Get Medical Device Approval in Ukraine
•
1 like•14,314 views
1. To get a medical device approved in Ukraine, manufacturers must appoint an authorized representative in Ukraine who can be a distributor or representative office. The manufacturer then identifies the appropriate technical regulations for the device and chooses a conformity assessment procedure. 2. The manufacturer prepares a technical file demonstrating compliance with the requirements in the chosen regulations. They then select a Notified Body to review the file and certify compliance with quality standards. 3. Upon approval, the authorized representative's name and address and the Ukrainian conformity assessment mark must be placed on the device labels and packaging.
Report
Share
Report
Share
Download to read offline
Recommended
mock 510(k) for UCSC Extension Regulatory Submissions Devices and Diagnostics...

mock 510(k) for UCSC Extension Regulatory Submissions Devices and Diagnostics...Joanne Pelaschier, RAC, CQA, CQE
Recommended
mock 510(k) for UCSC Extension Regulatory Submissions Devices and Diagnostics...

mock 510(k) for UCSC Extension Regulatory Submissions Devices and Diagnostics...Joanne Pelaschier, RAC, CQA, CQE
More Related Content
Similar to Get Medical Device Approval in Ukraine
Similar to Get Medical Device Approval in Ukraine (20)
Medical device as per india and usa special reference with 510(k)

Medical device as per india and usa special reference with 510(k)
Procedure for getting the manufacturing license of notified IVDs Products in ...

Procedure for getting the manufacturing license of notified IVDs Products in ...
Conduction of a remote audit in Ukraine. How is it organized?

Conduction of a remote audit in Ukraine. How is it organized?
Clinical study approval process in the Russian Federation

Clinical study approval process in the Russian Federation
Medical devices: 7 steps to CE-mark, and post-market surveillance

Medical devices: 7 steps to CE-mark, and post-market surveillance
More from Alexey Stepanov
More from Alexey Stepanov (12)
Eurasian integration alexey stepanov-informa v20150619

Eurasian integration alexey stepanov-informa v20150619
Kazakhstan: What information is mandatory on medical device label?

Kazakhstan: What information is mandatory on medical device label?
5 questions on safety reporting for medical devices in CIS region

5 questions on safety reporting for medical devices in CIS region
How to provide proper photos for medical device registration dossier? 

How to provide proper photos for medical device registration dossier?
How Can You Group Medical Devices for Registration in Russia? 

How Can You Group Medical Devices for Registration in Russia?
Recently uploaded
Model Call Girl in Subhash Nagar Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Subhash Nagar Delhi reach out to us at 🔝9953056974🔝9953056974 Low Rate Call Girls In Saket, Delhi NCR
Russian Call Girls Gurgaon Swara 9711199012 Independent Escort Service Gurgaon

Russian Call Girls Gurgaon Swara 9711199012 Independent Escort Service GurgaonCall Girls Service Gurgaon
Hot Call Girl In Chandigarh 👅🥵 9053'900678 Call Girls Service In Chandigarh

Hot Call Girl In Chandigarh 👅🥵 9053'900678 Call Girls Service In ChandigarhVip call girls In Chandigarh
Russian Call Girls in Dehradun Komal 🔝 7001305949 🔝 📍 Independent Escort Serv...

Russian Call Girls in Dehradun Komal 🔝 7001305949 🔝 📍 Independent Escort Serv...Call Girls Lucknow Aaradhya Independent Escort Service Lucknow
Hot Call Girl In Ludhiana 👅🥵 9053'900678 Call Girls Service In Ludhiana

Hot Call Girl In Ludhiana 👅🥵 9053'900678 Call Girls Service In LudhianaRussian Call Girls in Ludhiana
College Call Girls Dehradun Kavya 🔝 7001305949 🔝 📍 Independent Escort Service...

College Call Girls Dehradun Kavya 🔝 7001305949 🔝 📍 Independent Escort Service...Call Girls Lucknow Aaradhya Independent Escort Service Lucknow
indian Call Girl Panchkula ❤️🍑 9907093804 Low Rate Call Girls Ludhiana Tulsi

indian Call Girl Panchkula ❤️🍑 9907093804 Low Rate Call Girls Ludhiana TulsiHigh Profile Call Girls Chandigarh Aarushi
Russian Call Girls in Chandigarh Ojaswi ❤️🍑 9907093804 👄🫦 Independent Escort ...

Russian Call Girls in Chandigarh Ojaswi ❤️🍑 9907093804 👄🫦 Independent Escort ...High Profile Call Girls Chandigarh Aarushi
Recently uploaded (20)
Call Now ☎ 9999965857 !! Call Girls in Hauz Khas Escort Service Delhi N.C.R.

Call Now ☎ 9999965857 !! Call Girls in Hauz Khas Escort Service Delhi N.C.R.
Model Call Girl in Subhash Nagar Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Subhash Nagar Delhi reach out to us at 🔝9953056974🔝
Russian Call Girls Gurgaon Swara 9711199012 Independent Escort Service Gurgaon

Russian Call Girls Gurgaon Swara 9711199012 Independent Escort Service Gurgaon
Hot Call Girl In Chandigarh 👅🥵 9053'900678 Call Girls Service In Chandigarh

Hot Call Girl In Chandigarh 👅🥵 9053'900678 Call Girls Service In Chandigarh
Russian Call Girls in Dehradun Komal 🔝 7001305949 🔝 📍 Independent Escort Serv...

Russian Call Girls in Dehradun Komal 🔝 7001305949 🔝 📍 Independent Escort Serv...
❤️♀️@ Jaipur Call Girls ❤️♀️@ Meghna Jaipur Call Girls Number CRTHNR Call G...

❤️♀️@ Jaipur Call Girls ❤️♀️@ Meghna Jaipur Call Girls Number CRTHNR Call G...
Leading transformational change: inner and outer skills

Leading transformational change: inner and outer skills
Hot Call Girl In Ludhiana 👅🥵 9053'900678 Call Girls Service In Ludhiana

Hot Call Girl In Ludhiana 👅🥵 9053'900678 Call Girls Service In Ludhiana
Call Girl Raipur 📲 9999965857 ヅ10k NiGhT Call Girls In Raipur

Call Girl Raipur 📲 9999965857 ヅ10k NiGhT Call Girls In Raipur
Dehradun Call Girls Service 8854095900 Real Russian Girls Looking Models

Dehradun Call Girls Service 8854095900 Real Russian Girls Looking Models
College Call Girls Dehradun Kavya 🔝 7001305949 🔝 📍 Independent Escort Service...

College Call Girls Dehradun Kavya 🔝 7001305949 🔝 📍 Independent Escort Service...
Udaipur Call Girls 📲 9999965857 Call Girl in Udaipur

Udaipur Call Girls 📲 9999965857 Call Girl in Udaipur
VIP Call Girl Sector 25 Gurgaon Just Call Me 9899900591

VIP Call Girl Sector 25 Gurgaon Just Call Me 9899900591
💚😋Chandigarh Escort Service Call Girls, ₹5000 To 25K With AC💚😋

💚😋Chandigarh Escort Service Call Girls, ₹5000 To 25K With AC💚😋
💚😋Kolkata Escort Service Call Girls, ₹5000 To 25K With AC💚😋

💚😋Kolkata Escort Service Call Girls, ₹5000 To 25K With AC💚😋
💚😋Mumbai Escort Service Call Girls, ₹5000 To 25K With AC💚😋

💚😋Mumbai Escort Service Call Girls, ₹5000 To 25K With AC💚😋
Call Girl In Zirakpur ❤️♀️@ 9988299661 Zirakpur Call Girls Near Me ❤️♀️@ Sexy...

Call Girl In Zirakpur ❤️♀️@ 9988299661 Zirakpur Call Girls Near Me ❤️♀️@ Sexy...
pOOJA sexy Call Girls In Sector 49,9999965857 Young Female Escorts Service In...

pOOJA sexy Call Girls In Sector 49,9999965857 Young Female Escorts Service In...
indian Call Girl Panchkula ❤️🍑 9907093804 Low Rate Call Girls Ludhiana Tulsi

indian Call Girl Panchkula ❤️🍑 9907093804 Low Rate Call Girls Ludhiana Tulsi
Russian Call Girls in Chandigarh Ojaswi ❤️🍑 9907093804 👄🫦 Independent Escort ...

Russian Call Girls in Chandigarh Ojaswi ❤️🍑 9907093804 👄🫦 Independent Escort ...
Get Medical Device Approval in Ukraine
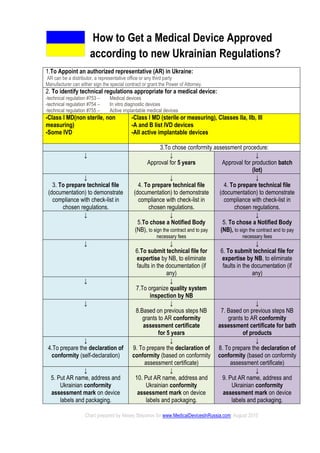
- 1. How to Get a Medical Device Approved according to new Ukrainian Regulations? 1.To Appoint an authorized representative (AR) in Ukraine: AR can be a distributor, a representative office or any third party Manufacturer can either sign the special contract or grant the Power of Attorney. 2. To identify technical regulations appropriate for a medical device: -technical regulation #753 – Medical devices -technical regulation #754 – In vitro diagnostic devices -technical regulation #755 – Active implantable medical devices -Class I MD(non sterile, non measuring) -Some IVD -Class I MD (sterile or measuring), Classes IIa, IIb, III -A and B list IVD devices -All active implantable devices 3.To chose conformity assessment procedure: ↓ ↓ Approval for 5 years ↓ Approval for production batch (lot) ↓ 3. To prepare technical file (documentation) to demonstrate compliance with check-list in chosen regulations. ↓ 4. To prepare technical file (documentation) to demonstrate compliance with check-list in chosen regulations. ↓ 4. To prepare technical file (documentation) to demonstrate compliance with check-list in chosen regulations. ↓ ↓ 5.To chose a Notified Body (NB), to sign the contract and to pay necessary fees ↓ 5. To chose a Notified Body (NB), to sign the contract and to pay necessary fees ↓ ↓ 6.To submit technical file for expertise by NB, to eliminate faults in the documentation (if any) ↓ 6. To submit technical file for expertise by NB, to eliminate faults in the documentation (if any) ↓ ↓ 7.To organize quality system inspection by NB ↓ ↓ 8.Based on previous steps NB grants to AR conformity assessment certificate for 5 years ↓ 7. Based on previous steps NB grants to AR conformity assessment certificate for bath of products ↓ 4.To prepare the declaration of conformity (self-declaration) ↓ 9. To prepare the declaration of conformity (based on conformity assessment certificate) ↓ 8. To prepare the declaration of conformity (based on conformity assessment certificate) ↓ 5. Put AR name, address and Ukrainian conformity assessment mark on device labels and packaging. ↓ 10. Put AR name, address and Ukrainian conformity assessment mark on device labels and packaging. ↓ 9. Put AR name, address and Ukrainian conformity assessment mark on device labels and packaging. Chart prepared by Alexey Stepanov for www.MedicalDevicesInRussia.com, August 2015
