Turning Tides in Targeted Therapy for Early-Stage EGFR-Mutated NSCLC: Latest Data and Practical Guidance for Thoracic Surgeons and the Multidisciplinary Team on the Emerging Role of EGFR-Targeted Therapy in Resectable Lung Cancer
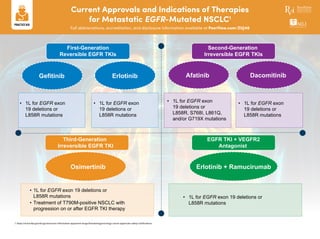
- 1. First-Generation Reversible EGFR TKIs Second-Generation Irreversible EGFR TKIs Third-Generation Irreversible EGFR TKI EGFR TKI + VEGFR2 Antagonist Gefitinib AfatinibErlotinib Dacomitinib Erlotinib + RamucirumabOsimertinib • 1L for EGFR exon 19 deletions or L858R mutations • 1L for EGFR exon 19 deletions or L858R mutations • 1L for EGFR exon 19 deletions or L858R, S768I, L861Q, and/or G719X mutations • 1L for EGFR exon 19 deletions or L858R mutations • 1L for EGFR exon 19 deletions or L858R mutations • Treatment of T790M-positive NSCLC with progression on or after EGFR TKI therapy • 1L for EGFR exon 19 deletions or L858R mutations Current Approvals and Indications of Therapies for Metastatic EGFR-Mutated NSCLC1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZUJ40 1. https://www.fda.gov/drugs/resources-information-approved-drugs/hematologyoncology-cancer-approvals-safety-notifications.
- 2. Addressing Unmet Needs and Improving Outcomes in Early-Stage NSCLC: The Emerging Role of EGFR-Targeted Therapy Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZUJ40 NSCLC 5-Year Overall Survival About 30% of patients with NSCLC present with resectable disease at diagnosis1-3 Surgery with curative intent is the primary treatment for these patients4 Adjuvant cisplatin-based chemotherapy is recommended for patients with resectable stage II-IIIA NSCLC and select patients with stage IB disease5 Adjuvant impact depends on stage, and there is much room for improvement6 Rates of disease recurrence following surgery remain high across disease stages, regardless of postoperative chemotherapy use7 Stage I (IB) Stage II Stage III CALGB JBR.10 ALPI IALT ANITA LACE 34 57 9 30 7 63 36 60 4 33 65 2 36 60 33 64 3 32 45 23 53 32 15 53 43 4 43 39 18 51 39 10 76 19 5 55 30 15 51 26 61 26 13 234 Death (%) with/without chemotherapy Survival without chemotherapy Survival due to chemotherapy Death due to chemotherapy
- 3. Addressing Unmet Needs and Improving Outcomes in Early-Stage NSCLC: The Emerging Role of EGFR-Targeted Therapy Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZUJ40 ADAURA: Phase 3 Double-Blind Studya DFS According to Investigator Assessment10 Stage II to IIIA Disease Stage IB to IIIA Disease Adjuvant osimertinib is the first targeted agent to show a statistically significant and clinically meaningful improvement in disease-free survival (DFS) in patients with stage IB/II/IIIA EGFRmut NSCLC10 EGFR TKIs are standard of care for patients with EGFRmut advanced NSCLC Previous studies have suggested there may be a role for EGFR TKIs in the resected setting, but results have been inconclusive8,9 ADAURA: Based on efficacy and safety data, adjuvant osimertinib represents a big opportunity to improve outcomes in more patients with early-stage NSCLC10 At 24 months, 90% of the patients with stage II to IIIA disease in the osimertinib group (95% CI, 84-93) and 44% of those in the placebo group (95% CI, 37-51) were alive and disease free (overall HR for disease recurrence or death, 0.17; 99.06% CI, 0.11-0.26; P < .001) → This HR = 83% reduction in risk of disease recurrence or death In the overall population, 89% of the patients in the osimertinib group (95% CI, 85-92) and 52% of those in the placebo group (95% CI, 46-58) were alive and disease free at 24 months (overall HR for disease recurrence or death, 0.20; 99.12% CI, 0.14-0.30; P < .001) → This HR = 80% reduction in risk of disease recurrence or death R • Primary endpoint: DFS, by investigator assessment, in stage II/IIIA patients; designed for superiority under the assumed DFS HR of 0.70 • Secondary endpoints: DFS in the overall population;f DFS at 2, 3, 4, and 5 years; OS; safety; health-related quality of life • Following IDMC recommendation, the study was unblinded early because of efficacy; here, an unplanned interim analysis is reported • At the time of unblinding, the study had completed enrollment and all patients were followed up for at least 1 year Patients with completely resected stageb IB, II, IIIA NSCLC, with or without adjuvant chemoc Key inclusion criteria: • ≥18 y (Japan/Taiwan, ≥20 y) • WHO performance status 0/1 • Confirmed primary nonsquamous NSCLC • Ex19del/L858Rd • Brain imaging, if not completed preoperatively • Complete resection with negative marginse • Maximum interval between surgery and randomization: 10 wk without adjuvant chemo; 26 wk with adjuvant chemo Placebo Once daily Osimertinib 80 mg, once daily Stratified by: • Stage (IB vs II vs IIIA) • EGFRmut (ex19del vs L858R) • Race (Asian vs non-Asian) 1:1 Planned treatment duration: 3 y Treatment continues until: • Disease recurrence • Treatment completed • Discontinuation criterion met Follow up: • Until recurrence: week 12 and 24, then every 24 weeks to 5 years, then yearly • After recurrence: every 24 weeks for 5 years, then yearly N = 682 Time Since Randomization, mo ProbabilityofDFS 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 0 6 12 18 24 30 36 42 48 97% 90% 80% 61% 44% 28% 1.0 Osimertinib Placebo 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 0 6 12 18 24 30 36 42 48 Time, mo ProbabilityofDFS 97% 89% 79% 69% 53% 41% Osimertinib Placebo
- 4. Subgroup HR 95% CI Overall (N = 682) Stratified log-rank Unadjusted Cox PH 0.21 0.20 0.16-0.28 0.14-0.29 Sex Male (n = 204) Female (n = 478) 0.21 0.20 0.11-0.38 0.12-0.30 Age <65 y (n = 380) ≥65 y (n = 302) 0.18 0.24 0.10-0.28 0.14-0.38 Smoking status Smoker (n = 194) Nonsmoker (n = 488) 0.14 0.23 0.06-0.27 0.15-0.34 Race Asian (n = 434) Non-Asian (n = 248) 0.22 0.17 0.14-0.33 0.08-0.31 Stage Stage IB (n = 212) Stage II (n = 236) Stage IIIA (n = 234) 0.50 0.17 0.12 0.25-0.96 0.08-0.31 0.07-0.20 EGFRmut Exon 19 deletion (n = 378) L858R (n = 304) 0.12 0.35 0.07-0.20 0.21-0.55 Adjuvant chemo Yes (n = 378) No (n = 304) 0.18 0.23 0.11-0.29 0.13-0.38 0.01 0.1 1 Favors osimertinib Favors placeboHR for DFS (95% CI) Subgroup Analysis of Disease Recurrence or Death According to Investigator Assessment10 Benefit favoring osimertinib with respect to disease-free survival was observed consistently across all predefined subgroups Safety profile was consistent with the established safety profile of osimertinib, with mild EGFR TKI class effects reported • Patients who received osimertinib had fewer local/regional and distant relapses than those who received placebo, with a lower incidence of metastatic disease in those patients with recurrence, including fewer CNS recurrence events • Adjuvant osimertinib demonstrated a clinically meaningful improvement in CNS DFS compared with placebo HR = 0.18 (95% CI, 0.10-0.33; P < .0001) = 82% reduction in risk of CNS disease recurrence or death Consistent improvement in DFS was seen regardless of whether patients received prior adjuvant chemotherapy DFS Per Investigator Assessment With and Without Adjuvant Chemotherapy10 Adverse Events10 CNS DFS According to Investigator Assessment in the Overall Population10 ADAURA → Adjuvant osimertinib represents a highly effective treatment for patients with stage IB/II/IIIA EGFRmut NSCLC after complete resection 0 0.2 0.4 0.6 0.8 1.0 0 6 12 18 24 30 36 42 48 56 ProbabilityofDFS Time, mo 0 0.2 0.4 0.6 0.8 0 6 12 18 24 30 36 42 48 ProbabilityofDFS Time, mo DFS in Patients Who Received Adjuvant Chemo DFS in Patients Who Did Not Receive Adjuvant Chemo Osimertinib Placebo HR = 0.16 (95% CI, 0.10-0.26) Osimertinib Placebo HR = 0.23 (95% CI, 0.13-0.40) 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 0 6 12 18 24 30 36 42 48 ProbabilityofCNSDFS Time, mo 100% 98% 98% 97% 85% 82% Osimertinib (N = 337), N (%) Placebo (N = 343), N (%) Adverse Event Any Grade Grade 1 Grade 2 Grade 3 Any Grade Grade 1 Grade 2 Grade 3 Diarrhea 156 (46) 116 (34) 32 (19) 8 (2) 68 (20) 54 (16) 13 (4) 1 (<1) Paronychia 85 (25) 31 (9) 50 (15) 3 (1) 5 (1) 3 (1) 2 (1) 0 Dry skin 79 (23) 75 (22) 3 (1) 1 (<1) 22 (6) 18 (5) 4 (1) 0 Pruritus 65 (19) 49 (15) 16 (5) 0 30 (9) 28 (8) 2 (1) 0 Cough 62 (18) 43 (13) 19 (6) 0 57 (17) 42 (12) 15 (4) 0 Stomatitis 59 (18) 35 (10) 18 (5) 6 (2) 14 (4) 10 (3) 4 (1) 0 Nasopharyngitis 47 (14) 30 (9) 17 (5) 0 35 (10) 25 (7) 10 (3) 0 Upper respiratory infection 45 (13) 24 (7) 19 (6) 2 (1) 35 (10) 19 (6) 16 (5) 0 Decreased appetite 44 (13) 29 (9) 13 (4) 2 (1) 13 (4) 9 (3) 4 (1) 0 Mouth ulceration 39 (12) 37 (11) 32 (9) 7 (2) 0 8 (2) 6 (2) 2 (1) 0 Dermatitis acneiform 29 (9) 8 (2) 0 16 (5) 12 (3) 4 (1) 0 1.0 a NCT0251106; ADAURA data cutoff: January 17, 2020. b AJCC, 7th edition. c Prior, post, and planned radiotherapy was not allowed. d Centrally confirmed in tissue. e Patients received a CT after resection and within 28 days before treatment.f Stage IB/II/IIIA. 1. Datta D, Lahiri B. Chest. 2003;123:2096-2103. 2. Le Chevalier T. Ann Oncol. 2010;21(suppl 7):vii196-vii198. 3. Cagle PT et al. Arch Pathol Lab Med. 2013;137:1191-1198. 4. Chansky K et al. J Thorac Oncol. 2017;12:1109-1121. 5. Postmus PE et al. Ann Oncol. 2017;28(suppl 4):iv1-iv21. 6. Kris MG et al. J Clin Oncol. 2017;35:2960-2974. 7. Pignon J et al. J Clin Oncol. 2008;26:3552-3559. 8. Wu Y-L et al. J Clin Oncol. 2020;38(suppl 15):9005. 9. Huang Q et al. Chest. 2016;149:1384-1392. 10. Wu Y et al. N Engl J Med. 2020;383:825-835. Addressing Unmet Needs and Improving Outcomes in Early-Stage NSCLC: The Emerging Role of EGFR-Targeted Therapy Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZUJ40
