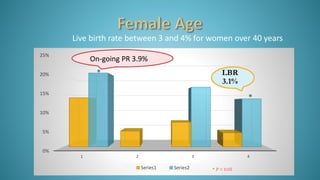
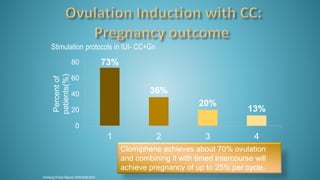
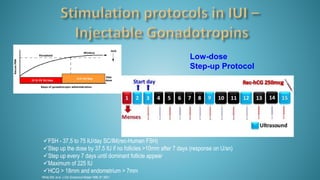
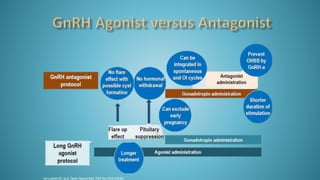
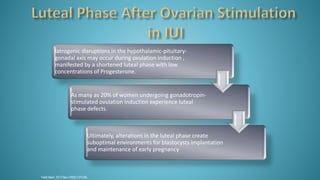
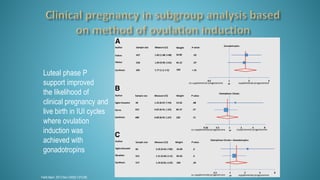
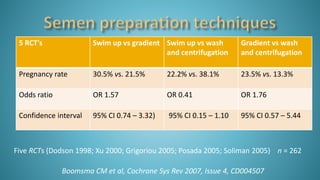
Dr. Vandana Bansal is a senior gynaecologist and obstetrician who specializes in infertility and IVF. She directs the Arpit Test Tube Baby Centre in Prayagraj, India. The document discusses intrauterine insemination (IUI), providing rationales for its use, details on techniques and protocols, success rates based on factors like age and ovarian stimulation methods, and alternatives when IUI is unsuccessful. It summarizes evidence from clinical studies on optimizing IUI outcomes.