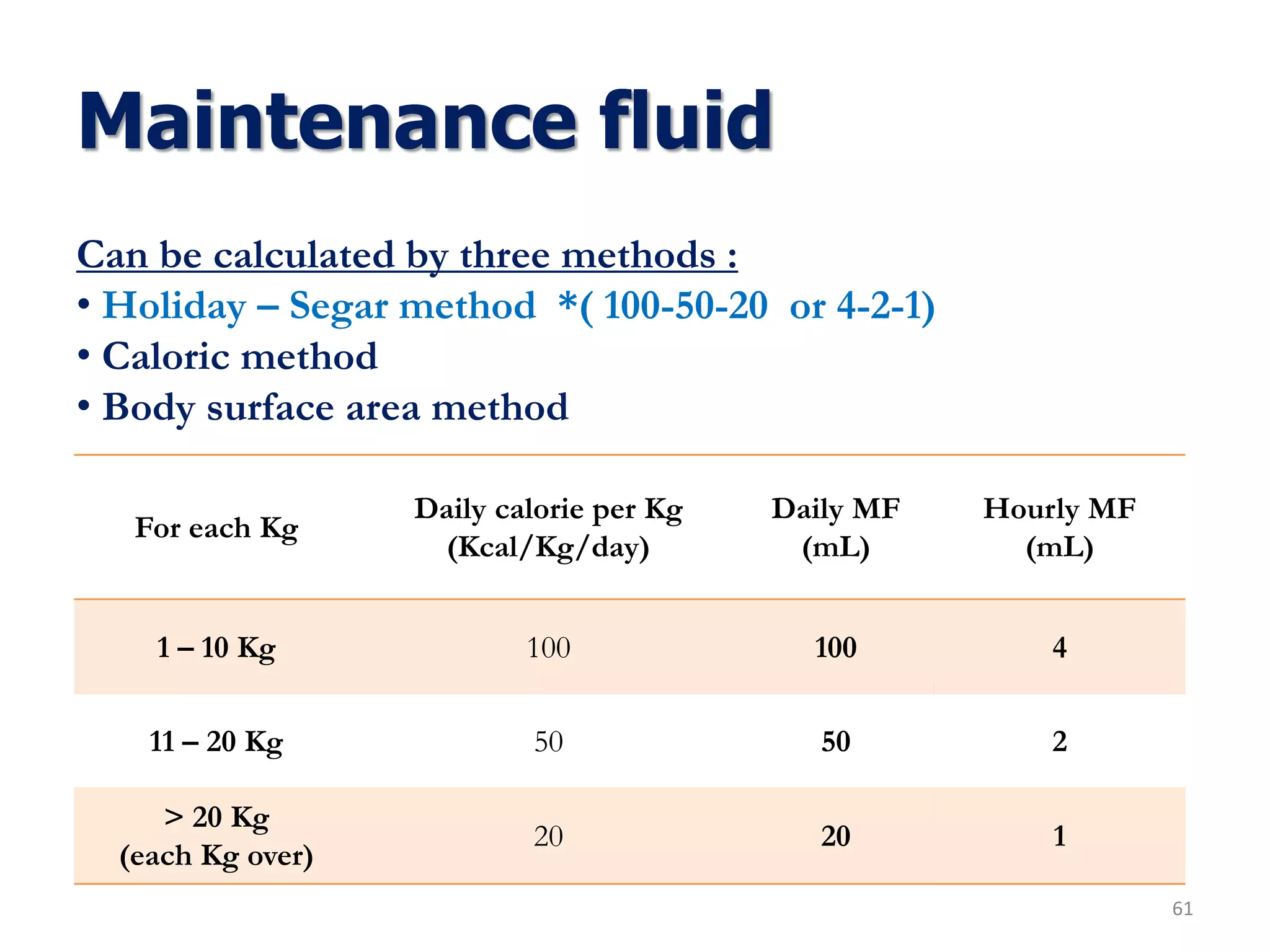
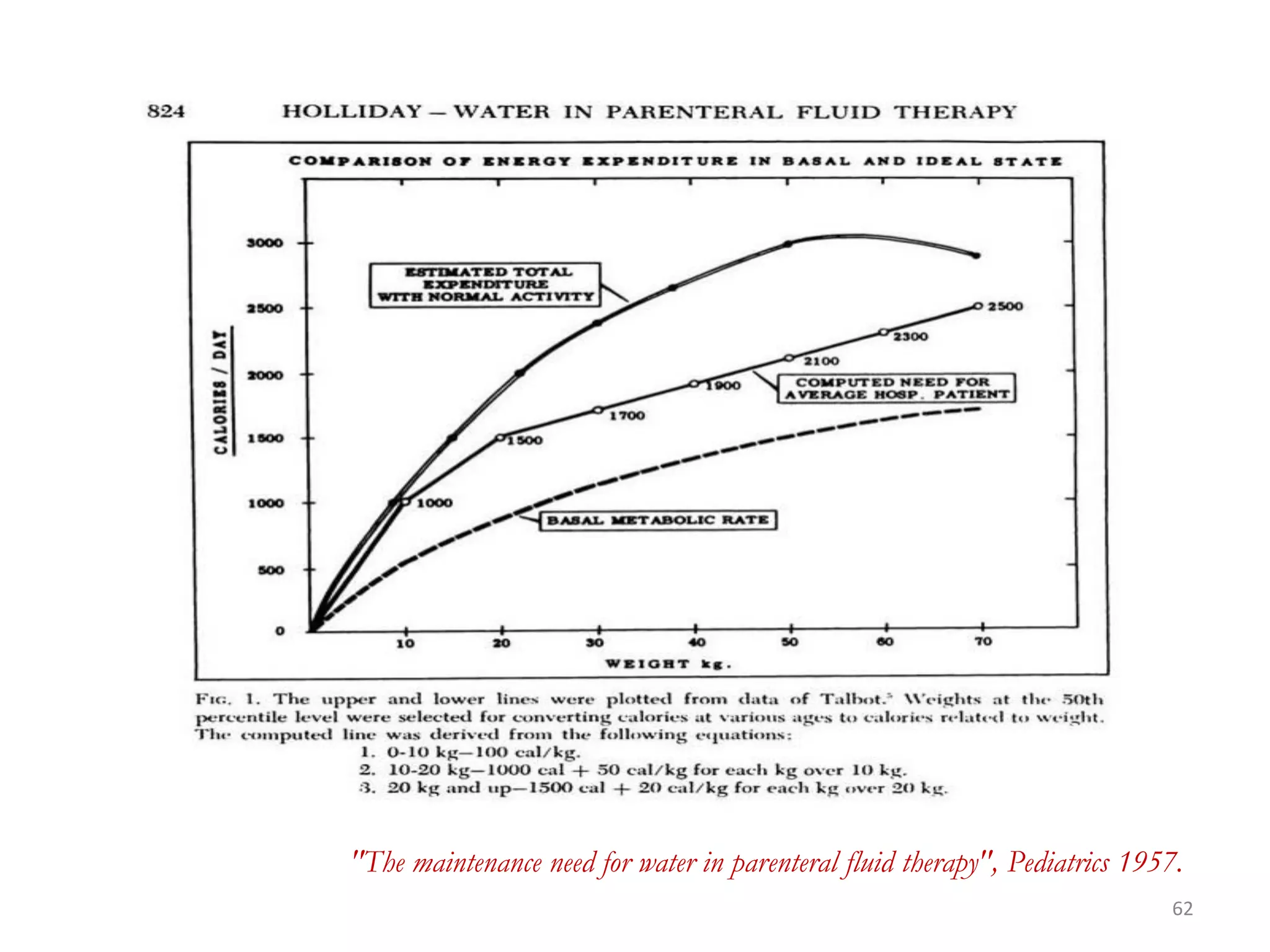
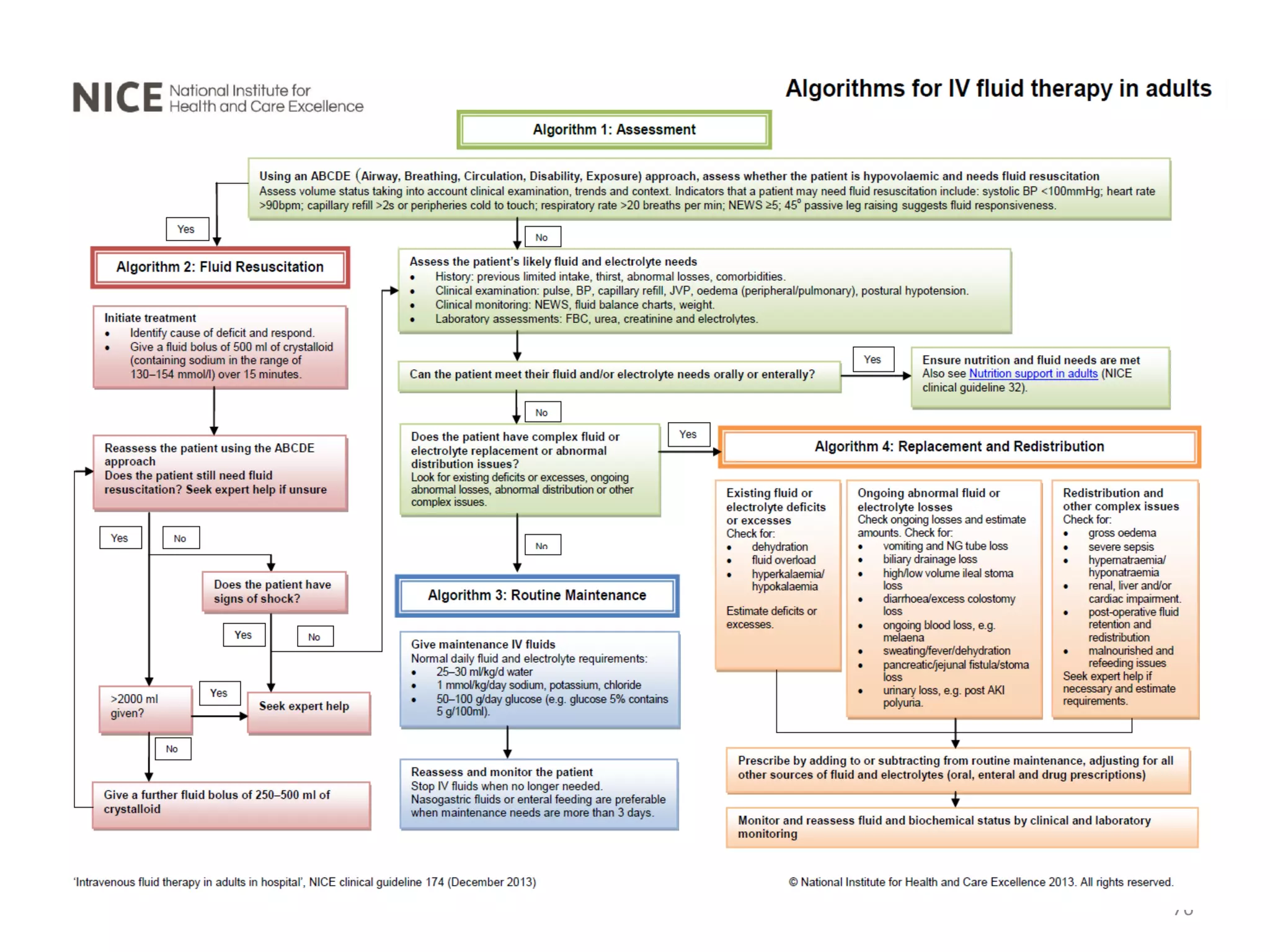
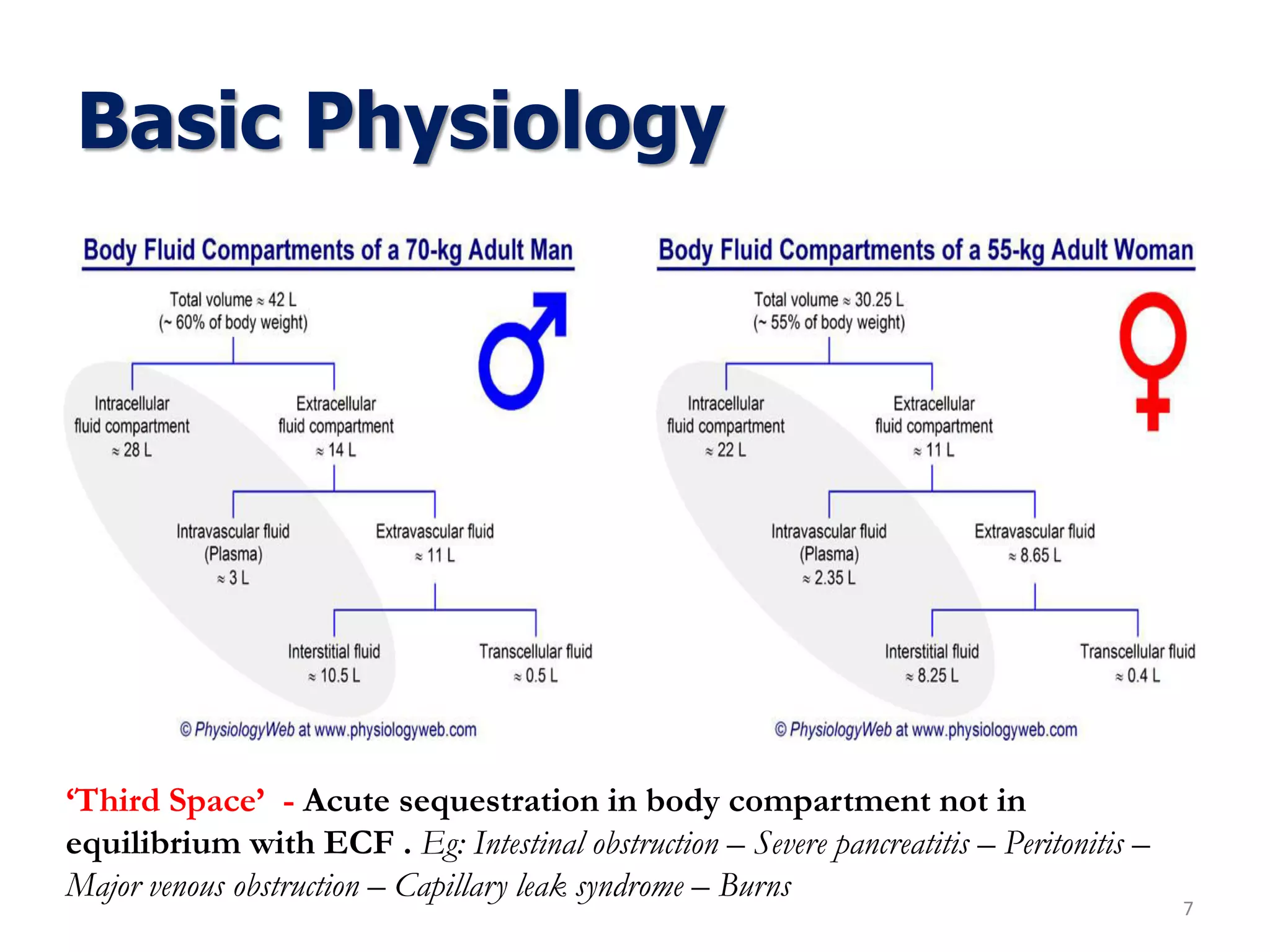
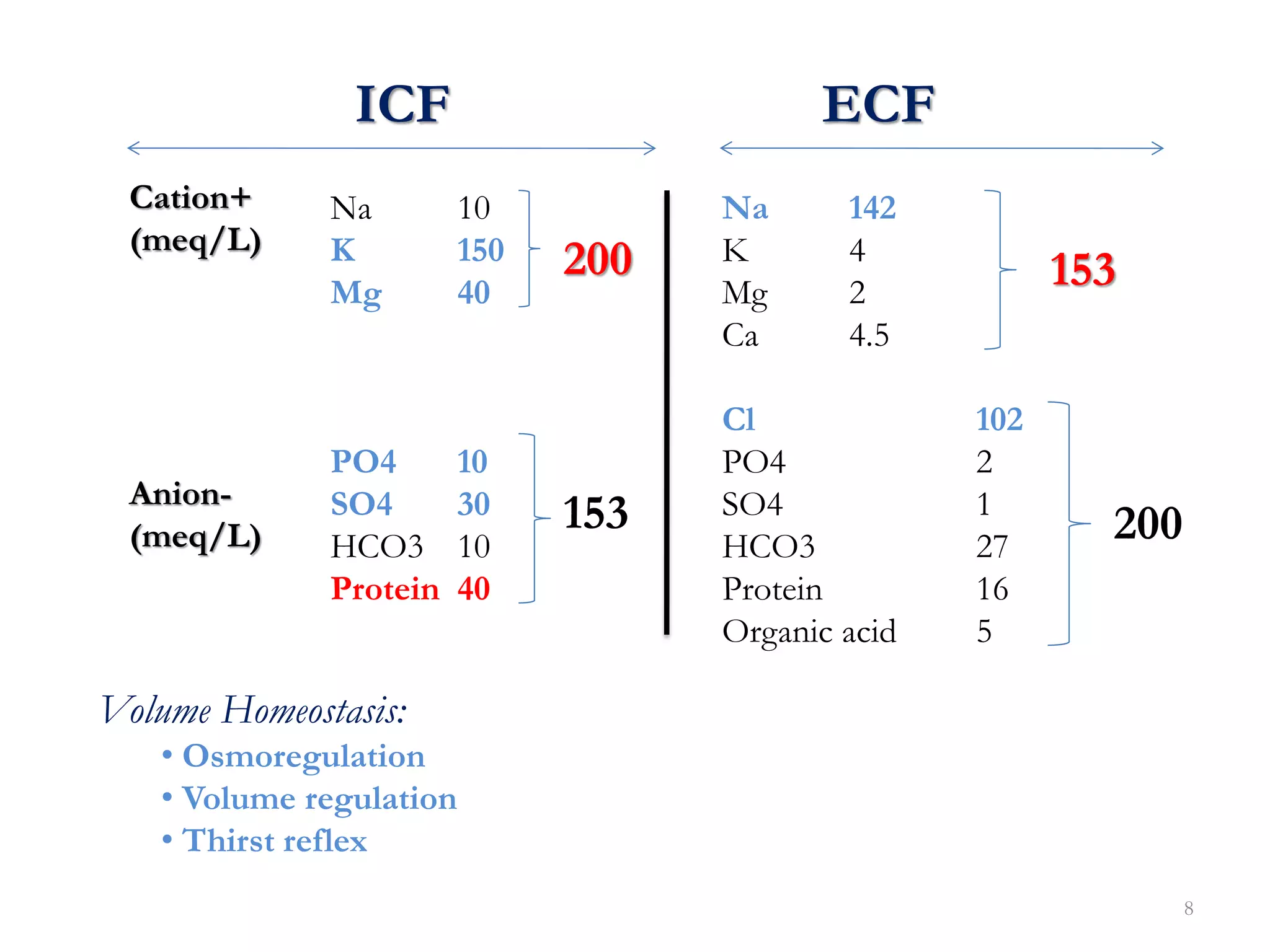
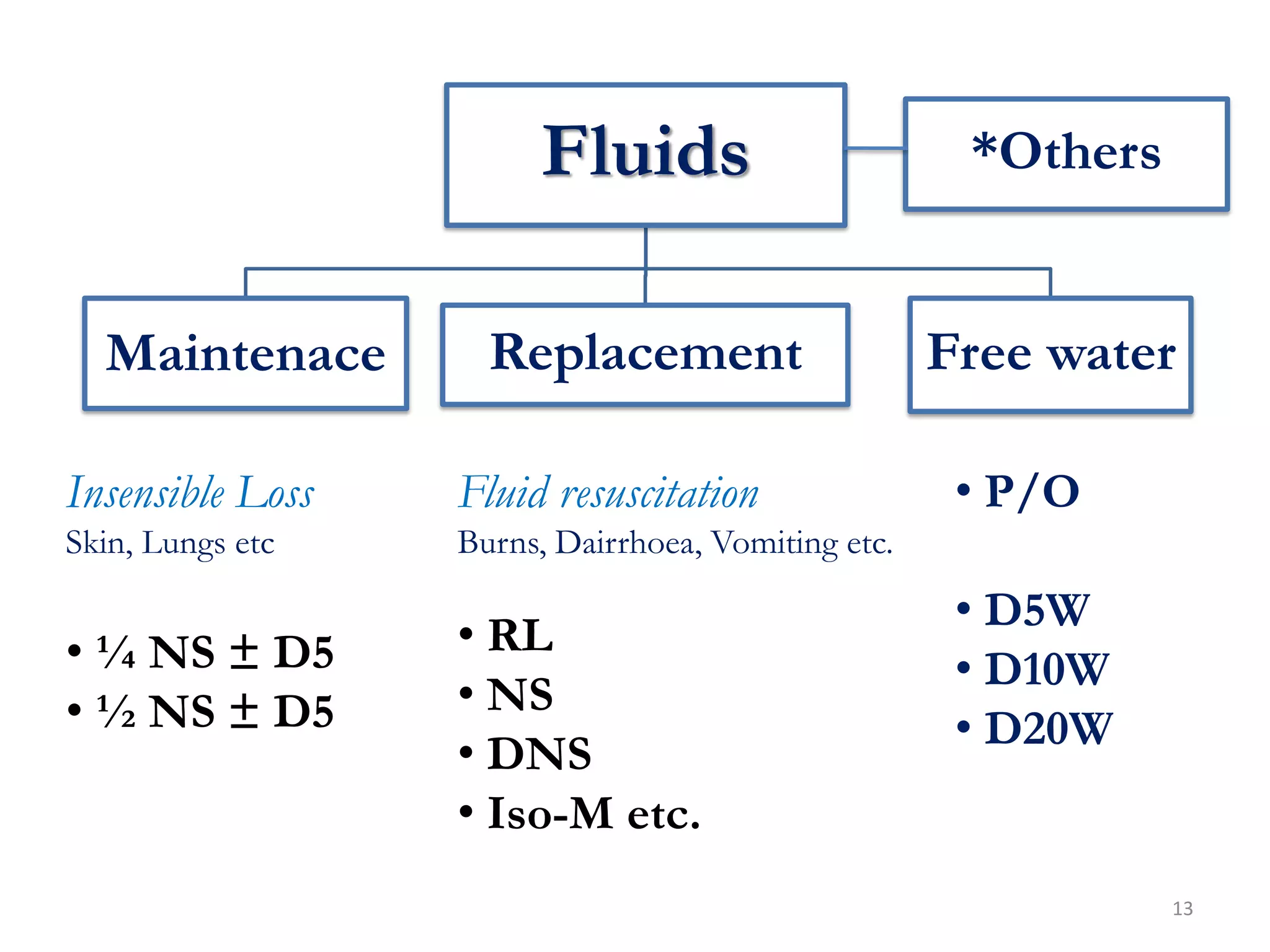
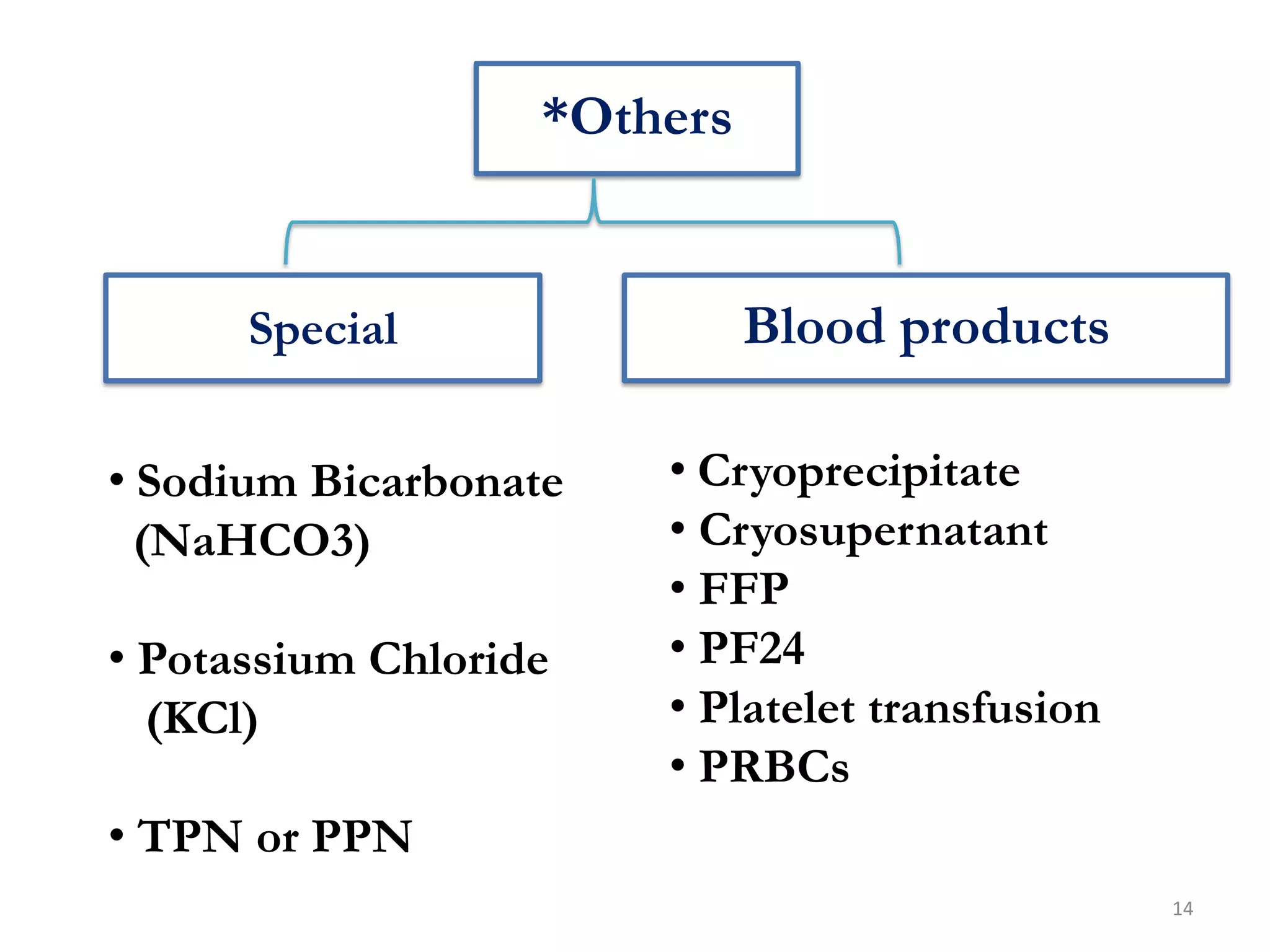
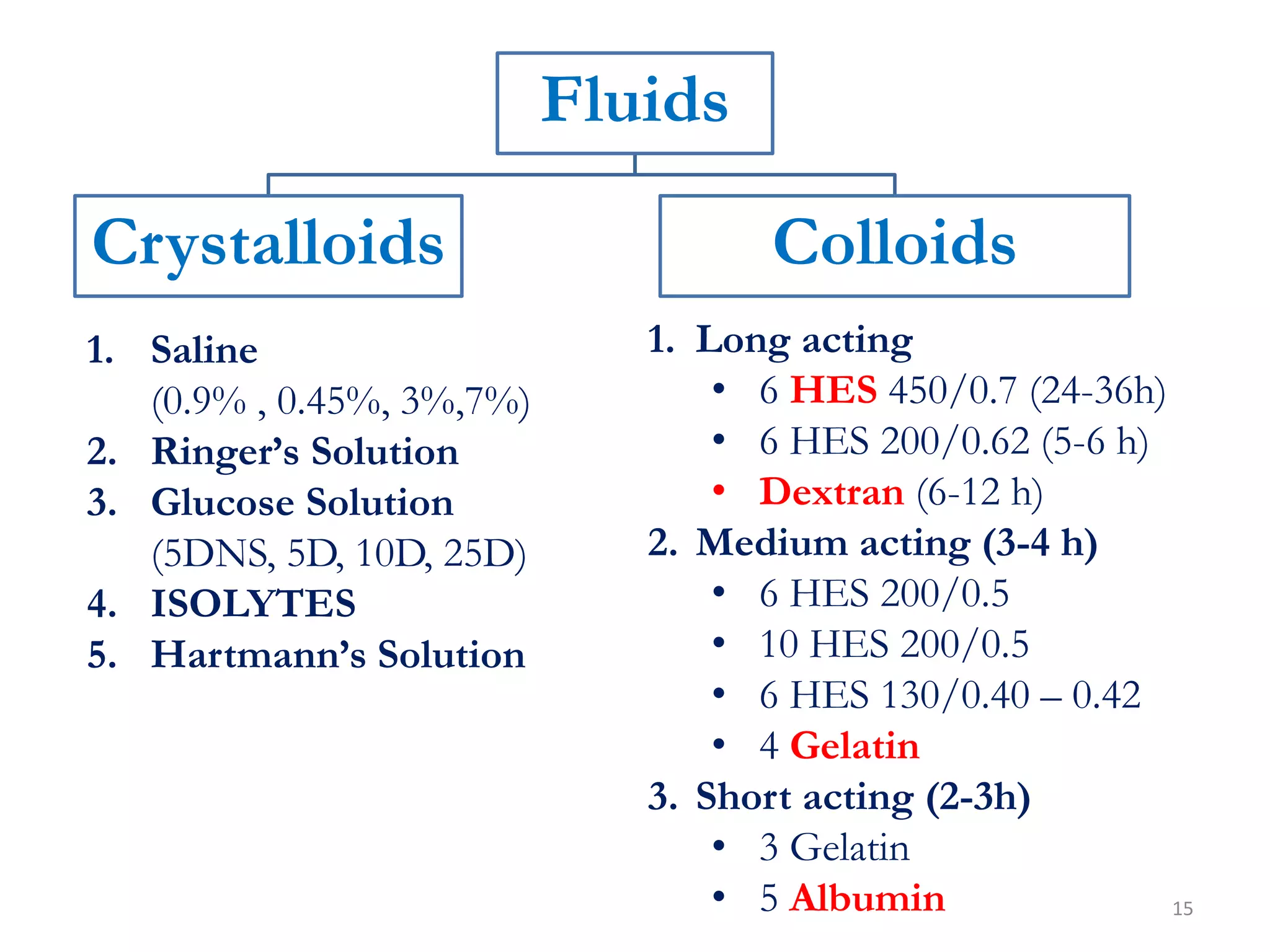
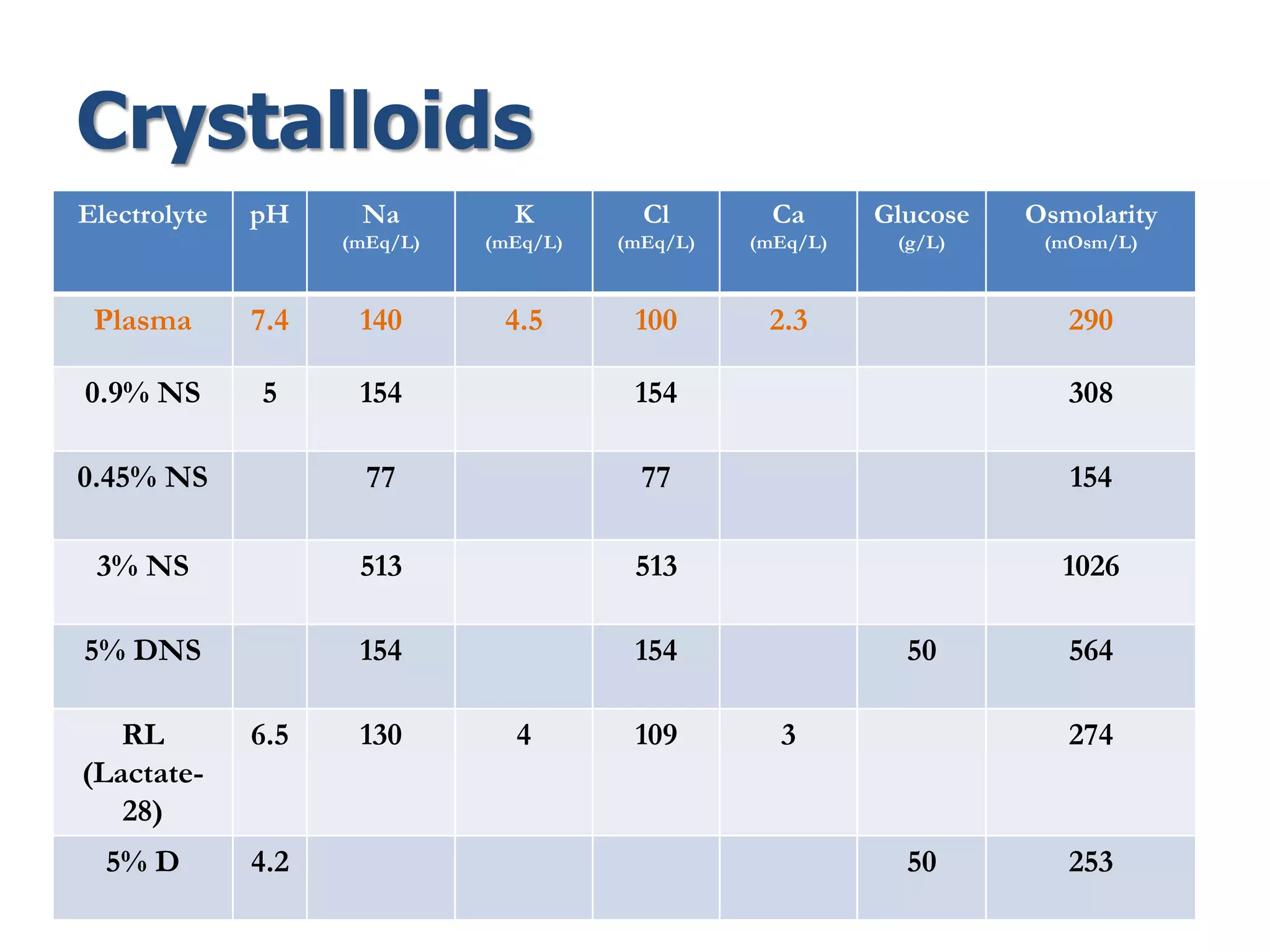
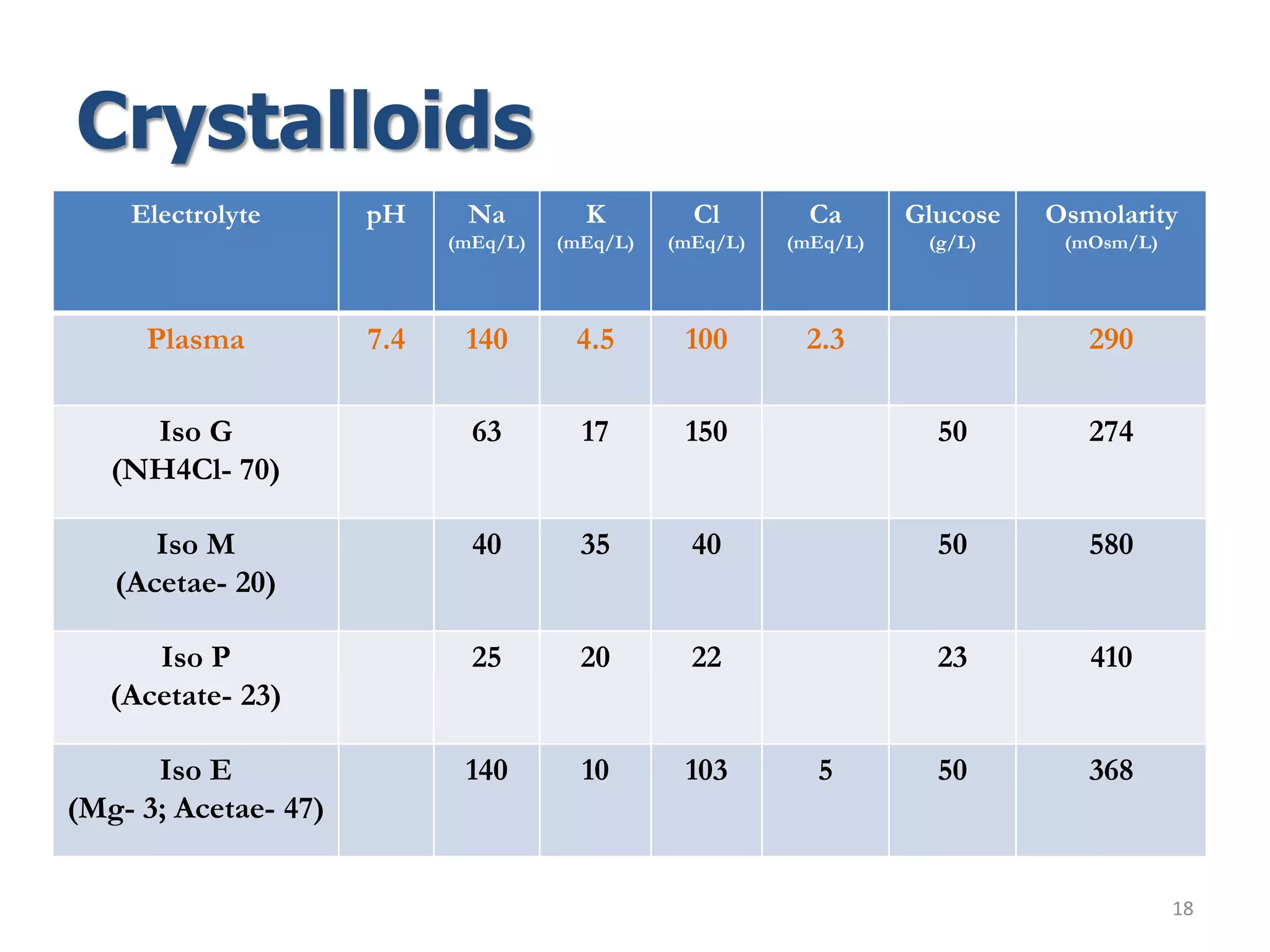
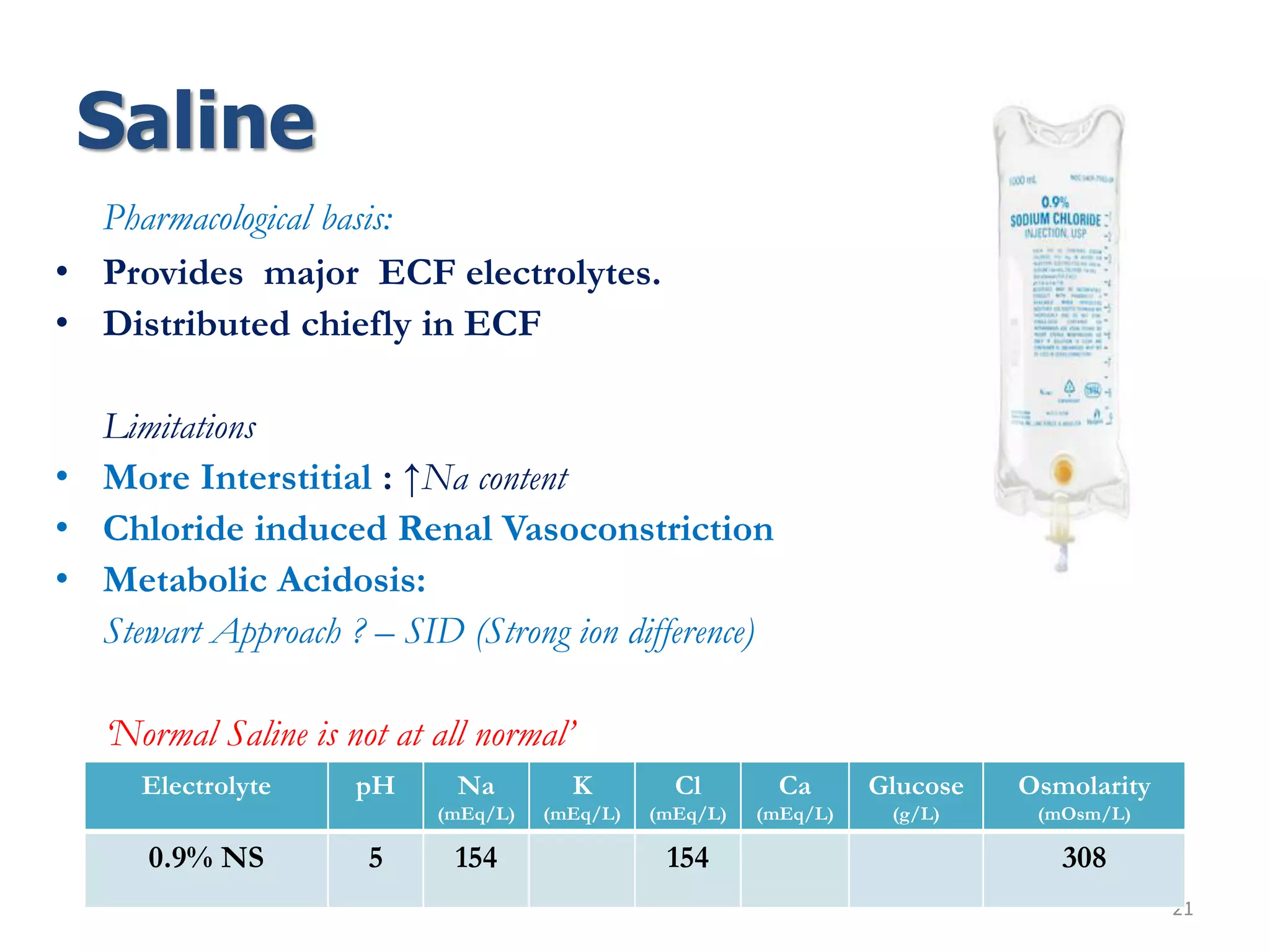
Fluid therapy involves replacing lost fluids and electrolytes to maintain homeostasis. Historically, saline solutions and Ringer's solution were developed and used to treat cholera victims and experimented on frog hearts. Common fluids include 0.9% saline, Ringer's lactate, 5% dextrose, Hartmann's solution, and Isolyte solutions. Indications for fluid selection depend on the fluid deficit and electrolyte abnormalities present. Special situations may require specific fluids like sodium bicarbonate or blood products.













































![Volume deficit ?
57
Volume deficits are best estimated by acute changes in Weight
Deficit may be Pure water deficit or Combined water & electrolyte deficit
Pure water deficit is reflected biochemically by hypernatremia, increase in plasma, osmolality,
concentrated urine, and low urine [Na+] (<15mEq/L)
Treatment involves replacement of enough water to restore plasma [Na+] to normal
The excess Na+ for which water must be provided can be estimated from the
following equation:
ΔNa = (140 Na – plasma Na) × TBW
ΔNa : represents the total Na+ (mEq) in excess of water
*Divide ΔNa by 140 to obtain the amount of water required to return serum Na+ to
140mEq/L](https://image.slidesharecdn.com/fluidtherapy11-180926154735/75/Fluid-therapy-Principles-57-2048.jpg)
![Volume deficit ?
58
This fluid deficit must be corrected in addition to giving maintenance fluids
for ongoing obligatory losses.
Combined water and electrolyte deficit is commonly associated with “Third
spacing’
Urine Na+ is often < 10mEq/L as a result of Na+conservation from RAAS
system
Decreased blood volume diminishes renal perfusion and often produces
Pre-renal azotemia (BUN: Creatinine = 20-25: 1)
The composition of the correction fluid should take into account the plasma
[Na+]. If it is normal, fluid and electrolyte losses are probably isotonic, and
the replacement fluid should be isotonic NS or its equivalent.](https://image.slidesharecdn.com/fluidtherapy11-180926154735/75/Fluid-therapy-Principles-58-2048.jpg)