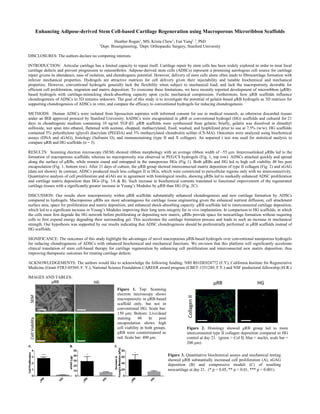
This study investigated using microribbon (μRB) scaffolds to support chondrogenesis of adipose-derived stem cells (ADSCs) for cartilage regeneration. The μRB scaffolds were macroporous and gelatin-based, while conventional hydrogel (HG) scaffolds lacked macroporosity. ADSCs encapsulated in μRB scaffolds attached, spread, and proliferated more than in HG scaffolds. After 3 weeks of culture, ADSCs in μRB scaffolds deposited more interconnected type II collagen and sulfated glycosaminoglycans (sGAG), and the resulting neocartilage had a higher compressive modulus than ADSCs in HG scaffolds. The enhanced chondrogenesis and mechanical properties of