Creating an HIV Prevention–Certified Provider Workforce: A Training and Certificate Program Designed to Improve the Competencies of Providers to Deliver Comprehensive HIV Prevention
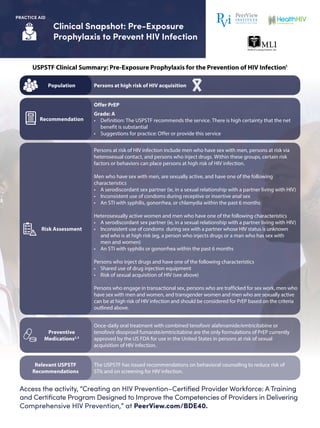
- 1. Clinical Snapshot: Pre-Exposure Prophylaxis to Prevent HIV Infection Access the activity, “Creating an HIV Prevention–Certified Provider Workforce: A Training and Certificate Program Designed to Improve the Competencies of Providers in Delivering Comprehensive HIV Prevention,” at PeerView.com/BDE40. PRACTICE AID USPSTF Clinical Summary: Pre-Exposure Prophylaxis for the Prevention of HIV Infection1 Persons at high risk of HIV acquisitionPopulation Offer PrEP Grade: A • Definition: The USPSTF recommends the service. There is high certainty that the net benefit is substantial • Suggestions for practice: Offer or provide this service Recommendation Persons at risk of HIV infection include men who have sex with men, persons at risk via heterosexual contact, and persons who inject drugs. Within these groups, certain risk factors or behaviors can place persons at high risk of HIV infection. Men who have sex with men, are sexually active, and have one of the following characteristics • A serodiscordant sex partner (ie, in a sexual relationship with a partner living with HIV) • Inconsistent use of condoms during receptive or insertive anal sex • An STI with syphilis, gonorrhea, or chlamydia within the past 6 months Heterosexually active women and men who have one of the following characteristics • A serodiscordant sex partner (ie, in a sexual relationship with a partner living with HIV) • Inconsistent use of condoms during sex with a partner whose HIV status is unknown and who is at high risk (eg, a person who injects drugs or a man who has sex with men and women) • An STI with syphilis or gonorrhea within the past 6 months Persons who inject drugs and have one of the following characteristics • Shared use of drug injection equipment • Risk of sexual acquisition of HIV (see above) Persons who engage in transactional sex, persons who are trafficked for sex work, men who have sex with men and women, and transgender women and men who are sexually active can be at high risk of HIV infection and should be considered for PrEP based on the criteria outlined above. Risk Assessment Once-daily oral treatment with combined tenofovir alafenamide/emtricitabine or tenofovir disoproxil fumarate/emtricitabine are the only formulations of PrEP currently approved by the US FDA for use in the United States in persons at risk of sexual acquisition of HIV infection. Preventive Medications2,3 The USPSTF has issued recommendations on behavioral counseling to reduce risk of STIs and on screening for HIV infection. Relevant USPSTF Recommendations
- 2. Clinical Snapshot: Pre-Exposure Prophylaxis to Prevent HIV Infection Access the activity, “Creating an HIV Prevention–Certified Provider Workforce: A Training and Certificate Program Designed to Improve the Competencies of Providers in Delivering Comprehensive HIV Prevention,” at PeerView.com/BDE40. PRACTICE AID Two Ways to Identify Candidates for PrEP4 Passive Prescribing • Relies on patients self-identifying as being at risk of HIV infection and asking about PrEP; however, many at-risk patients do not recognize their need for PrEP Active Screening • Requires that physicians, or staff, take sexual history from all patients, which can be overwhelming – Commit to talking to subsets of potentially high-risk patients, such as MSM or transgender patients – Sexual orientation and gender identity are not direct risk factors; a nuanced sexual history is often needed to understand potential exposures – A diagnosis of syphilis or other bacterial STI is a marker of high risk of HIV acquisition Questions to Ask When Assessing Risk of HIV Infection5-7 Sexual risk Have you had sex in the past 6 months? If “Yes,” with how many partners? Do you have sex with men, women, or both men and women? How often do you use a condom with these partners? As far as you know, do any of your partners have HIV infection? Have you been treated for an STI, such as gonorrhea, chlamydia, or syphilis? If “Yes,” do you know which infection you had? Have you used methamphetamines, such as crystal or speed? Injection risk Have you ever injected drugs that were not prescribed for you? If “Yes,” have you injected in the past 6 months? Do you use needles or injection equipment after they have been used by someone else? Patient Identification
- 3. Clinical Snapshot: Pre-Exposure Prophylaxis to Prevent HIV Infection Access the activity, “Creating an HIV Prevention–Certified Provider Workforce: A Training and Certificate Program Designed to Improve the Competencies of Providers in Delivering Comprehensive HIV Prevention,” at PeerView.com/BDE40. PRACTICE AID HIV: human immunodeficiency virus; PrEP: pre-exposure prophylaxis; PWID: persons who inject drugs; STI: sexually transmitted infection; USPSTF: US Preventive Services Task Force. 1. US Preventive Services Task Force et al. JAMA. 2019;321:2203-2213. 2. Descovy (emtricitabine and tenofovir alafenamide) Prescribing Information. https://www.gilead.com/~/media/Files/pdfs/ medicines/hiv/descovy/descovy_pi.pdf. Accessed October 18, 2019. 3. Truvada (emtricitabine and tenofovir disoproxil fumarate) Prescribing Information. https://www.gilead.com/~/media/Files/pdfs/ medicines/hiv/truvada/truvada_pi.pdf. Accessed October 18, 2019. 4. Stewart J, Stekler JD. J Fam Pract. 2019;68:254-261. 5. www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2017.pdf. Accessed October 14, 2019. 6. Smith DK et al. J Addict Med. 2015;9:226-232. 7. Smith DK al. J Acquir Immune Defic Syndr. 2012;60:421-427. Recommended Indications for PrEP5 Men Who Have Sex With Men Heterosexual Women and Men People Who Inject Drugs • Adult or adolescent male patient weighing at least 35 kg (77 lbs) – Without acute or established HIV infection – Any male sex partners in past 6 months (if also has sex with women; see next box) – Not in a monogamous partnership with a recently tested, HIV-negative man • Adult or adolescent person weighing at least 35 kg (77 lbs) – Without acute or established HIV infection – Any sex with opposite sex partners in past 6 months – Not in a monogamous partnership with a recently tested, HIV-negative partner • Adult or adolescent person weighing at least 35 kg (77 lbs) – Without acute or established HIV infection – Any injection of drugs not prescribed by a clinician in past 6 months And at least one of the following • Any anal sex without condoms (receptive or insertive) in past 6 months • A bacterial STI (syphilis, gonorrhea, or chlamydia) diagnosed or reported in past 6 months And at least one of the following • Is a man who has sex with both women and men (also evaluate by criteria in previous boxes) • Infrequently uses condoms during sex with one or more partners of unknown HIV status who are known to be at substantial risk of HIV infection (PWID or bisexual male partner) • Is in an ongoing sexual relationship with an HIV-positive partner • A bacterial STI (syphilis, gonorrhea in women or men) diagnosed or reported in past 6 months And at least one of the following • Any sharing of injection or drug preparation equipment in past 6 months • Risk of sexual acquisition (also evaluate by criteria in previous boxes)
- 4. Flow Chart: Any Prescribing Healthcare Provider Can Provide PrEP Care1-3 Access the activity, “Creating an HIV Prevention–Certified Provider Workforce: A Training and Certificate Program Designed to Improve the Competencies of Providers in Delivering Comprehensive HIV Prevention,” at PeerView.com/BDE40. PRACTICE AID HIV: human immunodeficiency virus; PrEP: pre-exposure prophylaxis. 1. https://www.cdc.gov/vitalsigns/hivprep/index.html. Accessed October 14, 2019. 2. Truvada (emtricitabine and tenofovir disoproxil fumarate) Prescribing Information. http://www.gilead.com/~/ media/Files/pdfs/medicines/hiv/truvada/truvada_pi.pdf. Accessed October 14, 2019. 3. Descovy (emtricitabine and tenofovir alafenamide) Prescribing Information. https://www.gilead.com/~/media/ Files/pdfs/medicines/hiv/descovy/descovy_pi.pdf. Accessed October 14, 2019. • If HIV positive, provide or refer patient for HIV treatment and other services to maintain health and prevent further spread of HIV • If HIV negative, ask about sex and drug use behaviors – If HIV risk is low, discuss prevention methods – If HIV risk is high, discuss risk behaviors, PrEP, and use of other prevention methods • If tests show reason not to prescribe PrEP (eg, abnormal kidney function), discuss other prevention methods • If tests show patient still benefits from taking PrEP, go to next step • Prescribe PrEP, and instruct patient to take one pill every day • Currently tenofovir alafenamide/ emtricitabine or tenofovir disoproxil fumarate/emtricitabine are only medications approved by the FDA for PrEP • Most public and private insurance programs cover PrEP, and patients can get help with their copayments • Drug assistance programs can help patients without insurance pay for PrEP • Schedule appointments every 3 months for follow-up, including HIV testing and prescription refills HELP ORDER PRESCRIBE FOLLOW UP TEST
- 5. Patient Assessment: Before and After Initiating PrEP Access the activity, “Creating an HIV Prevention–Certified Provider Workforce: A Training and Certificate Program Designed to Improve the Competencies of Providers in Delivering Comprehensive HIV Prevention,” at PeerView.com/BDE40. PRACTICE AID Signs/symptoms of acute HIV infection anytime in prior 4 weeks Retest antibody in 1 month; defer PrEP decision HIV- Eligible for PrEP HIV+ Not eligible for PrEP HIV status unclear Defer PrEP decision No Option 1 Option 3 Link to care and treatment Consider HIV + (pending confirmatory testing) Yes HIV+ VL 3,000 copies/mL VL 3,000 copies/mL HIV- VL level of detection No signs/symptoms on day of blood draw VL level of detection Signs/symptoms on day of blood draw Retest in 1 month Defer PrEP decision Send blood for HIV-1 VL assay HIV immunoassay blood test Negative Indeterminate HIV+ Option 2 (preferred) HIV- Send blood for HIV antibody/ antigen assaya Positive Negative Positive? HIV Status Algorithm1 Retest VL Defer PrEP decision
- 6. Patient Assessment: Before and After Initiating PrEP Access the activity, “Creating an HIV Prevention–Certified Provider Workforce: A Training and Certificate Program Designed to Improve the Competencies of Providers in Delivering Comprehensive HIV Prevention,” at PeerView.com/BDE40. PRACTICE AID a Use only HIV/antigen tests that are approved by FDA for diagnostic purposes. HIV: human immunodeficiency virus; PrEP: pre-exposure prophylaxis; STI: sexually transmitted infections; VL: viral load. 1. https://www.cdc.gov/stophivtogether/library/prescribe-hiv-prevention/brochures/cdc-lsht-php-brochure-prep-faq.pdf. Accessed October 14, 2019. 2. www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2017.pdf. Accessed October 14, 2019. • Assess adherence • Provide risk-reduction counseling • Offer condoms • Manage side effects At every visit • HIV testing (fingerstick or other blood test) STI symptom screen and testing For male patients • Nucleic Acid Amplification Test (NAAT) to screen for gonorrhea and chlamydia, based on exposure site • Test for syphilis For female patients • Nucleic Acid Amplification Test (NAAT) to screen for gonorrhea, based on exposure site • Test for syphilis • Serum creatinine and calculated creatinine clearance • Pregnancy testing • Every 3 months and • Whenever there are symptoms of acute infection Symptom screen • At every visit Testing • Every 3 months for sexually active persons with symptoms • Every 3 months for asymptomatic MSM at high risk for recurrent STIs (eg, those with recent STIs or multiple sex partners) • Every 6 months for sexually active persons, even if asymptomatic • Whenever symptoms are reported • At 3 months after initiation, and then every 6 months • Every 3 months CDC Guidelines: Monitoring While on PrEP2 Prevention and Medication Support Laboratory Testing Frequency Frequency
- 7. Information for Patients: Effectiveness of Prevention Strategies to Reduce the Risk of Acquiring or Transmitting HIV1 Access the activity, “Creating an HIV Prevention–Certified Provider Workforce: A Training and Certificate Program Designed to Improve the Competencies of Providers in Delivering Comprehensive HIV Prevention,” at PeerView.com/BDE40. PRACTICE AID a Optimal use of oral daily PrEP is defined as taking PrEP daily. In studies, optimal or daily PrEP use has been determined by levels of TFV-DP detected in dried blood spots equivalent to seven pills/week. Consistent use is defined as taking PrEP at least four pills/week and has been measured in studies by levels of TFV-DP detected in dried blood spots or other objective adherence measures, consistent with at least four pills/week. HIV: human immunodeficiency virus; TFV-DP: tenofovir-diphosphate. 1. https://www.cdc.gov/hiv/risk/estimates/preventionstrategies.html. Accessed October 14, 2019. Oral Daily Pre-Exposure Prophylaxis (PrEP) for HIV-Negative Persons Population Effectiveness Estimate “Optimal or Consistent Use” (Taking PrEP daily or at least 4 times per week)a Men who have sex with men (MSM) ~99% Heterosexual men and women ~99% Persons who inject drugs (PWIDs) 74% to 84% Antiretroviral Therapy (ART) for HIV-Positive Persons to Prevent Sexual Transmission Population Effectiveness Estimate “Optimal Use” (Taking ART daily as prescribed and achieving and maintaining viral suppression) Heterosexual men and women 100% Men who have sex with men (MSM) 100% Circumcision of Adult Males Population Effectiveness Estimate MSM, insertive anal sex Inconclusive MSM, receptive anal sex Inconclusive Heterosexual men 50% Heterosexual women Inconclusive Male Condom Use Population Effectiveness Estimate “Optimal Use” (Used consistently and correctly during every sex act) MSM or heterosexual men and women N/A “Consistent Use” (Always used during sex per self-report) Heterosexual men and women 80% MSM, receptive anal sex 72% to 91% MSM, insertive anal sex 63%
- 8. CDC Guidance Follow-Up and Monitoring Follow-Up Evaluate for All Persons Receiving PrEP for HIV Visit Checklist At least every 3 months • HIV testing (4th generation p24 Ag/Ab test) • Clinical visit and medication adherence assessment and counseling • Side-effect assessment • Behavioral risk reduction review and support • 90-day supply (TDF/FTC or TAF/FTC) • Pregnancy testing • Answer any new questions ❑HIV test ❑Assess for acute infection ❑Check for side effects ❑Pregnancy testing ❑Prescribe 90-day supply of medication At least every 6 months • Assess renal function by serum creatinine (estimated CrCl) • Test for bacterial STIs (three sites for chlamydia and gonorrhea; syphilis); every 3 months in MSM ❑HIV test ❑STI tests ❑Pregnancy testing ❑Renal function ❑90-day prescription Every 9 months • Test for bacterial STIs (three sites for chlamydia and gonorrhea; syphilis); every 3 months in MSM ❑HIV test ❑Pregnancy test ❑90-day prescription At least every 12 months • Evaluate need to continue PrEP ❑HIV test ❑STI tests ❑Pregnancy testing ❑Renal function ❑90-day prescription ❑Assess the need to continue PrEP Many experts recommend more frequent follow-up (ie, monthly) of patients on PrEP (ie, TDF/FTC or TAF/FTC) to assess adherence and monitor for STIs, including HIV.
- 9. Pre-Exposure Prophylaxis (PrEP) Myths and Urban Legends • PrEP will protect me from HIV and other STDs. • PrEP will lower my gender-affirming hormone levels and interfere with my gender expression. • Studies in Africa have shown that PrEP doesn’t work for heterosexual cis-gender women. • Studies have shown that PrEP does not work for transgender women. • PrEP causes serious liver and kidney disease. • As long as I take at least half of my PrEP tablets, I will be protected from HIV. • My doctor told me that I can take my first tablet of PrEP this morning and be protected from HIV this evening. • Since U=U is true for PLWH taking ART, and PrEP is 97% effective in preventing HIV acquisition, there’s no reason to use condoms anymore. • The increasing use of PrEP is directly responsible for the increasing numbers of STIs in the US. • Taking PrEP will not only protect me from HIV, but it will also prevent pregnancy. • If I start taking PrEP, I can never stop taking it, because if I acquire HIV it will be resistant to all antiretroviral medications. • Taking PrEP makes men and women more sexually promiscuous. • For cis-gender women like me, the best way to prevent HIV acquisition is to use PrEP gel inserted into my vagina before and after I have sex with a man. • If the PrEP tablets cause me to have side effects like nausea, I can always insert the tablet into my rectum (or vagina) before I have sex to protect me from acquiring HIV infection. • The cost of PrEP is very high and I do not have medical insurance, so there is no way for me to access PrEP. • The FDA recently approved a new type of PrEP which not only prevents HIV acquisition more than the old PrEP, but it will not harm my kidneys or bones. • The new PrEP medication approved by the FDA does not work in cis-gender women, which is why the FDA rejected it for cis-gender women who have vaginal sex. • HIV resistance to PrEP is increasing rapidly because many persons taking PrEP are not adherent with their medications. • There is a new way to take PrEP developed in France which is now FDA-approved in the US. It is called “as-needed” PrEP and is taken a couple of hours before sex and immediately after sex. • If I only have sex once or twice a month, my risk of acquiring HIV is too low to need PrEP. Key PrEP: Pre-exposure prophylaxis U=U: Undetectable=Untransmittable PLWH: People living with HIV ART: Antiretroviral therapy STI: Sexually transmitted infection
