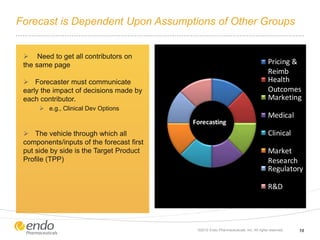
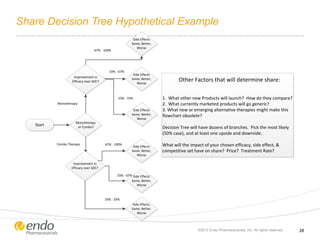
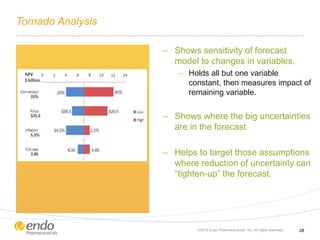
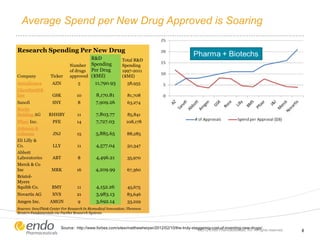
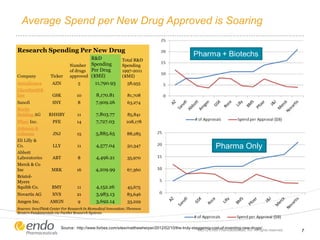
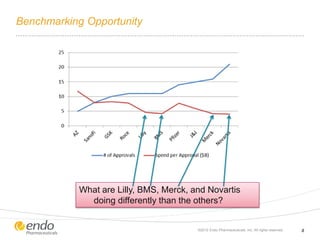
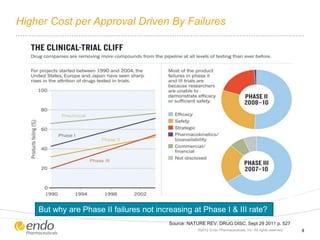
The document discusses risk assessment and management for new product planning in the pharmaceutical industry. It notes that [1] managing risk for new products is important given high development costs and failure rates, [2] forecasts often overestimate commercial potential which can lead to problems, and [3] using a target product profile and decision analysis can help capture development options and quantify risk.



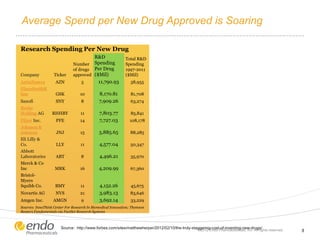
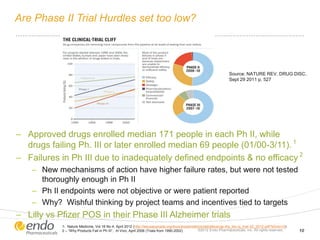
![Concern Over Early Stage New Product Development
– Commercial potential [value] is
being assessed at the beginning of
every development program1
– “Sales estimates” identified as the
parameter management most
wishes to be more reliable in
biotech valuations2
– 80% of products launched in past
decade did not live up to
expectations3
– # of FDA drugs approved per each
$1B of R&D spend halved every 9
years since 1950 4
– $4B-$12B = average R&D spend
for every drug approved (1997 –
2011)5
©2012 Endo Pharmaceuticals, Inc. All rights reserved. 4
1- John LaMattina, former Pfizer VP R&D, LifescienceLeader.com, Dec 2011
2 – Dec 2011 Linked in Survey by Avance (www.avance.ch)
3 – Campbell Alliance, PA Bio, March 29, 2012 “What Decisions Impact Value the Most”
4 – Nature Reviews, Drug Discovery, March 2012 p. 191
5 – Forbes, March 12, 2012 p. 38](https://image.slidesharecdn.com/8c11f0c0-0689-43e7-97ab-793534eb3fa2-160217011823/85/RiskAssessmentCBIFalcon25June2012-4-320.jpg)


![Forecasts and the Scientific Method
“Scientific method refers to a body of techniques
for…acquiring new knowledge, or correcting and integrating
previous knowledge” 1
“[A] basic expectation is to document, archive and share all
data and methodology so they are available for careful
scrutiny by other scientists, giving them the opportunity to
verify results by attempting to reproduce them.” 2
“The goal of a scientific inquiry is to obtain knowledge in the
form of testable explanations that can predict the results of
future experiments.” 3
©2012 Endo Pharmaceuticals, Inc. All rights reserved. 14
1 – Wikipedia http://en.wikipedia.org/wiki/Scientific_method
2 - Ibid
3 - Ibid](https://image.slidesharecdn.com/8c11f0c0-0689-43e7-97ab-793534eb3fa2-160217011823/85/RiskAssessmentCBIFalcon25June2012-14-320.jpg)