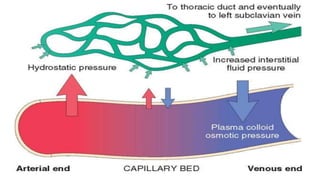
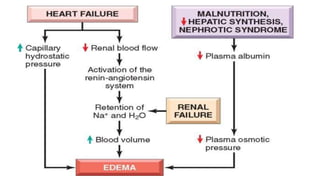
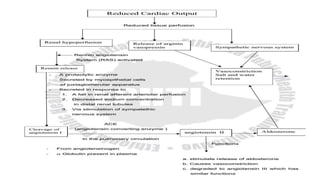
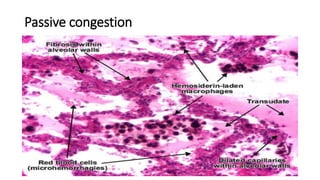
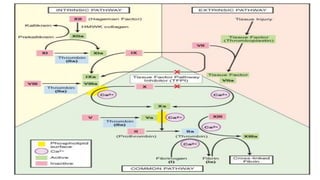
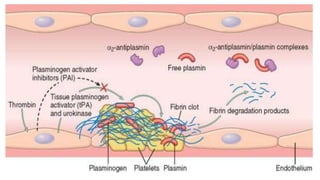
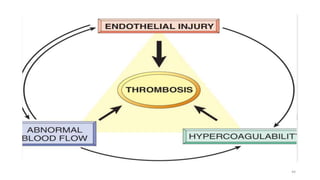
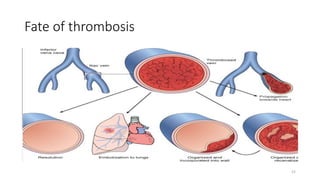
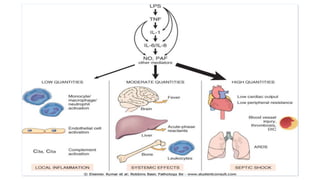
This document provides an overview of hemodynamics disorders and outlines various pathologies including edema, hyperemia, congestion, hemorrhage, hemostasis, thrombosis, embolism, and infarction. It discusses the normal balance of fluid movement across capillaries and how various pathologic conditions can disrupt this balance and cause edema. Key concepts covered include the Starling forces, categories of edema, factors influencing thrombosis, and the coagulation cascade. Morbidities associated with cardiovascular diseases are also briefly mentioned.