ozone layer
- 1. Ozone Layer By: Karina Cortez
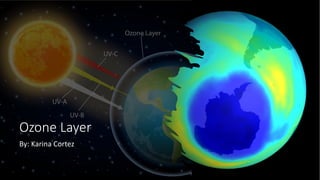
- 2. What is the ozone layer? • A region of the stratosphere on Earth called the ozone layer has a relatively high concentration of ozone (O3) molecules. A major percentage of the sun's harmful ultraviolet (UV) radiation is absorbed and blocked by the ozone molecule, which is made up of three oxygen atoms. Ozone is essential for protecting life on Earth. The majority of the sun's dangerous UV rays are blocked from reaching the Earth's surface by this layer, which serves as a shield.
- 3. Where is the Ozone Layer? • The ozone layer is located at the stratosphere, which is around 10 to 30 kilometers (6 to 19 miles) above the surface of the planet. The thickness and concentration of this layer, which is present all throughout the world, can change based on variables like latitude and altitude. The polar areas have seen the most ozone depletion, or the major drop of ozone in this layer, which has caused ozone holes to appear, especially over Antarctica. •
- 4. Why is it important? • For life to exist on Earth, the ozone layer's protective function is essential. It protects living things against ultraviolet (UV) radiation's negative effects, especially the UV-B and UV-C rays, which are the most destructive. These rays can harm ecosystems and alter food chains in addition to causing a number of health problems like skin cancer, cataracts, and sunburn. Significant threats to both human health and the environment can result from ozone depletion, which can increase the quantity of UV radiation that reaches the surface.
- 5. Ozone Depletion • A problem or an issue with the ozone layer is ozone depleting substances (ODS), which are compounds created by humans, are one of the main concerns with the ozone layer. In the past, ODS like carbon tetrachloride, halons, and chlorofluorocarbons (CFCs) were frequently utilized in items like foam-blowing agents, aerosol propellants, and refrigerants. These compounds break down ozone molecules when released into the atmosphere, which contributes to the thinning of the ozone layer and the development of ozone holes. •
- 6. Solutions for Ozone Depletion! • International cooperation has played a significant role in the success of the solutions to the ozone depletion problem. The Montreal Protocol, a global agreement to gradually phase down the manufacturing and use of ODS, was the most important action taken in 1987. This agreement has greatly reduced ODS production, which has caused the ozone layer to gradually recover. Maintaining and further repairing the ozone layer still depends on continued adherence to the Montreal Protocol as well as monitoring and enforcement efforts. By limiting the use of ODS and encouraging alternatives with a lower environmental impact, public awareness and education about the need of ozone protection also play a critical role in reducing ozone depletion.
- 8. Ozone Layer • All in all, the ozone layer is vital to keep life on Earth healthy and living as it protects us from the sun's harmful rays, so it is important to take measures to keep the ozone healthy as well. It will require cooperation from all of us to reduce the ozone hole. Urge your friends to purchase locally, cut back on meat consumption, drive less, and dispose of outdated fire extinguishers and cooling appliances that contain ozone-depleting materials safely.