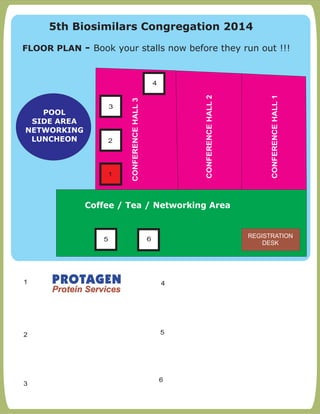
- The document outlines the agenda and speaker details for the 5th Biosimilars Congregation 2014 conference taking place on September 10th, 2014 in Mumbai, India.
- The conference will bring together leaders in the pharmaceutical, biotechnology, and regulatory fields to discuss challenges in biosimilar development and the evolving regulatory landscape.
- Key topics to be covered include assessing biosimilar similarity, strategic choices for biobetters and biosimilars, safety evaluation, regulatory updates, and emerging market opportunities for biosimilars in countries like India.