Report
Share
Recommended
E2 - Types of Star & HR Diagram

The document describes different types of stars:
1) Red giants are very large, cool stars that all main sequence stars evolve into. Nuclear fusion occurs in red giants, fusing helium into heavier elements.
2) White dwarfs are very small and dense remnants of red giants. They have high temperatures but low luminosities due to their small size.
3) Neutron stars form from massive stars and are very hot and dense, composed mostly of neutrons. Pulsars are rotating, magnetized neutron stars that emit beams of electromagnetic radiation.
Stars - Basic Properties/H- R Diagram

The document discusses properties of stars and methods astronomers use to study them. It describes the composition of the Sun and how its composition is similar to other stars and gas giants. It then explains different types of groups stars can be found in, including constellations, clusters, and binary systems. The document also discusses methods astronomers use to measure stellar distances and properties, including parallax, apparent and absolute magnitude, luminosity, and spectral classification. It introduces the Hertzsprung-Russell diagram and how it relates stellar properties like mass, luminosity, temperature and size.
Types of stars

This document describes different stages and types of stars:
- Main sequence stars like our Sun spend most of their lives fusing hydrogen into helium in their cores.
- Red giants are large, reddish stars that have exhausted hydrogen fusion and begun fusing helium.
- Planetary nebulae form when average-sized stars eject their outer layers after becoming red giants, leaving behind dense, hot cores called white dwarfs.
- Brown dwarfs are failed stars too small to sustain nuclear fusion.
- Variable stars change in brightness over timescales from seconds to years as they evolve.
- Binary stars are two gravitationally bound stars that orbit a common center of mass.
Hertzsprung russel diagram

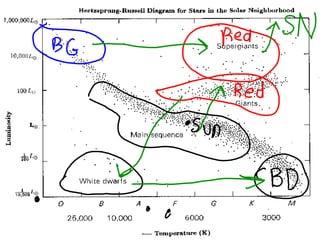
The Hertzsprung-Russell diagram is a chart that shows the relationship between a star's luminosity or absolute magnitude versus its spectral type, stellar classification, and effective temperature. It plots each star on a graph of magnitude against temperature and color. Most stars occupy the main sequence area of the diagram where they are fusing hydrogen. The diagram can also be used to measure the approximate distance between a star cluster and Earth by comparing apparent and absolute magnitudes.
H-R Diagram

This document discusses the Hertzsprung-Russell diagram (H-R diagram), which plots stars' brightness against their surface temperatures. It shows that 90% of stars are average "main sequence" stars that are not too bright, hot, or large. The remaining 10% are "giants", "supergiants", and "white dwarfs". The H-R diagram demonstrates that hotter stars are usually brighter, while cooler stars appear dimmer.
Hertzsprung Russell Diagram

The document summarizes the Hertzsprung–Russell diagram, which plots stars based on their color, temperature, and brightness over their life cycles. It explains that the diagram shows how stars change from blue and hot as O class stars to red and cool as M class stars. The document uses our sun as an example, showing where it is now on the main sequence and where it will be as a red giant and white dwarf. It notes that black dwarfs would not be shown on the diagram since they emit no light or heat.
9 Digestive System

The digestive system begins in the mouth where mechanical and chemical digestion starts breaking down starches. Food then moves to the pharynx and esophagus through peristalsis. In the stomach, food is further broken down chemically and mechanically by HCl and pepsin over 3-6 hours. The partially digested food, now called chyme, spends 3-5 hours in the small intestine where nutrients are absorbed and enzymes from the liver, gallbladder and pancreas aid digestion. Undigested waste spends 18-24 hours in the large intestine where water is absorbed before moving to the rectum and anus to be excreted from the body.
Recommended
E2 - Types of Star & HR Diagram

The document describes different types of stars:
1) Red giants are very large, cool stars that all main sequence stars evolve into. Nuclear fusion occurs in red giants, fusing helium into heavier elements.
2) White dwarfs are very small and dense remnants of red giants. They have high temperatures but low luminosities due to their small size.
3) Neutron stars form from massive stars and are very hot and dense, composed mostly of neutrons. Pulsars are rotating, magnetized neutron stars that emit beams of electromagnetic radiation.
Stars - Basic Properties/H- R Diagram

The document discusses properties of stars and methods astronomers use to study them. It describes the composition of the Sun and how its composition is similar to other stars and gas giants. It then explains different types of groups stars can be found in, including constellations, clusters, and binary systems. The document also discusses methods astronomers use to measure stellar distances and properties, including parallax, apparent and absolute magnitude, luminosity, and spectral classification. It introduces the Hertzsprung-Russell diagram and how it relates stellar properties like mass, luminosity, temperature and size.
Types of stars

This document describes different stages and types of stars:
- Main sequence stars like our Sun spend most of their lives fusing hydrogen into helium in their cores.
- Red giants are large, reddish stars that have exhausted hydrogen fusion and begun fusing helium.
- Planetary nebulae form when average-sized stars eject their outer layers after becoming red giants, leaving behind dense, hot cores called white dwarfs.
- Brown dwarfs are failed stars too small to sustain nuclear fusion.
- Variable stars change in brightness over timescales from seconds to years as they evolve.
- Binary stars are two gravitationally bound stars that orbit a common center of mass.
Hertzsprung russel diagram

The Hertzsprung-Russell diagram is a chart that shows the relationship between a star's luminosity or absolute magnitude versus its spectral type, stellar classification, and effective temperature. It plots each star on a graph of magnitude against temperature and color. Most stars occupy the main sequence area of the diagram where they are fusing hydrogen. The diagram can also be used to measure the approximate distance between a star cluster and Earth by comparing apparent and absolute magnitudes.
H-R Diagram

This document discusses the Hertzsprung-Russell diagram (H-R diagram), which plots stars' brightness against their surface temperatures. It shows that 90% of stars are average "main sequence" stars that are not too bright, hot, or large. The remaining 10% are "giants", "supergiants", and "white dwarfs". The H-R diagram demonstrates that hotter stars are usually brighter, while cooler stars appear dimmer.
Hertzsprung Russell Diagram

The document summarizes the Hertzsprung–Russell diagram, which plots stars based on their color, temperature, and brightness over their life cycles. It explains that the diagram shows how stars change from blue and hot as O class stars to red and cool as M class stars. The document uses our sun as an example, showing where it is now on the main sequence and where it will be as a red giant and white dwarf. It notes that black dwarfs would not be shown on the diagram since they emit no light or heat.
9 Digestive System

The digestive system begins in the mouth where mechanical and chemical digestion starts breaking down starches. Food then moves to the pharynx and esophagus through peristalsis. In the stomach, food is further broken down chemically and mechanically by HCl and pepsin over 3-6 hours. The partially digested food, now called chyme, spends 3-5 hours in the small intestine where nutrients are absorbed and enzymes from the liver, gallbladder and pancreas aid digestion. Undigested waste spends 18-24 hours in the large intestine where water is absorbed before moving to the rectum and anus to be excreted from the body.
8 Momentum

Momentum is defined as the product of an object's mass and velocity (p = m X v), measured in kg*m/sec. The law of conservation of momentum states that during collisions, the total momentum of interacting objects remains the same, relating to Newton's third law. The document provides the definition of momentum, its units, and introduces the law of conservation of momentum.
5 Newtons First Law

Newton's first law of motion, also known as the law of inertia, states that an object at rest stays at rest and an object in motion stays in motion with the same speed and in the same direction unless acted upon by an unbalanced force. This law is demonstrated when a car comes to a sudden stop - an unbelted crash dummy will continue moving forward at the car's original speed and fly through the windshield, coming to rest only upon colliding with another object exerting an outside force like the ground or gravity in a vacuum.
4 Gravity

The document discusses the universal law of gravitation and how it affects falling objects on Earth. Gravity causes all objects to attract each other, with the force depending on their masses and distance between them. When objects fall near Earth's surface, they accelerate at 9.8 m/s2 due to gravity. While gravity causes all objects to fall at the same rate in a vacuum, air resistance opposes smaller, lighter objects so their acceleration decreases until they reach a terminal velocity where air resistance equals the force of gravity.
7 Newtons Third Law

Newton's third law states that for every action, there is an equal and opposite reaction. When a bird flies, the wings push air downwards which causes an equal and opposite reaction force from the air pushing the bird upwards, allowing flight. The action-reaction force pairs between the wings and air make bird flight possible.
6 Newtons Second Law

Newton's second law states that the acceleration of an object is directly proportional to the net force acting on it and inversely proportional to its mass. It can be expressed by the equation F=ma, where F is the net force, m is the mass of the object, and a is the acceleration. The document provides an example problem of calculating the force needed to push a car with a mass of 1000 kg and an acceleration of 0.05 m/sec^2. Using the equation, the force is calculated to be 50 N.
3 Friction

There are three main types of friction: sliding friction which occurs between two solid objects in contact and sliding over each other, rolling friction which occurs between wheels or spherical objects that are rolling over a surface, and fluid friction which occurs when an object moves through a liquid or gas.
2 Forces

Forces can be pushes or pulls on an object. When balanced forces act on an object, there is no movement as the forces add up to zero, but unbalanced forces cause movement as the forces do not add up to zero. Forces are often shown with arrows to indicate their size and direction as a vector quantity.
1 Measuring Motion

This document discusses motion and related concepts like speed, velocity, and acceleration. It defines motion as an object's change in position relative to a reference point over time. Speed is the rate of change of distance over time, while velocity also includes direction. Acceleration is the rate of change of velocity over time. Examples are provided to illustrate calculating speed, velocity, and changes in velocity.
Acids And Bases

Acids turn litmus paper red, contain hydrogen, and donate protons. Bases turn litmus paper blue, contain hydroxide ions, and accept protons. When acids and bases are mixed in a neutralization reaction, they form salts and water by combining hydrogen ions with hydroxide ions. The pH scale measures the hydrogen ion concentration to determine if a solution is acidic, basic, or neutral.
Tips For Predicting 6

This document provides tips for predicting bond type, stating that ionic bonds form between metals and nonmetals or metals and polyatomic ions, covalent bonds form between nonmetals, and metallic bonds form between metals. It then lists examples of bond predictions and practice problems predicting bond type between different elements.
Radioactivity 11

There are three main types of radioactive decay: alpha, beta, and gamma. Alpha decay releases alpha particles, beta decay releases electrons, and gamma decay releases gamma rays. The half-life of a radioactive isotope is the amount of time it takes for half of the radioactive substance to decay, and is used to determine the age of fossils and rocks. In the examples given, calculations are shown for determining the amount of radioactive material remaining after a given number of half-lives based on the original amount and the half-life of the specific isotope.
Notes Acids&Bases 10

The document lists common acids and bases, including their chemical formulas and some common uses. It identifies hydrochloric acid, sulfuric acid, and nitric acid as strong acids commonly used for cleaning, manufacturing, and making explosives. Weaker acids mentioned include carbonic acid, boric acid, phosphoric acid, acetic acid, and citric acid which are used in beverages, personal care, detergents, and food production. Strong bases listed are sodium hydroxide, potassium hydroxide, calcium hydroxide, and magnesium hydroxide used in soaps, cleaners, leather processing, and pharmaceuticals. Weaker bases include ammonium hydroxide and aluminum hydroxide used in household products and
Energy And Rates Of Reactions 9

The document discusses how energy relates to chemical reactions. It explains that exothermic reactions release energy by breaking bonds, while endothermic reactions absorb energy by forming new bonds. All chemical reactions require a minimum amount of energy, called activation energy, to get started. The rates of reactions are affected by temperature, concentration, surface area, catalysts, and inhibitors. Catalysts lower the activation energy needed to start a reaction.
Writing And Balancing Chem Rxn’S 8

This document discusses writing and balancing chemical reactions. It explains that when writing chemical equations, reactants are written on the left and products are written on the right. It also notes that every atom must be balanced according to the law of conservation of mass, which states that mass cannot be created or destroyed in a chemical reaction. The document indicates that if an equation is not balanced, a coefficient can be placed in front of a molecule to balance the number of atoms, but no bonds can be broken in the process of balancing equations.
Covalent Bonding 5

Covalent bonds form when atoms share electrons to achieve a full outer shell, giving the molecule an overall charge of zero. Hydrogen always forms covalent bonds because it has only one electron to share. Examples of covalent bonds include hydrogen gas (H2) and hydrogen chloride (HCl), which can be represented using electron-dot diagrams showing the shared electrons. Molecules are combinations of atoms held together by covalent bonds, such as polyatomic ions.
Chemical Reactions 7

Chemical reactions start with one or more substances (A) and result in different substances (B) through the breaking and rearranging of bonds. Signs that a chemical reaction occurred may include the formation of a gas (bubbles), release of energy (light, sound, heat), a color change, or the production of a precipitate (solid). The document asks if these signs were present for reactions between sulfuric acid and sugar, and nitric acid and a penny.
Electrons And Chemical Bonding 3

Electrons and Chemical Bonding summarizes how atoms bond together to form new substances. Atoms bond by sharing or transferring valence electrons in their outermost shells to achieve full outer shells, which provides stability. Noble gases have full outer shells and do not bond since they are the most stable configuration for atoms.
Periodic Table Presentation 1

The document provides a key for understanding a periodic table. It indicates that solid elements are black, gases are red, and liquids are blue. It also notes that all states of elements can be changed and clear elements are unstable. The periodic table boxes list the element symbol, atomic number representing protons, and atomic mass. Groups have vertical columns of similar chemical properties, while periods have different properties horizontally. Elements are categorized as metals, metalloids, or nonmetals.
Ionic Bonding 4

Ionic bonding occurs when atoms transfer electrons, forming positive and negative ions that bond together. Specifically, metals donate electrons to nonmetals to complete their outer energy levels, with the metal always written first in the compound. The final bonded ionic compound is stable with no overall charge.
Counting Atoms 2a

This document provides instructions on writing chemical formulas and counting atoms in compounds. It explains that capital letters are used for elemental symbols, subscripts indicate the number of atoms, and numbers distributed throughout a compound indicate how many of each atom are present. Examples are given for common compounds and ions to demonstrate how to count the total number of each type of atom.
More Related Content
More from jlevs295
8 Momentum

Momentum is defined as the product of an object's mass and velocity (p = m X v), measured in kg*m/sec. The law of conservation of momentum states that during collisions, the total momentum of interacting objects remains the same, relating to Newton's third law. The document provides the definition of momentum, its units, and introduces the law of conservation of momentum.
5 Newtons First Law

Newton's first law of motion, also known as the law of inertia, states that an object at rest stays at rest and an object in motion stays in motion with the same speed and in the same direction unless acted upon by an unbalanced force. This law is demonstrated when a car comes to a sudden stop - an unbelted crash dummy will continue moving forward at the car's original speed and fly through the windshield, coming to rest only upon colliding with another object exerting an outside force like the ground or gravity in a vacuum.
4 Gravity

The document discusses the universal law of gravitation and how it affects falling objects on Earth. Gravity causes all objects to attract each other, with the force depending on their masses and distance between them. When objects fall near Earth's surface, they accelerate at 9.8 m/s2 due to gravity. While gravity causes all objects to fall at the same rate in a vacuum, air resistance opposes smaller, lighter objects so their acceleration decreases until they reach a terminal velocity where air resistance equals the force of gravity.
7 Newtons Third Law

Newton's third law states that for every action, there is an equal and opposite reaction. When a bird flies, the wings push air downwards which causes an equal and opposite reaction force from the air pushing the bird upwards, allowing flight. The action-reaction force pairs between the wings and air make bird flight possible.
6 Newtons Second Law

Newton's second law states that the acceleration of an object is directly proportional to the net force acting on it and inversely proportional to its mass. It can be expressed by the equation F=ma, where F is the net force, m is the mass of the object, and a is the acceleration. The document provides an example problem of calculating the force needed to push a car with a mass of 1000 kg and an acceleration of 0.05 m/sec^2. Using the equation, the force is calculated to be 50 N.
3 Friction

There are three main types of friction: sliding friction which occurs between two solid objects in contact and sliding over each other, rolling friction which occurs between wheels or spherical objects that are rolling over a surface, and fluid friction which occurs when an object moves through a liquid or gas.
2 Forces

Forces can be pushes or pulls on an object. When balanced forces act on an object, there is no movement as the forces add up to zero, but unbalanced forces cause movement as the forces do not add up to zero. Forces are often shown with arrows to indicate their size and direction as a vector quantity.
1 Measuring Motion

This document discusses motion and related concepts like speed, velocity, and acceleration. It defines motion as an object's change in position relative to a reference point over time. Speed is the rate of change of distance over time, while velocity also includes direction. Acceleration is the rate of change of velocity over time. Examples are provided to illustrate calculating speed, velocity, and changes in velocity.
Acids And Bases

Acids turn litmus paper red, contain hydrogen, and donate protons. Bases turn litmus paper blue, contain hydroxide ions, and accept protons. When acids and bases are mixed in a neutralization reaction, they form salts and water by combining hydrogen ions with hydroxide ions. The pH scale measures the hydrogen ion concentration to determine if a solution is acidic, basic, or neutral.
Tips For Predicting 6

This document provides tips for predicting bond type, stating that ionic bonds form between metals and nonmetals or metals and polyatomic ions, covalent bonds form between nonmetals, and metallic bonds form between metals. It then lists examples of bond predictions and practice problems predicting bond type between different elements.
Radioactivity 11

There are three main types of radioactive decay: alpha, beta, and gamma. Alpha decay releases alpha particles, beta decay releases electrons, and gamma decay releases gamma rays. The half-life of a radioactive isotope is the amount of time it takes for half of the radioactive substance to decay, and is used to determine the age of fossils and rocks. In the examples given, calculations are shown for determining the amount of radioactive material remaining after a given number of half-lives based on the original amount and the half-life of the specific isotope.
Notes Acids&Bases 10

The document lists common acids and bases, including their chemical formulas and some common uses. It identifies hydrochloric acid, sulfuric acid, and nitric acid as strong acids commonly used for cleaning, manufacturing, and making explosives. Weaker acids mentioned include carbonic acid, boric acid, phosphoric acid, acetic acid, and citric acid which are used in beverages, personal care, detergents, and food production. Strong bases listed are sodium hydroxide, potassium hydroxide, calcium hydroxide, and magnesium hydroxide used in soaps, cleaners, leather processing, and pharmaceuticals. Weaker bases include ammonium hydroxide and aluminum hydroxide used in household products and
Energy And Rates Of Reactions 9

The document discusses how energy relates to chemical reactions. It explains that exothermic reactions release energy by breaking bonds, while endothermic reactions absorb energy by forming new bonds. All chemical reactions require a minimum amount of energy, called activation energy, to get started. The rates of reactions are affected by temperature, concentration, surface area, catalysts, and inhibitors. Catalysts lower the activation energy needed to start a reaction.
Writing And Balancing Chem Rxn’S 8

This document discusses writing and balancing chemical reactions. It explains that when writing chemical equations, reactants are written on the left and products are written on the right. It also notes that every atom must be balanced according to the law of conservation of mass, which states that mass cannot be created or destroyed in a chemical reaction. The document indicates that if an equation is not balanced, a coefficient can be placed in front of a molecule to balance the number of atoms, but no bonds can be broken in the process of balancing equations.
Covalent Bonding 5

Covalent bonds form when atoms share electrons to achieve a full outer shell, giving the molecule an overall charge of zero. Hydrogen always forms covalent bonds because it has only one electron to share. Examples of covalent bonds include hydrogen gas (H2) and hydrogen chloride (HCl), which can be represented using electron-dot diagrams showing the shared electrons. Molecules are combinations of atoms held together by covalent bonds, such as polyatomic ions.
Chemical Reactions 7

Chemical reactions start with one or more substances (A) and result in different substances (B) through the breaking and rearranging of bonds. Signs that a chemical reaction occurred may include the formation of a gas (bubbles), release of energy (light, sound, heat), a color change, or the production of a precipitate (solid). The document asks if these signs were present for reactions between sulfuric acid and sugar, and nitric acid and a penny.
Electrons And Chemical Bonding 3

Electrons and Chemical Bonding summarizes how atoms bond together to form new substances. Atoms bond by sharing or transferring valence electrons in their outermost shells to achieve full outer shells, which provides stability. Noble gases have full outer shells and do not bond since they are the most stable configuration for atoms.
Periodic Table Presentation 1

The document provides a key for understanding a periodic table. It indicates that solid elements are black, gases are red, and liquids are blue. It also notes that all states of elements can be changed and clear elements are unstable. The periodic table boxes list the element symbol, atomic number representing protons, and atomic mass. Groups have vertical columns of similar chemical properties, while periods have different properties horizontally. Elements are categorized as metals, metalloids, or nonmetals.
Ionic Bonding 4

Ionic bonding occurs when atoms transfer electrons, forming positive and negative ions that bond together. Specifically, metals donate electrons to nonmetals to complete their outer energy levels, with the metal always written first in the compound. The final bonded ionic compound is stable with no overall charge.
Counting Atoms 2a

This document provides instructions on writing chemical formulas and counting atoms in compounds. It explains that capital letters are used for elemental symbols, subscripts indicate the number of atoms, and numbers distributed throughout a compound indicate how many of each atom are present. Examples are given for common compounds and ions to demonstrate how to count the total number of each type of atom.