves Principle of wave–particle duality.
- 6. Quantum Weirdness – Is Light/electron a Wave or Particle? Pass electron through a 2 slit Particles Particles/photon •single slit – single band •double slit – double band Does electron behave as Click here to view simulation Interference patterns of many bands Waves Pass electron through a 2 slit Particles Waves •Crest/trough appearing •Double slit – interference behind screen – bright/dark bands Particles/photon •single slit – single band •double slit – double band Does electron behave as Measuring /observing it - which slit it passes through Waves Waves •Crest/trough appearing •Double slit – interference behind screen – bright/dark bands Conclusion Single electron behave like particle but travel like a wave Try to measure it. Principle of wave–particle duality. Wave of possibility.
- 7. Is light a Wave or Particle? Photoelectric effect Light hits metal Electrons are release (photoelectrons) Current measured Click here to view video Light – Shows photoelectric effect Shine light to metal Current measured Is Light a Wave or Particle? Is Light a Wave or Particle? Wave Theory Particle Theory VS Click here to view video Wave Theory •Light energy arrives continuously, regardless of freq •Energy proportional to amplitude •Intensity light – depends on amplitude •Low intensity – low amplitude •High intensity – high amplitude Particle Theory •Light energy comes in tiny packets -photons •Amt energy directly proportional to frequency, E = hf •Red light – low freq – low energy •Blue light – high freq – high energy
- 8. Is light a Wave or Particle? Light – Shows photoelectric effect Shine light to metal Current measured Is Light a Particle or Wave? Is Light a Wave or Particle? Wave Theory Particle Theory VS Click here to view video Wave Theory •Light energy arrives continuously, regardless of freq •Energy proportional to amplitude •Intensity light – depends on amplitude •Low intensity – low amplitude •High intensity – high amplitude Particle Theory •Light energy comes in tiny packets -photons •Amt energy directly proportional to frequency, E = hf •Red light – low freq – low energy •Blue light – high freq – high energy Particle Theory Wave Theory Colour/Frequency light Light energy arrives continuously, regardless of frequency Intensity light Low intensity – Low amplitude High intensity –High amplitude Colour/Frequency light Red light Low frequency – Low energy Colour/Frequency light Blue light High frequency – High energy
- 9. Is light a Wave or Particle? Is Light a Wave or Particle? Wave Theory Particle Theory VS Click here to view video Colour/Frequency light Colour/Frequency light Intensity light If light is Wave • Colour/Intensity of light NOT important ! • Regardless of colour/freq/intensity Colour/Frequency light If light is Particle • Colour/Frequency of light is important ! Shine RED light Shine BLUE light Shine light LONG enough Red light Low freq – Low energy Blue light High freq – High energy No electrons released Electrons are released Are electrons released? No electrons released Result!
- 10. Is light a Wave or Particle? Is Light a Wave or Particle? Stream of light Stream of photon Fixed amt of energy – E = hf Is Light a Wave or Particle? Click here to view video Is Light a Wave or Particle? Video Is Light a Wave or Particle? Click here to view video Click here wave and particle Click here wave and particle Click here wave and particle
