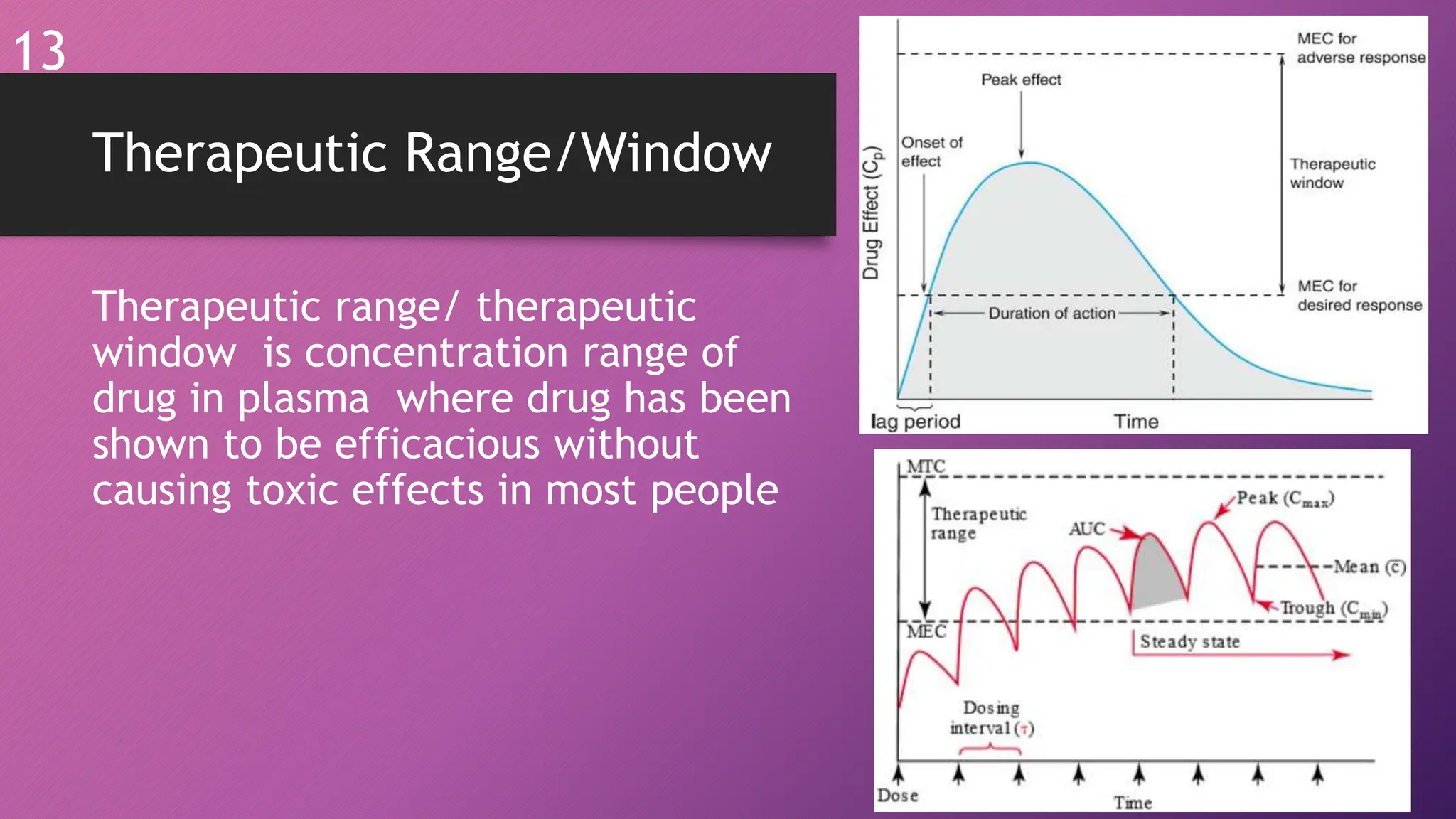
The document discusses therapeutic drug monitoring (TDM), including its definition, basic concepts, and drugs that commonly require monitoring. TDM involves measuring drug levels in blood to ensure doses are effective while preventing toxicity. Key concepts covered include pharmacokinetics, pharmacodynamics, therapeutic ranges, and factors affecting drug levels like metabolism and protein binding. Drugs highlighted as commonly monitored include lithium, phenytoin, digoxin, aminoglycosides, theophylline, tacrolimus, and methotrexate due to their narrow therapeutic windows. Accurate analysis methods and clinical interpretation of results are important for effective TDM.