Catalysts for Change in SCLC: Optimizing the Use of Standard Therapies and Accelerating the Transition of Emerging Strategies to the Clinic
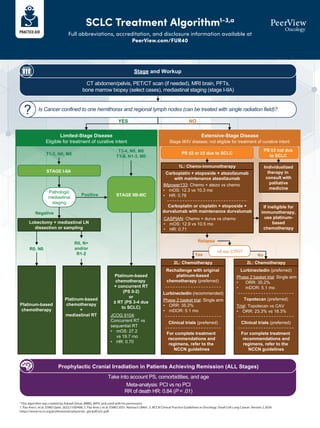
- 1. SCLC Treatment Algorithm1-3,a Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUR40 a This algorithm was created by Aakash Desai, MBBS, MPH, and used with his permission. 1. Paz-Ares L et al. ESMO Open. 2022;7:100408. 2. Paz-Ares L et al. ESMO 2021. Abstract LBA61. 3. NCCN Clinical Practice Guidelines in Oncology. Small Cell Lung Cancer. Version 2.2024. https://www.nccn.org/professionals/physician_gls/pdf/sclc.pdf. CT abdomen/pelvis, PET/CT scan (if needed), MRI brain, PFTs, bone marrow biopsy (select cases), mediastinal staging (stage I-IIA) Stage and Workup Is Cancer confined to one hemithorax and regional lymph nodes (can be treated with single radiation field)? !"#"$%&'($)*% ?#*@*&#($2,'$+'(%+3(-+$,2$0"'%+*)($*-+(-+ Limited-Stage Disease Eligible for treatment of curative intent %4$%5("6%'($)*% Extensive-Stage Disease Stage III/IV disease; not eligible for treatment of curative intent YES NO STAGE I-IIA STAGE IIB-IIIC Pathologic mediastinal staging Positive Negative Platinum-based chemotherapy + concurrent RT (PS 0-2) or ± RT (PS 3-4 due to SCLC) JCOG 9104: Concurrent RT vs sequential RT • mOS: 27.2 vs 19.7 mo • HR: 0.70 Lobectomy + mediastinal LN dissection or sampling Platinum-based chemotherapy Platinum-based chemotherapy + mediastinal RT R0, N0 R0, N+ and/or R1-2 Take into account PS, comorbidities, and age Meta-analysis: PCI vs no PCI RR of death HR: 0.84 (P = .01) Prophylactic Cranial Irradiation in Patients Achieving Remission (ALL Stages) PS ≤2 or ≥3 due to SCLC PS ≥3 not due to SCLC Individualized therapy in consult with palliative medicine 1L: Chemo-immunotherapy If ineligible for immunotherapy, use platinum- based chemotherapy >6 mo CTFI? Relapse Yes No 2L: Chemotherapy 2L: Chemotherapy Carboplatin + etoposide + atezolizumab with maintenance atezolizumab IMpower133: Chemo + atezo vs chemo • mOS: 12.3 vs 10.3 mo • HR: 0.76 Carboplatin or cisplatin + etoposide + durvalumab with maintenance durvalumab CASPIAN: Chemo + durva vs chemo • mOS: 12.9 vs 10.5 mo • HR: 0.71 Rechallenge with original platinum-based chemotherapy (preferred) Lurbinectedin (recommended) Phase 2 basket trial: Single arm • ORR: 35.2% • mDOR: 5.1 mo For complete treatment recommendations and regimens, refer to the NCCN guidelines Lurbinectedin (preferred) Phase 2 basket trial: Single arm • ORR: 35.2% • mDOR: 5.1 mo Topotecan (preferred) Trial: Topotecan vs CAV • ORR: 23.3% vs 18.3% Clinical trials (preferred) For complete treatment recommendations and regimens, refer to the NCCN guidelines Clinical trials (preferred) T1-2, N0, M0 T3-4, N0, M0 T1-4, N1-3, M0 ?
- 2. Research Trends and Emerging Therapies in SCLC Full abbreviations, accreditation, and disclosure information available at PeerView.com/FUR40 1. Gay MG et al. Cancer Cell. 2021;39:346-360. Clinical Studies Informing the Use of Current Therapy Transcriptional Signatures, Subtypes, and Therapeutic Vulnerabilities1 Novel Therapeutic Approaches on the Horizon Selected Studies for Immunotherapy in LS-SCLC Selected Studies for First- and Later-Line ES-SCLC Bispecific T-Cell Engager (BiTE) Antibody–Drug Conjugates (ADCs) ADRIATIC Phase 3 trial of durvalumab or durvalumab + tremelimumab as consolidation treatment in LS-SCLC NRG/Alliance LU005 Phase 2/3 trial of atezolizumab ± cCRT in LS-SCLC ASTRUM-005 Phase 3 trial of 1L serplulimab + chemo vs chemo in ES-SCLC KEYVIBE-008 Phase 3 trial of 1L vibostolimab + pembrolizumab + chemo vs atezolizumab + chemo in ES-SCLC NCT04702880 Phase 2 trial of 1L BMS-986012 + nivolumab + chemo in ES-SCLC LAGOON Confirmatory phase 3 trial of lurbinectedin in relapsed SCLC IMforte Phase 3 trial of lurbinectedin + atezolizumab as maintenance therapy in ES-SCLC following 1L immunochemotherapy LUPER Phase 1/2 trial of lurbinectedin + pembrolizumab in relapsed SCLC DeLLphi-300 Phase 1 trial of tarlatamab monotherapy or in combination with ICIs in SCLC ORR: 23%, mPFS: 3.7 mo; mOS: 13.2 mo DeLLphi-301 Phase 2 trial of tarlatamab in ≥3L SCLC ORR: 40% for 10-mg group, 32% for 100-mg group mPFS: 4.9 mo for 10-mg group, 3.9 mo for 100-mg group NCT04145622 Phase 1/2 FIH trial of ifinatamab deruxtecan (B7-H3 targeting ADCs) in solid tumor, including SCLC NCT05280470 Phase 2 trial of ifinatamab deruxtecan in pre-treated ES-SCLC IMMU-132-01 Phase 1/2 trial of sacituzumab govitecan (TROP2 targeting ADCs) in epithelial cancer, including SCLC Antibody Chemo drug + = Antibody–drug conjugates FDA priority review
