‘Pharmacy to the world can’t make diapers, hospital beds’ by Dr.Mahboob Ali Khan Phd
The document discusses India's lag in manufacturing medical devices despite being a major exporter of pharmaceuticals. It notes that India imports many basic medical devices like hospital beds and monitors. The secretary cites the example of linear accelerator machines used for cancer treatment, which cost over Rs. 5 crore to import but could cost Rs. 1.5 crore if manufactured in India, allowing more hospitals to offer cancer treatment. Recent duty hikes on medical device imports are aimed to boost domestic manufacturing over time by correcting inverted duty structures that currently favor imports. The government is also working to develop its own quality standards for medical devices rather than relying only on international ones like USFDA.
Recommended
Recommended
More Related Content
Similar to ‘Pharmacy to the world can’t make diapers, hospital beds’ by Dr.Mahboob Ali Khan Phd
Similar to ‘Pharmacy to the world can’t make diapers, hospital beds’ by Dr.Mahboob Ali Khan Phd (20)
More from Healthcare consultant
More from Healthcare consultant (20)
Recently uploaded
Recently uploaded (20)
‘Pharmacy to the world can’t make diapers, hospital beds’ by Dr.Mahboob Ali Khan Phd
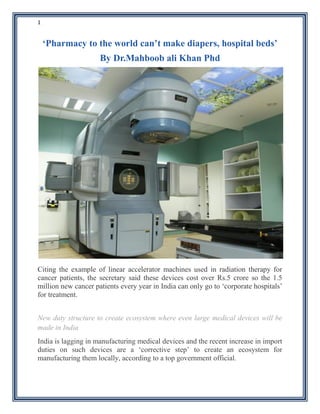
- 1. 1 ‘Pharmacy to the world can’t make diapers, hospital beds’ By Dr.Mahboob ali Khan Phd Citing the example of linear accelerator machines used in radiation therapy for cancer patients, the secretary said these devices cost over Rs.5 crore so the 1.5 million new cancer patients every year in India can only go to ‘corporate hospitals’ for treatment. New duty structure to create ecosystem where even large medical devices will be made in India India is lagging in manufacturing medical devices and the recent increase in import duties on such devices are a ‘corrective step’ to create an ecosystem for manufacturing them locally, according to a top government official.
- 2. 2 While India is known as the pharmacy of the world, exporting to over 200 countries, it is falling behind in medical devices whose regulation was recently entrusted with the Department of Pharmaceuticals, “We are producing so many things, sending even satellites to Mars in a very cost-effective way. But we are not able to produce ordinary medical devices. You go to any hospital you will find that all the equipment is coming from other countries, even the ordinary monitors, hospital beds. Can’t we produce hospital cots and beds here?”I reiterate. Citing the example of linear accelerator machines used in radiation therapy for cancer patients, the secretary said these devices cost over Rs.5 crore so the 1.5 million new cancer patients every year in India can only go to ‘corporate hospitals’ for treatment. “If you manufacture such a technology within India, and it is available, it will cost Rs.1.5 crore. But we don’t manufacture in India. We all think only foreign equipment will work that are available only in topmost tertiary care hospitals. “Suppose you manufacture them here, even district hospitals can have this equipment and treat cancer,” he said. Imports from Ireland or China are not only convenient but also cheaper owing to inverted duty structures that hamper domestic manufacturing activity, he said referring to the decision to hike duties on several medical devices in January. “Recently, we initiated correction of duty structures. If you import the equipment as it is, it is cheaper, it is convenient to get (them) into the hospital from Ireland or China. “If you manufacture the same thing in India, duty is there. So it is convenient for all of us, but we want to rectify this… It wouldn’t happen overnight,” I know.I think this approach will help in manufacturing medical devices in India over time. “You will be surprised baby diapers used by the millions of babies born in our country, are also called a medical device. “We import Rs.200 crore of diapers, (but) now, a few companies have come into India. We will create an ecosystem that even large medical devices are made in India over a period of time,argubly there was no reason why India should import ordinary medical devices, including hospital beds and even diapers.
- 3. 3 I think said the government is working on standards for medical devices rather than rely on USFDA norms currently prescribed in government procurements for medical equipment. He was speaking at an event to introduce a voluntary certification process for domestically-produced medical devices organised by the Quality Council of India with the Association of Indian Medical Device Industry. I think such a certification would help create confidence among doctors and consumers about the reliability of locally manufactured products. The government is also prodding all pharma producers to adapt World Health Organisation (WHO) standards to reassure the world that Indian drugs are not only cost-effective but are also of good quality. “Whatever be the quality constraints that people keep complaining of (in pharma products), that’s not a major issue. Our own people undermine our own products,” I think. Though Indian standards are sufficient, the move to WHO standards would help bolster quality perception. According to India Ratings, a Fitch company, the Indian pharmaceutical industry is estimated to grow at 20 per cent compound annual growth rate over the next five years. The Indian pharmaceutical industry, which is expected to grow over 15 per cent every year between 2015 and 2020, will outperform the global pharmaceutical industry, which is set to grow at an annual rate of five per cent between the same period. Presently the market size of the pharmaceutical industry in India stands at U.S. $ 20 billion. As on March 2014, Indian pharmaceutical manufacturing facilities registered with the U.S. Food and Drug Administration (FDA) stood at 523, highest for any country outside the U.S. By 2020, India is likely to be among the top three pharmaceutical markets by incremental growth and sixth largest market globally in absolute size. India’s cost of production is significantly lower than that of the US and almost half of that of Europe. It gives a competitive edge to India over others, according to a report by the Indian Brand Equity Foundation.
