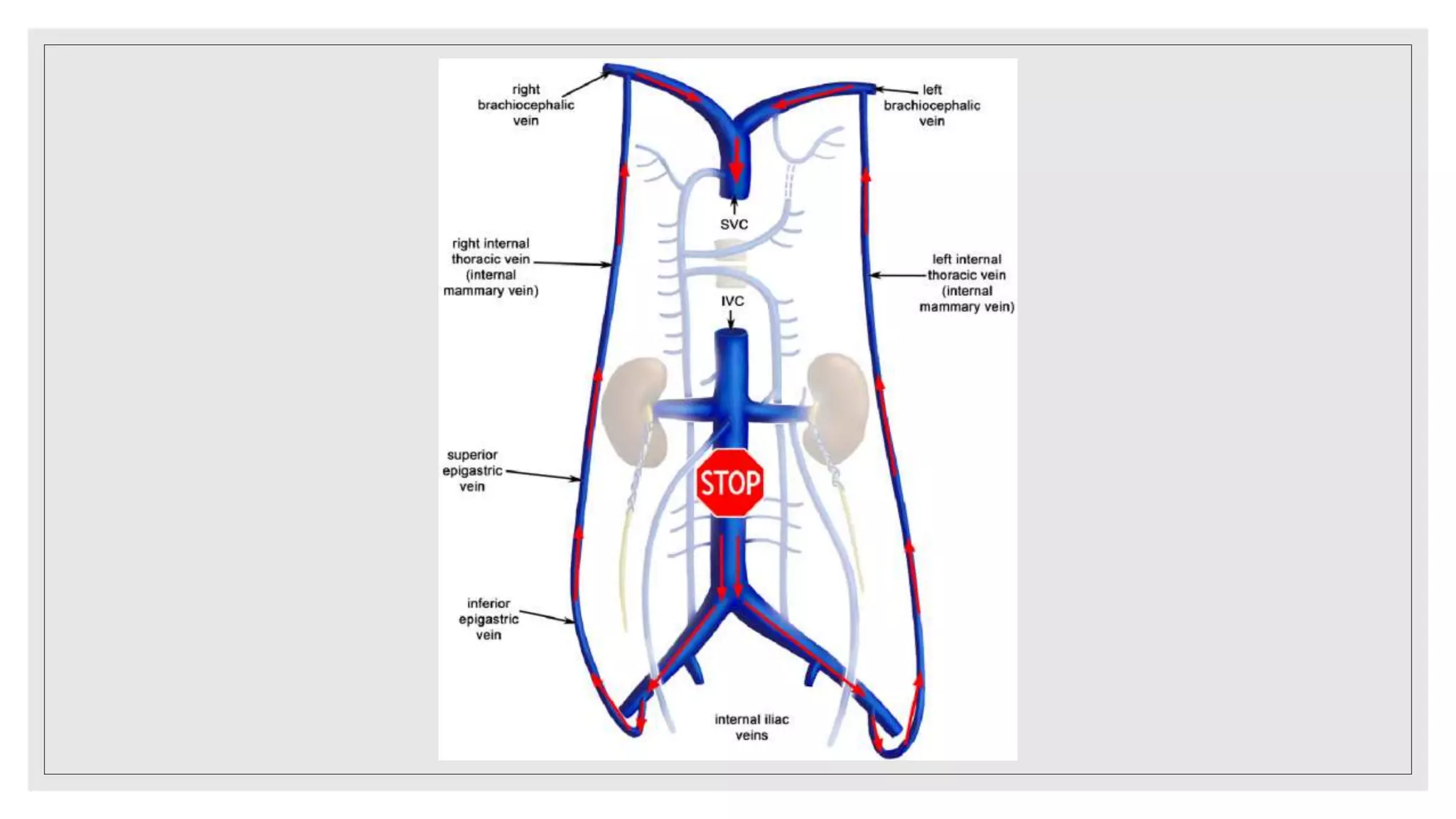
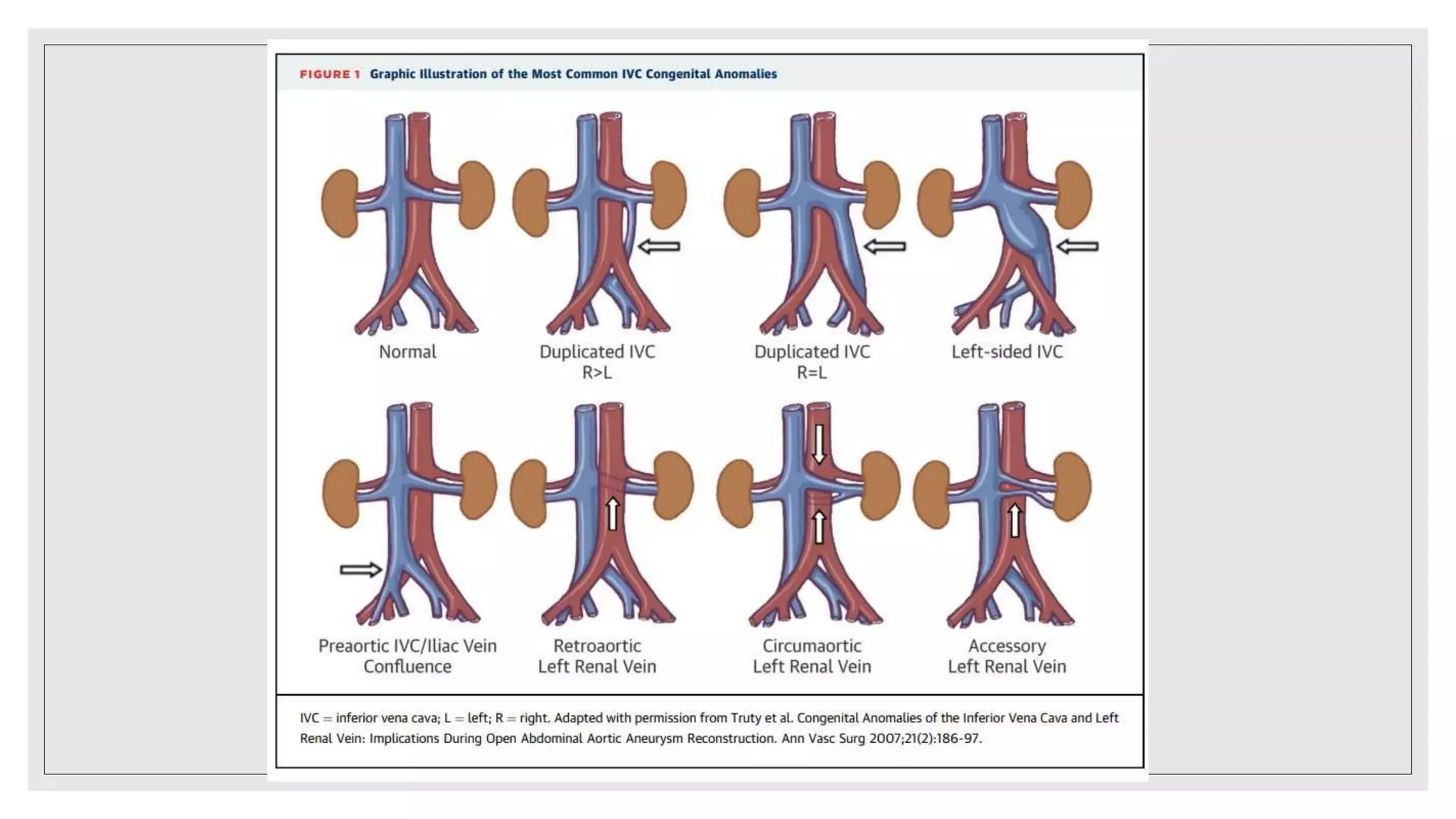
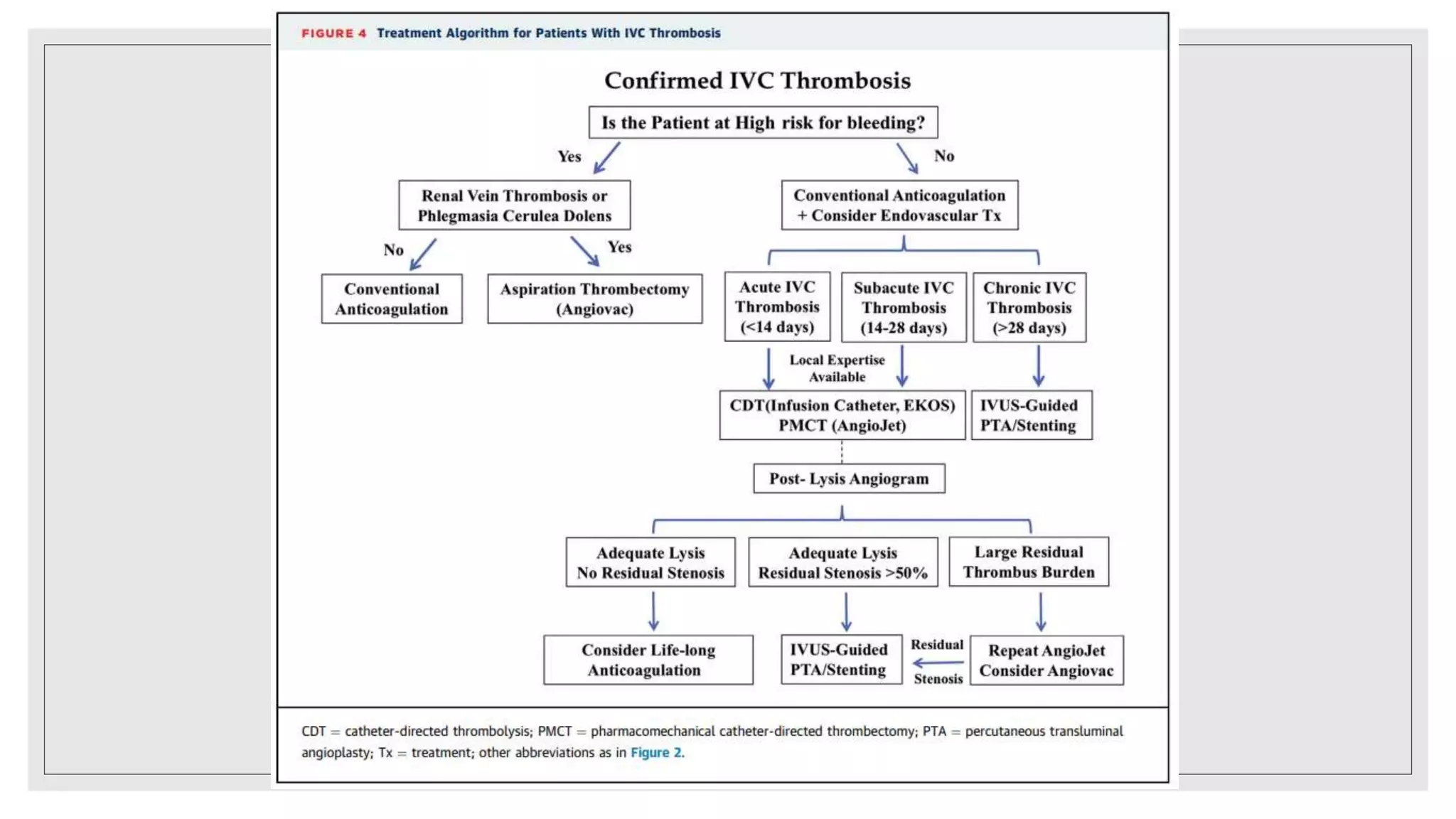
This document summarizes a clinical meeting discussing a case of inferior vena cava (IVC) thrombosis presented by Dr. Mehak Trehan. The case involved a 21-year-old male who presented with distended abdominal veins and lower back pain. Investigations revealed splenomegaly, IVC thrombosis, and a positive JAK2 mutation, leading to a diagnosis of JAK2-positive essential thrombocythemia causing the IVC thrombosis. The discussion covered the presentation, evaluation, and treatment of IVC thrombosis, noting it can be caused by hypercoagulable states, compression from adjacent structures, or congenital IVC anomalies. Evaluation involves imaging like ultrasound, CT, or MRI ven
































![Laboratory Evaluation
◦ Gayer et al recommended that all patients with an IVC thrombosis be screened for a thrombophilic disorder. [14] In
their series, 77.7% of patients with IVC thrombosis had a positive thrombophilia screen.
◦ The decision to initiate a laboratory workup for thrombophilia is complex and difficulty, because identification of
underlying thrombophilia might not affect therapeutic strategy.
◦ In addition to the challenge of deciding who should be tested, clinicians need to be aware of when to test—or more
precisely, when not to.
Gayer, G., et al. "Congenital anomalies of the inferior vena cava revealed on CT in patients with deep vein thrombosis." American Journal of Roentgenology 180.3 (2003): 729-732.](https://image.slidesharecdn.com/clinicalmeet-220424081650/75/CLINICAL-MEET-IVC-thrombosis-pptx-35-2048.jpg)















![REFERENCES
◦ McAree BJ, O'Donnell ME, Fitzmaurice GJ, Reid JA, Spence RA, Lee B. Inferior vena cava thrombosis: a review of
current practice. Vasc Med. 2013 Feb;18(1):32-43. doi: 10.1177/1358863X12471967. PMID: 23439778
◦ Alkhouli M, Morad M, Narins CR, Raza F, Bashir R. Inferior Vena Cava Thrombosis. JACC Cardiovasc Interv. 2016 Apr
11;9(7):629-43. doi: 10.1016/j.jcin.2015.12.268. Epub 2016 Mar 4. PMID: 26952909.
◦ Hollingsworth CM, Mead T. Inferior Vena Caval Thrombosis. [Updated 2021 Jul 10]. In: StatPearls [Internet]. Treasure
Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537175/
◦ https://emedicine.medscape.com/article/1933035-overview#a6](https://image.slidesharecdn.com/clinicalmeet-220424081650/75/CLINICAL-MEET-IVC-thrombosis-pptx-52-2048.jpg)
