Effect of Base Volume on pH in Acid-Base Titration Experiment
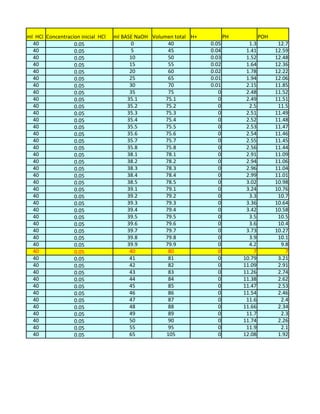
- 1. ml HCl Concentracion inicial HCl ml BASE NaOH Volumen total H+ PH POH 40 0.05 0 40 0.05 1.3 12.7 40 0.05 5 45 0.04 1.41 12.59 40 0.05 10 50 0.03 1.52 12.48 40 0.05 15 55 0.02 1.64 12.36 40 0.05 20 60 0.02 1.78 12.22 40 0.05 25 65 0.01 1.94 12.06 40 0.05 30 70 0.01 2.15 11.85 40 0.05 35 75 0 2.48 11.52 40 0.05 35.1 75.1 0 2.49 11.51 40 0.05 35.2 75.2 0 2.5 11.5 40 0.05 35.3 75.3 0 2.51 11.49 40 0.05 35.4 75.4 0 2.52 11.48 40 0.05 35.5 75.5 0 2.53 11.47 40 0.05 35.6 75.6 0 2.54 11.46 40 0.05 35.7 75.7 0 2.55 11.45 40 0.05 35.8 75.8 0 2.56 11.44 40 0.05 38.1 78.1 0 2.91 11.09 40 0.05 38.2 78.2 0 2.94 11.06 40 0.05 38.3 78.3 0 2.96 11.04 40 0.05 38.4 78.4 0 2.99 11.01 40 0.05 38.5 78.5 0 3.02 10.98 40 0.05 39.1 79.1 0 3.24 10.76 40 0.05 39.2 79.2 0 3.3 10.7 40 0.05 39.3 79.3 0 3.36 10.64 40 0.05 39.4 79.4 0 3.42 10.58 40 0.05 39.5 79.5 0 3.5 10.5 40 0.05 39.6 79.6 0 3.6 10.4 40 0.05 39.7 79.7 0 3.73 10.27 40 0.05 39.8 79.8 0 3.9 10.1 40 0.05 39.9 79.9 0 4.2 9.8 40 0.05 40 80 0 7 7 40 0.05 41 81 0 10.79 3.21 40 0.05 42 82 0 11.09 2.91 40 0.05 43 83 0 11.26 2.74 40 0.05 44 84 0 11.38 2.62 40 0.05 45 85 0 11.47 2.53 40 0.05 46 86 0 11.54 2.46 40 0.05 47 87 0 11.6 2.4 40 0.05 48 88 0 11.66 2.34 40 0.05 49 89 0 11.7 2.3 40 0.05 50 90 0 11.74 2.26 40 0.05 55 95 0 11.9 2.1 40 0.05 65 105 0 12.08 1.92
- 2. 14 13 Concentracion inicial PH 12 HCl 11 10 9 8 7 6 5 4 3 2 1 0 Row 4 Row 8 Row 12 Row 16 Row 20 Row 24 Row 28 Row 32 Row 36 Row 40 Row 44 Row 2 Row 6 Row 10 Row 14 Row 18 Row 22 Row 26 Row 30 Row 34 Row 38 Row 42
- 3. OH- Base exceso 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1.00E-07 6.17E-04 1 1.22E-03 2 1.81E-03 3 2.38E-03 4 2.94E-03 5 3.49E-03 6 4.02E-03 7 4.55E-03 8 5.06E-03 9 5.56E-03 10 7.89E-03 15 1.19E-02 25